Cardiac
Incidence
Up to 25% on autopsy but clinically apparent in 5% of sarcoid patients [1,2].
Clinical presentation
Variable and range from no symptoms to arrhythmia,
heart block,
congestive cardiac failure,
angina,
ventricular aneurysm,
pericardial effusion or sudden death.
Imaging findings
Cardiac findings are rarely seen on CT,
with myocardial thinning at the site of disease the only potential sign [2]; however,
extra-cardiac stigmata of sarcoidosis may be seen in the thorax.
Gated cardiac MR is the modality of choice with sites of disease demonstrating T2 hyperintesity and late gadolinium enhancement (LGE) following contrast administration,
reflecting myocardial scarring and/or inflammation (Fig. 1) [3].
The left ventricular free wall and septum are typically involved with localisation to the mid-myocardial wall or epicardium,
although,
transmural and/or subendocardial enhancement (mimicking myocardial infarction) may be seen [4].
A non-ischaemic pattern of distribution should raise suspicion for sarcoidosis,
especially in healthy or symptomatic young or middle-aged patients.
Pericardial effusion and impaired cardiac function are other potential findings.
PET can be used as an adjunct to cardiac MR with areas of abnormally increased FDG uptake in the myocardium +/- other extra-cardiac sites of disease (Fig. 2).
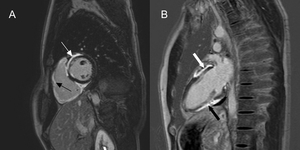
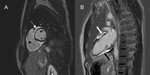
Fig. 1: Cardiac sarcoidosis in a 33 year-old man presenting with arrhythmia and no past medical history. Late-gadolinium cardiac MR in the ventricular short axis (A) and vertical long axis (B). Mid-wall and epicardial myocardial hyper-enhancement in the basal anterior, antero-septum and inferior segments of the left ventricle (A, narrow white arrow) and extensive enhancement of the right ventricle myocardium (A, narrow black arrow). Similar enhancement pattern in the anterior (B, thick white arrow) and inferior (B, thick black arrow) left ventricular walls. Note subendocardial sparing.
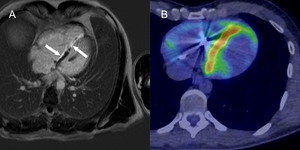
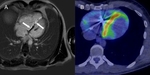
Fig. 2: Cardiac sarcoidosis in a young male with known pulmonary sarcoidosis. Late-gadolinium cardiac MR in the horizontal long axis (A) demonstrating septal hyper-enhancement (white arrows). Fused PET/CT demonstrating abnormal septal FDG uptake (B) in keeping with active myocardial inflammation.
Abdominal
Liver and Spleen
Incidence
50-80% on autopsy but clinically detected in 5-15% of sarcoid patients [2,5]
Clinical presentation
Typically asymptomatic and incidentally found.
Organomegaly,
abnormal liver function tests,
abdominal pain or pruritus may be present.
Imaging findings
Organomegaly may be the only manifestation but 5-15% of patients demonstrate multiple well-defined hypoechoic or low-attenuating nodules on ultrasound and CT respectively,
representing coalescing granulomas (Fig. 3) [5].
These are more frequently seen in the spleen than the liver,
where they are also typically larger [6].
The liver lesions represent a diagnostic challenge as they mimic metastases and lymphoma,
especially when features of pulmonary sarcoidosis are absent ( Fig. 4).
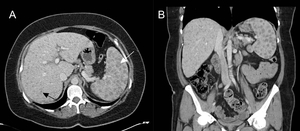
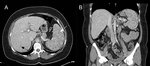
Fig. 3: Hepatic and splenic sarcoidosis in a patient with pulmonary sarcoidosis. Contrast-enhanced abdominal CT in axial (A) and coronal (B) planes demonstrating widespread low-attenuating nodules in the liver and spleen. Splenic nodules (white arrow) are typically larger than hepatic nodules (black arrow).
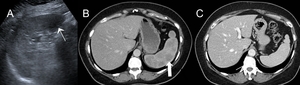
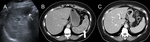
Fig. 4: Spleen involvement in a 56 year-old female with a previous history of breast carcinoma two years prior. Routine ultrasound demonstrating a hypoechoic splenic lesion (A, narrow white arrow) with additional low-attenuating splenic lesions demonstrated on contrast-enhanced CT (B, thick white arrow). Mediastinal and hilar lymphadenopathy was also demonstrated on CT, raising suspicion for malignant breast carcinoma recurrence. Splenic biopsy revealed granulomatous lesions and endobronchial ultrasound-guided fine-needle aspirations (EBUS-FNA) of mediastinal nodes were normal. A trial of steroid therapy was initiated, resulting in reduction in the size of mediastinal/hilar lymph nodes and resolution of the splenic nodules (C).
Gastrointestinal
Gastrointestinal tract sarcoidosis is rare (<1%) and often associated with pulmonary involvement,
with stomach the commonest site of disease [2].
Radiological findings are not typically apparent and may include ulceration,
mucosal thickening or features similar to linitis plastica [6].
Intra-abdominal lymphadenopathy may be present.
Urogenital
Incidence
Kidney and testicular sarcoidosis are extremely rare; both 5% incidence on autopsy [2].
Clinical presentation
Rarely any clinical or biochemical manifestation in kidney sarcoidosis.
Normally asymptomatic epididymitis in the scrotum but scrotal pain or mass may be present [2].
Imaging findings
Hypercalcaemia-induced nephrocalcinosis is the commonest renal manifestation.
Direct granulomatous involvement of the kidneys can cause glomerulonephritis and interstitial nephritis,
with the latter demonstrated as striated nephrograms on contrast-enhanced CT [2].
Rarely,
multiple low-attenuating renal nodules may be seen and differentiating these from renal malignancy is particularly challenging.
Ultrasound and MRI may demonstrate an enlarged epididymis (asymptomatic epididymitis) that can be bilateral in 33% of cases [4].
Single or multiple hypoechoic testicular nodules may be seen on ultrasound which cause a particular diagnostic challenge in the absence of a known history of sarcoid,
where malignancy needs to be ultimately excluded with further additional imaging and/or histological diagnosis (Fig. 5) [7]. Testicular nodules demonstrate T2 hypointensity on MRI and enhancement on T1 sequences following contrast administration [2].
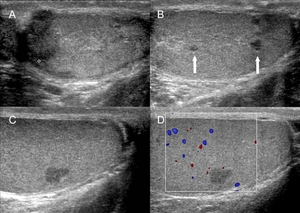
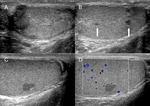
Fig. 5: Testicular sarcoidosis in a 50 year-old man presenting with testicular swelling and no scrotal pain or past medical history. Ultrasonography demonstrates mildly enlarged bilateral hypoechoic epididymides (A) and multiple focal hypoechoic nodules measuring up to 5 mm (B, arrows) with no evidence of hypervascularity (C, D). Further imaging revealed splenomegaly and bilateral nodular pulmonary consolidations atypical for sarcoidosis. Histological diagnosis was confirmed following orchidectomy.
Neurological systems
Incidence
Up to 25% on autopsy but clinically detected in <10% of patients [2,8]
Clinical presentation
Variable and dependent on site of disease.
Up to 75% of patients with neurosarcoidosis present with neurological symptoms ranging from headache to cranial nerve neuropathy (especially facial nerve palsy,
which when bilateral in young adults,
is strongly suggestive of sarcoidosis),
hypothalamic/pituitary endocrine dysfunction (e.g.
diabetes insipidus),
aseptic meningitis and psychiatric symptoms [9].
Imaging findings
Brain
Non-specific solitary or multiple T2 hyperintense periventricular and deep white matter lesions are demonstrated in up to 60% of neurosarcoidosis cases,
thought to reflect spread along perivascular spaces (Fig. 6) [4].
These enhance on contrast-enhanced imaging.
Mass-like lesions have also been described which may show a ring-like appearance and enhancement following contrast administration (Fig. 7 and Fig. 8) [2].
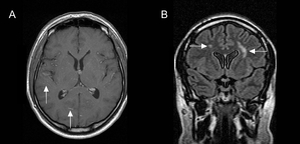
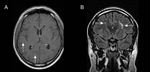
Fig. 6: Parenchymal neurosarcoidosis in a patient presenting with headaches. Patchy high signal is demonstrated in the deep white matter on post-gadolinium T1-weighted (A) and fluid-attenuation inversion-recovery (FLAIR; B) MRI (arrows).
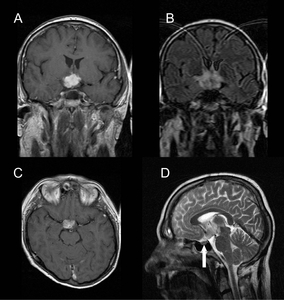
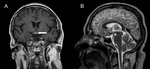
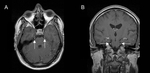
Fig. 7: Neurosarcoidosis in a 37 year-old male with deterioration in visual fields. Large avidly enhancing lobulated suprasellar mass involving the optic chiasm and hypothalamus on post-gadolinium T1-weighted (A, C), fluid-attenuation inversion-recovery (FLAIR; B) and T2-weighted (D) sequences. The pituitary gland is normal within a non-expanded sella (D, arrow). Pulmonary sarcoidosis was demonstrated on HRCT and steroid treatment initiated.
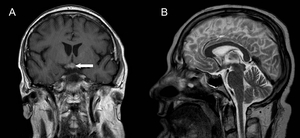
Fig. 8: Post-steroid treatment post-gadolinium T1-weighted (A) and T2-weighted MRI (B) of the neurosarcoidosis patient in Figure 7. Reduction in the size of the hypothalamic/chiasmic sarcoid nodule with a residual focal enhancing nodule (arrow) is seen.
Cranial nerve involvement,
most commonly the facial nerve,
are seen in up to 30% of patients,
demonstrated as a “tram-line” pattern of leptomeningeal enhancement on T1 contrast-enhanced sequences and hyperintensity on fluid-attenuation inversion-recovery (FLAIR) (Fig. 9) [9,10].
The optic nerve can be involved anywhere along its course.
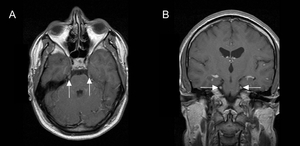
Fig. 9: Neurosarcoidosis with involvement of thickened and enhancing trigeminal nerves on post-gadolinium T1-weighted MRI (arrows).
The hypothalamic-pituitary axis is affected in 5-10% of cases with radiological signs including thickening of the infundibulum,
cystic pituitary lesions,
glandular enlargement and contrast enhancement (Fig. 10) [9,10].
Loss of posterior pituitary T1 hyperintensity in diabetes insipidus may be seen secondary to depletion of intracellular neurosecretory granules [2].
Dural involvement is possible,
even in the form of dural-based extra-axial lesions mimicking malignancy or meningiomas [4,9].
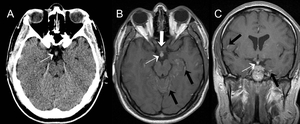
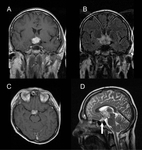
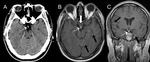
Fig. 10: Neurosarcoidosis in a 28 year-old male patient presenting with dizziness and headaches. CT demonstrates a thickened pituitary infundibulum (A, narrow white arrow). A thickened enhancing pituitary infundibulum is confirmed on post-gadolinium T1-weighted MRI (B and C, narrow white arrow), along with a thickened optic chiasm (B, thick white arrow). Diffuse nodular leptomeningeal and white matter enhancement also demonstrated (black arrows).
Meninges
Diffuse or nodular leptomeningeal enhancement is seen in up to 40% of cases with a predilection for basal meninges (Fig. 10 and Fig. 11) [10].
This can mimic granulomatous meningitis including tuberculosis or carcinomatous meningitis [2].
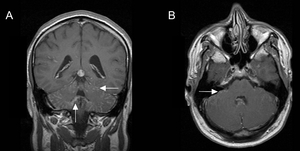
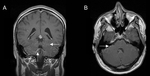
Fig. 11: Diffuse nodular leptomeningeal neurosarcoidosis along the cerebellar folia and brainstem (arrows) on post-gadolinium T1-weighted MRI.
Ocular
Ocular involvement is very common (up to 80%) and typically seen as bilateral uveitis [11].
Bilateral enlarged enhancing lacrimal glands may be seen radiologically on CT or MRI [2].
Spinal cord
Solitary or diffuse enhancing intramedullary lesion(s) with or without cord oedema [4].
Linear or nodular leptomeningeal disease may be seen on the cord surface.
Musculoskeletal
Incidence
Uncommon with reported incidence ranging from 1 to 25% [2,4,12].
Clinical presentation
Arthralgia,
acute arthritis (frequently as Löfgren’s syndrome: bilateral hilar adenopathy,
erythema nodosum and bilateral ankle swelling),
chronic arthritis (dactylitis) or myalgia.
Imaging findings
Radiological findings are variable.
Bone radiographs can demonstrate a reticular,
lacy pattern and isolated or multiple cystic lesions,
with or without malalignment deformities [12].
Resorption of the distal phalanges (acro-osteolysis) secondary to granulomatous inflammations have a “punched-out” appearance [12].
Cutaneous involvement may be seen as sausage-like dactylitis.
MR appearances of bone involvement is highly variable,
ranging from well-defined intramedullary lesions to irregular marrow infiltration; these are typically T2 or short-tau inversion recovery (STIR) hyperintense and T1 hypointense [4].
Symptomatic muscle involvement is extremely rare (0.5%) with acute and chronic subtypes demonstrating non-specific inflammation,
muscle atrophy or no radiological signs at all [4].
The nodular subtype is the most challenging as it can be mistaken for soft tissue tumours.
Muscular sarcoid nodules are typically bilateral,
multifocal and seen at myotendinous junctions [13].
They are hypointense centrally on all MR sequences and peripherally hyperintense on T2 and following gadolinium administration [2,13].
Cutaneous
Not subject to radiological imaging,
cutaneous involvement is common (up to 25%),
with erythema nodosum a feature of acute sarcoidosis,
and lupus pernio a feature of chronic sarcoidosis and pulmonary fibrosis [11].