INTRODUCTION
Definition
Schistosomiasis (ie,
bilharziasis) is a human disease syndrome caused by infection from one of several species of parasitic trematodes of the genus Schistosoma.
Epidemiology
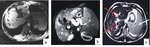
According to the World Health Organization,
schistosomiasis is a millenary disease,
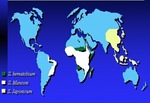
which affects more than 200 million individuals distributed in 76 countries in Africa,
Asia or America (Fig.
1).
Among these,
10% present the severe form of disease and 50 to 60% of infected people,
that is more than 100 million people,
present clinical manifestations of the disease,
constituting a huge public health problem (WHO 1993).
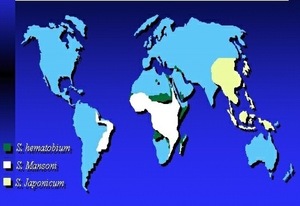
Fig. 1: Global distribuition of schistosomiasis.
References: internet
Historic background
Although discovered only in 1851,
by Theodor Bilharz (Fig.
2),
who confirmed the presence of the worm known today as Schistosoma hematobium in mesenteric vessels of an autopsied Egyptian peasant,
Shistosoma eggs were found in Egyptian mummies dating from 3500 b.C. In 1902,
Manson found eggs with lateral spine,
in patients from Antilles,
admitting a new species of Schistosoma,
which was classified as S.
mansoni, by Sambon,
in 1907.
This same species had its presence confirmed in Brazil,
through the description of four cases,
the first ones registered by Pirajá da Silva,
in Bahia,
in 1908.

Fig. 2: Theodor Maximillian Bilharz
References: internet
Schistosomiasis is considered to be the most important helminthic disease of man.In Brazil,
a total of 2.5 million individuals are estimated to be infected by Schistosoma mansoni,
and 25 million people are at risk to be infected,
representing one of our major public health challenges.
Subjects are affected in different manners and most infected individuals are asymptomatic carriers of the parasite.
The most serious form is the hepatosplenic one,
whose major complication is upper digestive hemorrhage secondary to portal hypertension.
In pure and uncomplicated cases,
hepatocellular function is usually preserved up to the more advanced phases of the disease.
PATHOPHYSIOLOGY
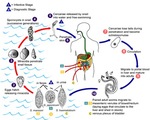
Life cycle (Fig.
3 and 4)
•Humans are the principal hosts for the major species
•Persistence of schistosomiasis depends on the presence of an appropriate snail as an intermediate host.
•Eggs excreted in stool (S mansoni and S japonicum) or urine (S haematobium) into fresh water hatch into motile miracidia,
which infect snails.
•After development in the snails,
cercariae emerge and penetrate the skin of humans encountered in the water.
•The cercariae pass through lungs to liver where they mature,
mate and pass down into mesenteric or vescical venules to begin egg production.
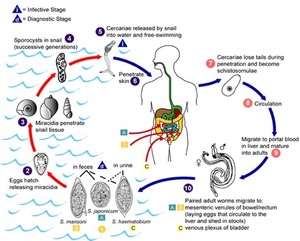
Fig. 3: Schistosomiasis mansoni. Life cycle.
References: internet
•Communicability lasts as long as live eggs are excreted in the urine and feces.
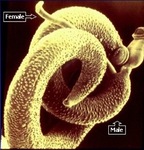
•Adult worms of the S mansoni species (Fig.
5) have been documented to live as long as 26 years in the human host.
•Thus,
schistosomiasis can be diagnosed in patients many years after they have left the endemic areas.
•Swimmer's itch occurs in all regions of the world after exposure to fresh,
brackish,
or salt water containing larvae that do not complete their life cycle in humans.
•The incubation period is variable but is approximately 8 weeks for S haematobium and 4 weeks for S mansoni and S japonicum.
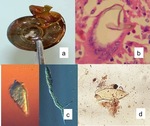
Fig. 4: Other forms of Schistosomes (a) Snail, (b) HISTOLOGY: Schistosoma mansoni egg, (c) Miracidium and cercaria and (d) Schistosoma mansoni eggs.
References: internet
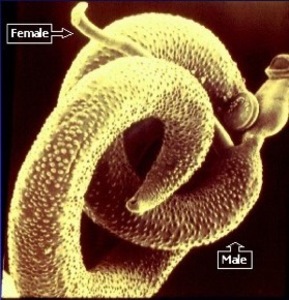
Fig. 5: Adult Schistosomes in copulation.
References: internet
The evolutionary history of schistosomotic patients comprises five major steps : 1) an appreciable contamination of the Schistosoma mansoni; 2) the fixation of live adult worms within the roots of the mesenteric veins; 3) the migration of the eggs deposited by the females and of dead worms towards the intra-hepatic portal roots in the liver,
causing reactions both inflammatory and scarring,
thus constituting Symmers fibrosis; 4) the development of portal hypertension; 5) the formation of esophageal varices,
hypertensive gastropathy and colopathy,
as well as varices in other organs.
ACUTE SCHISTOSOMIASIS MANSONI
The acute phase of schistosomiasis is usually asymptomatic but clinical signs of varying intensity may occur.
Those living in endemic areas may develop the syndrome after having contact for the first time with water contaminated by Schistosoma mansoni cercariae.
The most common manifestations are fever,
chills,
weakness,
weight loss,
headache,
nausea,
vomiting,
diarrhea,
hepatomegaly,
splenomegaly and marked eosinophilia.
Patients with acute schistosomiasis mansoni have been diagnosed at times as having typhoid fever,
hepatitis,
pancreatitis and appendicitis,
only to be cured when infection with schistosomiasis has been discovered after a long search.
UNUSUAL PRESENTATIONS OF ACUTE SCHISTOSOMIASIS MANSONI
The toxaemic form of schistosomiasis mansoni is considered an unusual clinical manifestation of the disease especially in endemic areas for this parasitosis.
This is due to the frequent tendency of schistosomiasis to exhibit a clinically non-apparent form in its early phase or to present clinical features similar to those of several infectious or non-infectious diseases prevalent in endemic areas for this helminthiasis.
CHRONIC SCHISTOSOMIASIS MANSONI
TOPIC FORMS
INTESTINAL (Fig.
6 and 7)
This is the most common form of chronic schistosomiasis.
The intestinal form is manifested more frequently by bowel irregularity of little relevance and the great majority of colitis caused by schistosomiasis mansoni is asymptomatic or oligosymptomatic.
It usually involves the rectal and sigmoid colons.
Some patients may present colon stenosis and intestinal obstruction.
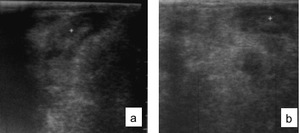
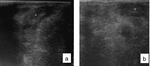
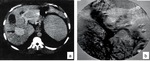
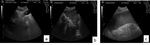
Fig. 6: Ultrasound showing thickening of the sigmoid (a) and descending colon (b) related to granuloma caused by schistosomiasis mansoni in two different patients.
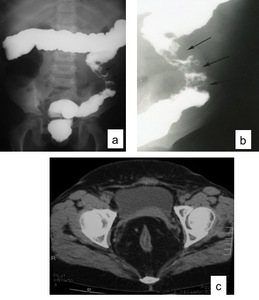
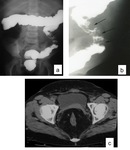
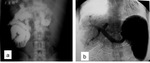
Fig. 7: Barium enema (a) and (b). Schistosomotic granuloma involving the descending colon in a 10 year-old boy presenting with bloody diarrhea. Pelvic CT showing abundant fat tissue around the rectum related to schistosomiasis (c).
HEPATOINTESTINAL
Depending on the worm burden,
immunological and nutritional factors of the patients as well as specific treatment,
they can develop the hepatointestinal form.
HEPATOSPLENIC (Fig.
8 and 9)
Hepatosplenic schistosomiasis(HS) is the most severe presentation of the disease,
which is characterized by extensive splenomegaly,
periportal fibrosis,
portal hypertension,
and upper digestive bleeding due to rupture of esophageal varices.
The principal deciding antigenic factor of hepatic fibrosis is the presence of the S.
mansoni eggs in the portal veins,
which causes a granulomatous inflammatory reaction leading to the formation of fibroid tissue.
During the sequence of events,
there occurs a subsequent intra-hepatic,
vascular neo-formation,
distributed around the portal ramifications that maintains the blood flow and preserves the hepatic cells.
It is for this reason that patients with schistosomiasis do not present hepatic insufficiency.
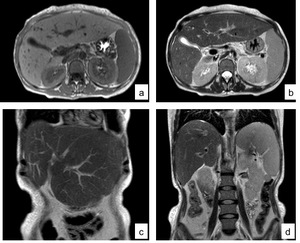
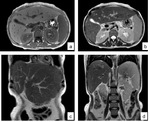
Fig. 8: MRI of the abdomen in a 43-year-old woman with hepatosplenic schistosomiasis. (a) Axial T1, (b) axial T2, (c) and (d) coronal T2.
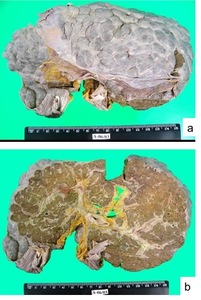
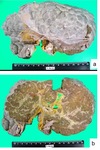
Fig. 9: Macroscopy - schistosomotic liver. Nodular appearance of the liver surface (a). resembling the aspect of coarse nodular cirrhosis. (b) Periportal fibrosis.
Periportal fibrosis
Periportal fibrosis,
also known as Symmers fibrosis,
was described in 1904 and constitutes a pathognomonic lesion of this chronic form.
The Niamey protocol comprises three stages,
which result into three scores assessing intraparenchymal and periportal findings as well as portal hypertension (IP,
PT,
PH).
In this protocol,
the assessment of periportal fibrosis is analyzed by two different,
previously described methods,
namely,
a qualitative one,
which takes into account the sonographic aspect of the liver evaluated based on characteristic patterns; the IP score in association with a quantitative method resulting from measurements of two or three second order portal branches,
the PT score.
In combination with those two criteria,
the presence of portal hypertension (PH score) is also analyzed with basis on indicators of its presence,
such as measurement of portal vein diameter,
presence of ascites and presence of collateral circulation.
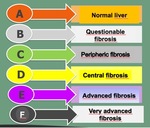
Such scores (IP,
PT and PH) are interpreted by means of a table(Fig.
10,
11 and 12) which classifies periportal fibrosis as follows: possible,
probable,
present,
present and advanced,
and present and advanced with portal hypertension.
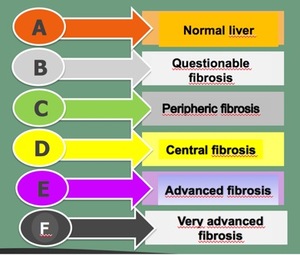
Fig. 10: Liver fibrosis classification.
References: internet
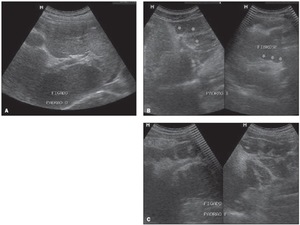
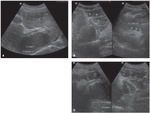
Fig. 11: Sonographic images of hepatic parenchyma demonstrating qualitative patterns with evaluation of the IP score according to the Niamey Working Group protocol, 2000. A: Pattern D: hyperechogenicity at the portal bifurcation and main trunk. B: Pattern E: highly echogenic signal extending from the portal vessels to the parenchyma. C: Pattern F: highly echogenic bands and strips extending from the main portal vein and its bifurcation to the periphery of the liver, retracting the surface of the organ.
References: -Fernandes DA. et al. Aspectos ultrassonográficos associados à morbidade de formas clínicas crônicas de esquistossomose mansônica, utilizando-se protocolo proposto pela Organização Mundial da Saúde. Radiologia Brasileira 2013; vol. 46 nº1-Jan./Fev.:1-6.
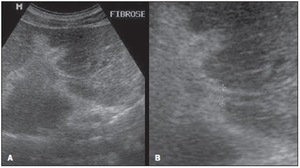
Fig. 12: Sonographic hepatic images demonstrating the quantitative data with evaluation of the IP score,
utilizing the measurement of second order portal branches, i.e., the first segmental branch originating
from the left or right branch of the main portal vein, according to the Niamey Working Group, 2000.
References: Fernandes DA. et al. Aspectos ultrassonográficos associados à morbidade de formas clínicas crônicas de esquistossomose mansônica, utilizando-se protocolo proposto pela Organização Mundial da Saúde. Radiologia Brasileira 2013; vol. 46 nº1-Jan./Fev.:1-6.
Portal Hypertension
Development of portal hypertension (PH),
regardless of its etiology,
is due to increased vascular resistance or increased portal venous flow or both.
In cirrhosis and schistosomiasis,
although increased vascular resistance appears to be the initial event,
mesenteric hyperflow is important in maintaining portal hypertension.
The pathophysiology of PH also frequently displays a systemic hyperdynamic state due to portal shunting of splanchnic vasoactive mediators.
Characteristically,
portosystemic collateral circulation and digestive hemorrhage through esophageal varices(Fig.
13,
14 and 15) occur independently from hepatocellular function.
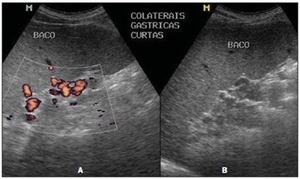
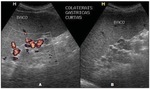
Fig. 13: Sonographic images demonstrating the PH score. Presence of collateral circulation, visualized
at color Doppler mapping (A) and at B mode (B).
References: Fernandes DA. et al. Aspectos ultrassonográficos associados à morbidade de formas clínicas crônicas de esquistossomose mansônica, utilizando-se protocolo proposto pela Organização Mundial da Saúde. Radiologia Brasileira 2013; vol. 46 nº1-Jan./Fev.:1-6.
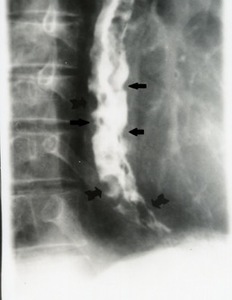
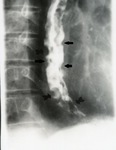
Fig. 14: Esophagogram reveals extensive varices in middle and lower esophagus (black arrows).
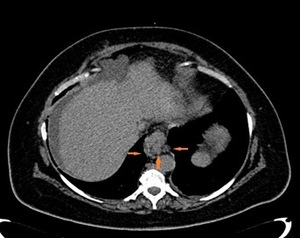
Fig. 15: CT scan shows varicose veins of the esophagus and gastric fundus (orange arrows).
Ultrasound
Ultrasound (US) represents an important contribution to the diagnosis of hepatosplenic mansonic schistosomiasis.
Abnormalities such as hyperechoic fibrotic bands along the portal vessels,
volumetric reduction of the right hepatic lobe,
enlargement of the left lobe,
perivesicular fibrotic thickening,
splenic nodules and splenomegaly can be recognized( Fig.
16).
Moreover,
the hemodynamic status of the portal venous system may also be evaluated by Doppler analysis,
including the identification of collateral veins and portosystemic venous shunts(Fig.
17).
Portal vein thrombosis may be present in a few cases.
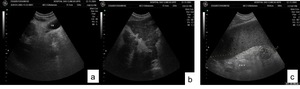
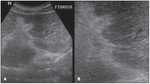
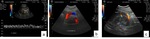
Fig. 16: Hepatosplenic schistosomiasis. US of the liver demonstrating perivesicular fibrotic thickening (a) and intense periportal fibrosis (b). US of the spleen (c) showing homogeneous splenomegaly.
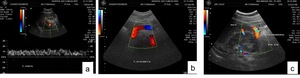
Fig. 17: Doppler US in a patient with portal hypertension due to schistosomiasis mansoni. (a) Dilated portal vein. (b) Proeminent splenic vein and hepatofugal flow and (c) Collateral circulation. Splenorenal.
Computed Tomography (CT) and Magnetic Resonance Imaging (MRI)
At CT(Fig.
18),
the periportal fibrosis manifests as low-attenuation rings around the portal vein branches throughout the liver,
with marked enhancement following intravenous administration of contrast material.
At MR(Fig.
19 and 20) imaging,
the periportal bands are isointense relative to normal liver parenchyma on T1-weighted images and hyperintense on T2-weighted images,
with marked enhancement following contrast material administration.
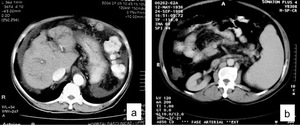
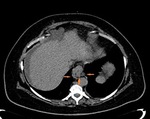
Fig. 18: CT of the abdomen showing ascites in a 69- year-old man with schistosomiasis mansoni (a). CT scan after contrast administration in a 62-year-old man with hepatosplenic schistosomiasis demonstrating splenomegaly (b).
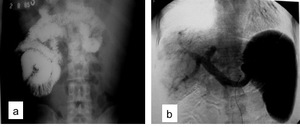
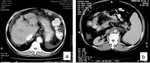
Fig. 19: Small bowel series (a) and angiography (b) demonstrating splenomegaly due to schistosomiasis mansoni. Note the dilated portal and splenic veins seen in angiography
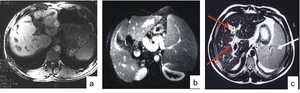
Fig. 20: MRI axial T1 in a 25-year-old man with hepatosplenic schistosomiasis and variceal bleeding. Note the hypointense periportal fibrosis. (b) MRI axial T1 after gadolinium showing periportal fibrosis, thickening of the gallbladder wall, splenomegaly and a dilated splenic vein. (c) MRI axial TSE T2 of the liver in a 34-year-old man with schistosomiasis reveals periportal thickening appearing as hyperintense signal (red arrows) and splenomegaly (white arrow).
MANIFESTATIONS RELATED TO PORTAL HYPERTENSION
PORTAL COLOPATHY
Portal colopathy is an important finding in hepatosplenic schistosomiasis and might be the cause of lower gastrointestinal bleeding in patients with severe portal hypertension.
The characteristic alterations of the portal colopathy in schistosomiasis are similar to those of portal colopathy in cirrhotics,
but they tend to be more frequent and accentuated.
Alterations in the colonic mucosa caused by Schistosoma mansoni eggs are described as increased vascular bed,
edema,
congestion,
scattered petechial spots with the aspect of "flea-bites",
ulceration,
and polyps.
ATYPICAL MANIFESTATIONS OF TOPIC FORMS
INTESTINAL POLYPS (Fig.
21)
Magalhães retrospectively reviewed 500 autopsies and 776 routine colon enemas.
He found polyps in 21 autopsies,
of which 38.8% were schistosomotic.
Lambertucci et al reported a case of a 35-year-old woman who presented with intestinal bleeding of recent onset.
She had hepatosplenic schistosomiasis,
portal hypertension and esophageal varices.
Colonoscopy showed 4 polyps (4 to 6mm in size).
Hyperplastic polyps were described and in all fragments there were mild to moderate inflammatory infiltrations around egg shells of Schistosoma mansoni.
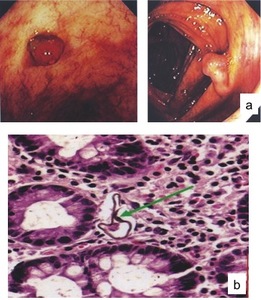
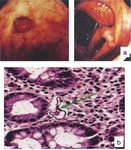
Fig. 21: Colonoscopy (a) demonstrating colonic polyps in the ascending colon and in the cecum in a 35-year-old woman with hepatosplenic schistosomiasis who presented with intestinal bleeding of recent onset. Also note the exuberant vasculature of the colon probably due to portal hypertension. (b) Microscopy: mild to moderate inflammatory infiltrations around egg shells of Schistosoma mansoni.
INTESTINAL OBSTRUCTION
Sigmoid colon obstruction complicated with perforation can be caused by schistosomiasis mansoni as the case reported by Atik et al. There are three other case reports of intestinal obstruction caused by schistosomiasis mansoni,
related to stenosing granulomatous processes and extensive pericolonic infiltration.
Elmasalme et al.
reported a case of sigmoid obstruction caused by a huge schistosomotic granuloma that was confused with a neoplasm; the diagnosis was established only in the post-operative period.
PORTAL VEIN ANEURYSM (Fig.
22)
Portal vein aneurysms have been considered a rare medical condition since they were first described about four decades ago.
They are most commonly found in the main portal vein branches or next to the junction of the mesenteric with the splenic vein as regards intra and extrahepatic aneurysms,
respectively.
By far,
the two most common causes are congenital malformations and chronic hepatic diseases.
Mucenic et al described the first case of portal vein aneurysm related to hepatosplenic schistosomiasis.
The described patient had a chronic liver disease with portal hypertension.
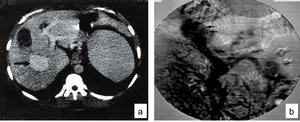
Fig. 22: CT of the abdomen (a) and angiography (b) showing a portal vein aneurysm (blaack arrows) in a patient with hepatosplenicschistosomiasis.
SCHISTOSOMOTIC GLOMERULOPATHY
The overall incidence of glomerular disease and schistosomiasis was shown to be about 5–6%,
whereas the hepatosplenic form of S.
mansoni infection may be accompanied by a glomerulopathy in 12-15% of cases.
This infection is clinically characterized by variable proteinuria ranging from asymptomatic to nephrotic syndrome.
The pathological changes vary.
Several patterns of glomerular pathology have been described associated with schistosomiasis mansoni and a histopathological classification was suggested by Barsoum.
Progression between these groups has been demonstrated.
Membranoproliferative glomerulonephritis was the most frequent pattern described in symptomatic patients (80%).
Immunofluorescence and electron microscopy (Fig.
23) reveal the presence of immune complexes containing IgM,
IgG,
IgA,
IgE,
complement components,
and schistosomal antigens in the mesangium and along the endothelial side of the capillary wall.
Sometimes renal histological changes are found to precede clinical manifestations.
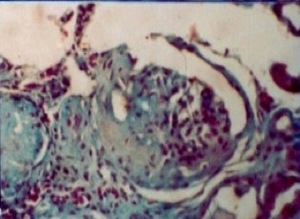
Fig. 23: Schistosomal glomerulopathy – microscopy demonstrating Focal Segmental Sclerosis (characterizing Class IV).
FEMALE GENITAL TRACT SCHISTOSOMIASIS
Egg-induced lesions in the upper and lower female reproductive tract are important complications of the infection with S.
mansoni.
Vaginal schistosomiasis was reported for the first time in Egypt in 1899.
It is estimated that at least 5% of girls and women with intestinal schistosomiasis also have genital lesions at an age when sexual activity peaks.
The understanding of the pathophysiology and pathology of genital lesions is only rudimentary.The frequency of genital lesions according to the topographic site reveal that the ovaries are the organs mainly affected,
followed by the cervix,
the tubes and the uterus.
Vulval lesions account for 6% of all case reports.
Recently it has been pointed out that schistosomiasis of the cervix,
together with or without concomitant human papilloma virus infection,
has to be considered a risk factor for the development of cancer of the cervix.
Furthermore since there is clinical,
epidemiological and immunological evidence of genital lesions facilitating the transmission and propagation of HIV,
genital schistosomiasis has to be regarded not only as an individual,
but also as a public health threat.
MALE GENITAL TRACT SCHISTOSOMIASIS (Fig.
24)
Epididymitis is a frequent inflammatory process.
It is related to sexually transmitted diseases,
urinary tract infections by E.
coli,
or scrotal trauma.
Rarely it can be related to schistosomiasis as the case reported by Alves et al.
The pathological examination showed the presence of chronic inflammatory process associated with eggs of the parasite Schistosoma mansoni in the epididymis.
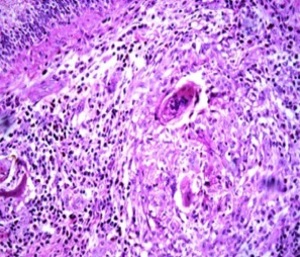
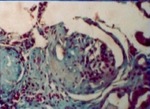
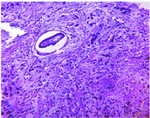
Fig. 24: Epididymitis – Photomicrography of epididymis slide showing granulomatous chronic inflammation with Langhans-type Giant cell reaction, presenting a large number of eosinophils around spiculated structures similar to S.mansoni eggs.
Schistosomiasis mansoni can also cause orchitis.
Schistosoma eggs have been described in this region (Fig 25).
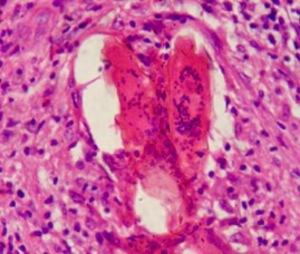
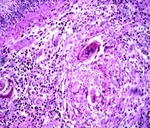
Fig. 25: Schistosomal orchitis. Histopathological study
Schistosomiasis has long been associated with malignant neoplasia.
A total of 5 cases of prostatic adenocarcinoma and concomitant gland schistosomiasis (S.
haematobium in 3 and S.
mansoni in 2) have been described in the literature.
One of them was reported by Oliveira et al in a Brazilian patient infected with S.
mansoni (Fig.
26).
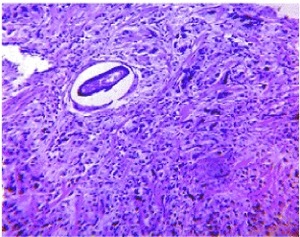
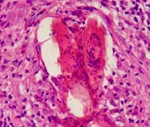
Fig. 26: Prostate cancer and schistosomiasis. Histopathological study of a tissue fragment obtained through a transrectal biopsy reveals a moderately differentiated prostatic adenocarcinoma seen as a malignant proliferation of acinar structures with hyperchromatic areas. An egg of S. mansoni with its lateral spines is also seen.
Rego et al reported a rare case of a huge abdominal cystic lesion caused by Schistosoma mansoni. CT demonstrated a cystic lesion in the abdominal cavity and after surgery schistosoma eggs were found in the wall of the cyst as well as the peritoneal cavity (Fig.
27)
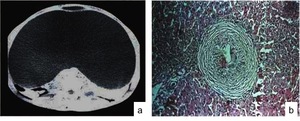
Fig. 27: CT of the abdomen (a) demonstrating a huge abdominal cystic lesion. Histopathological examination of the cyst wall (b) reveals granulomas with Schistosoma mansoni eggs.
ECTOPIC FORMS
Adult Schistosoma mansoni worms are usually lodged in venules of the mucosa and submucosa of the colon and rectum.
By virtue of this localization,
ova are frequently found in the colon and liver.
After the development of portal hypertension and the establishment of a portacaval shunt,
they can also be found in the lungs.
Despite high endemicity of this parasitosis in several parts of the world,
the finding of schistosome eggs or worms in other organs constitutes a rare phenomenon,
sometimes with serious clinical complications,
usually reported in the literature as pathological curiosities.
The possible implications that these ectopic localizations may have on the comprehension of the parasite behavior within host environment has been given little attention.