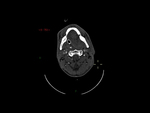
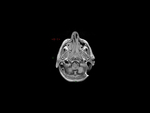
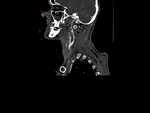
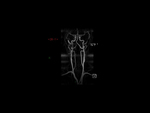
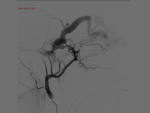
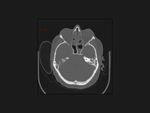
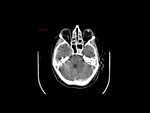
Our CT study protocol includes basal spiral acquisition of brain and neck (also with bone HR) and MPR reconstructions of the cervical tract. The examination is completed with TCA study of the apiaortic and intracranial vessels with biphasic technique (venous afetr 40 sec for the study of collateral circulation) and MPR and MIP reconstructions. The MRI study protocol includes FLAIR-weighted mutiplanar standard T1W-T2W sequences,
GE and DWI for the study of the brain that can be completed with MRA sequences for the study of arterial and venous vessels.
Arterial MRA sequences: without contrast medium (cm) for the study of arterial intracranial vessels (TOF),
and with cm for the study of epiaortic vessels and Willis circulation,
with CE Care bolus technique and 3D MIP reconstructions. In case of study of venous sinuses,
it is preferred to use a technique with cm (CE MR venography) to the less invasive ones without cm like 2D TOF or 3D Phase Contrast.
Regarding the arterial lesions,
and remaining faithful to the aforementioned radiological classification (tab 3),
we can make a first distinction between ischemic and hemorrhagic lesions.
Ischemic
Laceraction of the intimal layer of vessel wall,
with dissection flap and formation of an intramural hematoma,
that can evolve in the stenosis of the lumen itself until its complete occlusion.
However,
there is a further radiological presentation for different evolution of the lesion,
that is the formation of a pseudoaneurysm.
See table 4 and 5.
Tab 4.
Direct findings-arterial injuries
|
NECT
|
CTA
|
MRI-MRA
|
- Vessel wall haematoma in the involved portion of the ICA: spontaneus crescent-shaped hyperattenuating focus
|
- Narrowed eccentric lumen surrounded by a crescent-shaped mural thrombus and thin annular enhancement (vasa vasorum of the adventitia)
- Intimal flap and "double lumen" appearance
|
- Absent flow-void in ICA and VA segments (vs the healthy side)
- Hyperintense crescent sign (sickle-shaped hyperintensity) within the vessel wall (Fat saturated T1,
or T2 axial images through the neck and base of the skull)
- Abnormal vessel contour on MRA (stenosis,
pseudoaneurysm,
occlusion)
|
As for vertebral artery (VA),
dissections are mostly located in the segment V2 (~35%) and V3 (~34%).
An important feature is to assess if the dissection involves the intradural portion V4,
and thus the origin of the posterior inferior cerebellar artery.
As for the ICA,
the most exposed to traumatic injuries are C1-C2 segments.
This depends on the greater motility of these segments respect to the "fixed" tracts in the context of bone structures (high vulnerability to torsional forces).
The ICA,
like the VA,
have different distribution of elastic fibres compared to similar sized vessels elsewhere. Although the tunica media and tunica adventitia are present they are only a third as thick as their extracranial counterparts,
with the vast majority of elastic fibres located in a subendothelial elastic lamina.
This fundamental difference accounts for the markedly different natural history of intracranial arterial dissections compared to the extracranial ones.
When a tear breaches the aforementioned subendothelial elastic layer,
then there is little tissue preventing extension into the subarachnoid space,
thus accounting for the very high rate of subarachnoid haemorrhage.
Tab 5.
Indirect findings-arterial injuries
|
NECT
|
CTA
|
MRI-MRA
|
- Early ischaemic brain changes in anterior/posterior circulation's territories on CT an MRI images
- Complex skull fractures,
skull base fractures (including carotid canal ones),
cervical transverse process fractures
- Subarachnoid haemorrhage-SAH (V4 dissection)
|
-
|
- cerebral ischemia (hyperintense DWI-FLAIR)
- Hemorrhagic foci (hypointense GE)
|
Hemorrhagic
Complete tearing of the vessel wall,
accompanied by intraparenchymal (brain) or perivasal hematoma in the soft tissue contest (neck).
After administration of contrast medium,
the blush correspond to the presence of an active bleeding.
Based on the affected compartment,
we recognize: epidural,
subdural,
intraparenchymal hematomas,
subarachnoid haemorrhage,
laterocervical hematomas.
Carotid cavernous fistulae are also included among hemorrhagic lesions.
Carotid cavernous fistulas (CCF) are a subtype of artero-venous fistula, characterized by abnormal communications between arteries and veins located in the cavernous sinus (CS).
They are usually caused by severe head injuries or surgical traumas (2).
In penetrating trauma the laceration track crosses through the CS and cavernous segment of ICA.
In blunt trauma the CCF is the result of the transmission of shear stresses to the connective tissue fibers that connect cavernous segment of ICA to CS walls (for example central skull base fractures extended to CS region).
Patients who suffered severe facial traumas require a long follow-up because of direct CCF may be clinically and radiologically evident even several days after the trauma.
FCC rarely heal spontaneusly,
and a delay in diagnosis and treatment increase the chance of irreversible neurologic ocular damage.
Instead,
early endovascular treatment can improve clinical outcome.
The pathological comunication between arterial and venous system implicates a modification of the normal flow (speed and volume especially in venous compartment of cavernous sinus and tributary veins),
and for this reason clinical manifestation are chemosis,
conjunctival bleeding,
diplopia,
eyelid swelling,
intense proptosis and vision loss,
for oftalmic venous system cranial and nerve palsis (III,
IV,
Vc ,VI inside the CS) involvement.
Radiological findings of CCF are presented in the Tab 6.
Tab 6.CCF
|
CT/CTA
MRI/MRA
|
- proptosis
- orbital oedema
- enlargment of extraocular muscles (MR or LR greater than 4mm)
- SAH/ICH (rupture of cortical vein)
- fracture lines (CT)
- enlarged superior ophtalmic veins (>4mm)
- full-thickness defect within CS (abnormal arterialization of the flow)
- lateral bulging of CS,
asymmetrical and early enhancement vs contralateral one
- enlargment and asymmetrical enhancement in sphenoparietal sinus,
inferior petrosal sinus and superior petrosal sinus
|
|
DSA
|
- high flow shunt from cavernous ICA to CS
- retrograde flow from CS,
most commonly into the ophtalmic veins
- enlargement of draining veins
|
Venous injury may occur both after a penetrating or a blunt trauma.
Generally the involvement of the venous system is more common in penetrating injuries.
In penetrating trauma,
the wound results in a venous sinus mural distruption and epidural hematomas,
because of it crossing the site of dural sinus attachment.
The same extra axial hemorrhagic lesion may occur in case of a skull fracture crossing the dural attachment of the sinus itself.
In blunt injury the mechanism of the trauma lead to a venous thrombosis (whether the sinus is crossed by a fracture or not).
About 7% of patients with venous thrombosis may experience venous infarction.
(9)
For this reason in patients with fractures of the skullbase in a NCE-CT after a trauma,
a slight hyper-density near the course of the venous sinuses must make suspect a focal post-traumatic thrombosis,
and eventually a second diagnostic step with a CTA.
A delayed acquisition after the first arterial phase of CTA may show a focal full-thickness defect within the dural sinus involved,
maybe neighbouring a fracture.
DSA findings and treatment possibilities
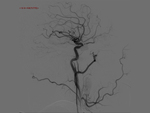
Angiography is still considered the gold standard for imaging cerebrovascular anatomy.
Modern technology and further technical refinements (rotational angiography,
3D angiography) made this technique able to perform more efficiently imaging.
DSA provides spatial and temporal resolution to evaluate dynamic and functional anatomy of cerebral circulation.
Biplane angiography (flat panel detectors) allows for orthogonal images to be simultaneously obtained,
with a single contrast injection,
limiting the time,
contrast,
radiations.
Angiographic images are acquired through low osmolality,
nonionic cm (less allergenic) selective injection.
DSA is an invasive technique. Its risks include stroke,
renal issues,
access site hematomas,
and others.
Complication are about the 1%.
(3)
High-grade dissection with suspicion of inadequate collaterals based on CT or MR techniques,
or in inconclusive CTA cases,
may be an indication to DSA,
to complete the study of the lesion,
and plan the best endovascular approach.
(3)
In his review,
Sianou suggests the following criteria for stenting: (3)
-patients with recurrent symptoms despite medical therapy,
-patients with hemodynamic hypoperfusion (involvement of multiple vessels or poor collateral vessels), patients with expanding or symptomatic pseudoaneurysm and contraindication to anticoagulation because of intracranial or systemic hemorrhage.
(11)
Stenting procedures need appropriate pre- and postprocedural antiplatelet therapy,
to guarantee patency of the stent and the target vessel itself.
When the patient presents other concomitant injuries with a high risk for severe bleeding,
the approach must be evaluated by a interdisciplinary team to identify optimal type and time of treatment.
(3)
Embolization therapy doesn’t need pharmacological premedication.
It is the first choice in patient,
with active bleeding.
Other purpose of this minimally invasive technique is to secure a impaired vessel,
also if not actively bleeding.
The neurointerventionalist has many arrows in his quiver.
The approach depends on case by case evaluation,
and basically on two different aims,
the first referred to the “kind” of occlusion to achieve,
and second the dimensions of the target vessel.
See the table 7.
Tab7.
Endovascular approaches
|
Vascular lesions (DSA/CTA aspect)
|
Endovascular approach
|
| Intimal irregularity with <25% narrowing |
Medical approach is preferred
|
|
Dissection with intramural hematoma and >25% narrowing (if not responding to medical therapy,
or in patient symptomatic)
|
Stenting
|
|
Pseudoaneurysm (if expanding or symptomatic)
|
Stenting with vessel wall reparation (ICA)
Embolization with vessel occlusion (ECA)
|
|
Symptomatic occlusion (acute)
|
Recanalization +/- stenting
|
|
Arterio-venous fistula
|
Embolization (coils)
|
|
Transection with extravasation
|
Embolization (coils or plug for large vessels,
other embolic agents for small vessels)
|