The mean age of a total of 48 patients was 53 years old (range,
21-83).
There was 33 women and 15 men.
42 patients presented with non-traumatic epigastric pain,
nausea,
vomiting,
of which and 20 with previous pancreatitis attack.
6 patients presented with trauma.
Mean lipasemia value was 1477,6 (range,
118-6736).
18 patients had infection signs (fever,
leucocytosis,
elevated CRP).
The Revised Atlanta Classification (RAC) distinguishes two phases of acute pancreatitis (AP):
- an early phase (1st week) with subsequent edema,
vascular damage,
hemorrhage,
and pancreatic necrosis
- a late phase (after the 1st week) that lasts for weeks to months,
with intrapancreatic and extrapancreatic inflammation.
In our study,
when patients arrived to the Emergency Department,
General surgeons evaluated the severity of AP during the early phase according to clinical parameters only: the presence or the absence of SIRS and organ/multiorgan failure using the BISAP score.
AP was categorized into:
- Mild: if no organ failure,
no local/systemic complications.
- Moderately severe: if transient organ failure,
local complications,
or exacerbation of co-morbid diseases.
- Severe: if persistent organ failure for more than 48 h.
SIRS is said to be present if 2 or more of these 4 criteria were present:
- temperature >38°C or <36°C
- pulse rate of >90 beats/min
- tachypnea (>20 breaths/min or PaCO2<32mmHg)
- deranged white blood cell count (>12,000/μL or <4,000/μL).
Organ failure was quantified using single clinical parameters or clinical severity scores combining clinical and laboratory parameters like Marshall Fig. 1 ,
Ranson’s criteria Fig. 2 ,
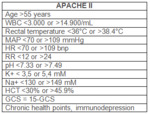
acute physiology and chronic health evaluation (APACHE)-II score Fig. 3 ,
or BISAP score.
The BISAP score was given at the time of admission as the number of criteria present of these five:
- blood urea nitrogen >25 mg/dL
- impaired mental status
- presence of SIRS
- age >60 years
- pleural effusion.
In the late phase,
patients were adressed to our department of Radiology to detect local complications and the severity of the disease using radiologic scoring systems.
Easy,
without contrast material but do not detect pancreatic necrosis.

Fig. 4: Radiological Scoring System based on the Balthazar classification system and CT Severity Index [Balthazar Radiology 1985 and 1990]: it categorizes the severity of pancreatitis in 5 grades, it does not require intravenous contrast material. Therefore, it does not caracterize the parenchymal necrosis.
References: N. Bharwani et al. / Clinical Radiology 66 (2011) 164-175
In our study,
we found:
Developed by Balthazar and his colleagues,
it needs contrast material,
is calculated within 48 h after admission and range from 0-10.

Fig. 5: A radiologic system developed by Balthazar: The CT Scoring Index with administration of intravenous contrast material detecting the pancreatic necrosis.
References: N. Bharwani et al. / Clinical Radiology 66 (2011) 164-175
AP is categorized as mild (0–3 points),
moderate (4–6 points),
or severe (7–10 points).
In our study,
we found:
- 16 patients with severe AP
- 24 patients with moderately severe AP
- 8 patients with mild AP
Developed by Mortele et al. Fig. 6,
it takes into account extrapancreatic complications and has a simpler quantification of amount of necrosis.
Points are given in increments of 2 comparing with the CTSI Fig. 14 Fig. 16 .
It has not been widely adopted.

Fig. 6: Modified CT severity index [Mortele AJR 2004]
References: N. Bharwani et al. / Clinical Radiology 66 (2011) 164-175
AP is categorized as mild (0–2 points),
moderate (4–6 points),
or severe (8–10 points).
Both CTSI and MCTSI showed significant correlation with clinical outcome parameters and severity grading as per the RAC.
It takes into account only the extrapancreatic changes and discounts pancreatic necrosis.
Range 0-7 and a score ≥ 4 means a severe AP.

Fig. 7: Radiologic EPIC score
References: N. Bharwani et al. / Clinical Radiology 66 (2011) 164-175
The prognostic significance of scoring systems like EPIC score is not clear.
CT findings
Clinical guidelines recommend CECT 48-72 hours after onset of symptoms in patients with suspected severe pancreatitis to identify necrosis and other complications that take few days to develop. Moreover,
it was suggested that early administration of intravenous contrast agents harms the already damaged pancreatic tissue.
The RAC distinguishes two subtypes:
- Interstitial edematous pancreatitis (IOP)
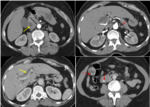
Characterised by localised or diffuse enlargement of the pancreas and homogeneous or slightly heterogeneous enhancement of the pancreatic parenchyma related to edema of following intravenous contrast administration Fig. 10 .
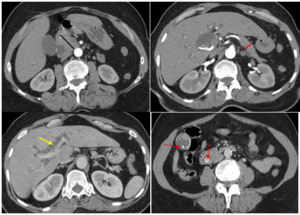
Fig. 10: A focal enlargement of the head of the pancreas (black measurement) in a 65 years-old women with a lipasemia value at 900. The body and the tail measurements (red arrow) are normal. The pancreatitis is classified Balthazar grade B. The is no pancreas necrosis: it is classified as mild pancreatitis using the CTSI (score:1). The gall bladder and the common bile duct contain stones (red arrows), leading to a intrahepatic (yellow arrow) and extrahepatic biliary dilatation.
References: Department of Radiology, Ibn Rochd Hospital, Casablanca/Morocco
Peak enhancement of normal pancreatic parenchyma is 80-150 HU at the parenchyma phase (40 seconds after intravenous contrast material administration).
Adjacent peri-pancreatic and retroperitoneal tissues can appear entirely normal or may demonstrate haziness and stranding Fig. 11 with peri-pancreatic fluid collections and over time,
with pseudocysts Fig. 12

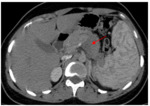
Fig. 11: Densification of the peripancreatic fat (yellow arrow). The pancreas volume is normal (black measurement). It is classified Balthazar grade C. There is no pancreatic necrosis: it is a mild pancreatitis (score: 2) using the CTSI.
References: Department of Radiology, Ibn Rochd Hospital, Casablanca/Morocco
Acute peripancreatic fluid collections develop within 4 weeks,
contain nonliquefied components Fig. 12.,
conform to anatomic boundaries of the retroperitoneum (especially the anterior pararenal fascia),
and are usually seen immediately next to the pancreas,
without discernable wall.

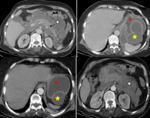
Fig. 12: Enlargement of the body and the tail of the pancreas (white measurement), with one ill-defined and fluid (homogenous) peripancreatic collections (yellow star). It was classified as Balthazar grade D. There is no pancreatic necrosis: it is classified using the CTSI as mild with a score of 3. A gallstone in the gallbladder (black arrow) was diagnosed as a cause of the pancreatitis.
References: Department of Radiology, Ibn Rochd Hospital, Casablanca/Morocco
APFC envolve 4 or more weeks after the initial disease into a pseudocyst that contains fluid amylase and lipase activity due to communication with the pancreatic ductal system.
It can be diagnosed as homogenous peripancreatic fluid collection with a well-defined enhancing wall. Fig. 25
Fig. 25
Fig. 25
Fig. 25

Fig. 26: Post ERCP pancreatitis: a rare iatrogenic cause of pancreatitis in a 78 years-old man with lipasemia value at 1007 and CRP at 134. CT scan shows a persistent fluid (homogenous) peripancreatic collection with a well-defined wall (white star). A catheter drainage (yellow arrows) of the fluid collection (pseudocyst) arriving to the left iliac fossa has been realized by surgeons.
The pancreatic parenchyma is normal without areas of nonenhancement (red arrows).
Aerobilia (black arrow) is due to the ERCP.
References: Department of Radiology, Ibn Rochd Hospital, Casablanca/Morocco
Sometimes,
APFC could develop a capsule earlier than 4 weeks after onset of IEP and it should be characterized as a pseudocyst.
Demonstrating communication with the pancreatic duct on CT may help determine management.
Both APFC and pseudocysts could be sterile or infected.
2. Necrotizing pancreatitis: has three morphologic subtypes:
When any region of pancreatic parenchyma demonstrates an attenuation of less than 30 HU during the pancreatic parenchymal phase.
It most often affects the peripheral parts of the gland while sparing the core.
In the 1st week of necrotizing pancreatitis,
pancreatic necrosis is homogeneous.
Later,
it appears heterogeneous as necrotic tissue gradually becomes liquefied.
Often the extent of parenchymal necrosis is divided into three categories:
o less than 30% of the gland involved Fig. 15

Fig. 15: Enlargement (1 point) and parenchymal necrosis (< 30%) of the body of the pancreas: 2 points. Area of nonenhancement (red arrow). Pancreatitis is classified as mild (score: 3) using the CTSI.
References: Department of Radiology, Ibn Rochd Hospital, Casablanca/Morocco
o 30%–50% Fig. 16

Fig. 16: Densification of the peripancreatic fat(red arrow). Heterogenous necrotic pancreatic collection (blue arrow: ANC)) replacing between 30 and 50% of the pancreatic parenchyma(score:4). There is no peripancreatic collection. Pancreatitis is categorized as Balthazar grade C and moderate (score: 6) using the CTSI. We can also note a left pleural effusion (yellow star). Using the MCTSI, it is a severe pancreatitis (score: 8).
References: Department of Radiology, Ibn Rochd Hospital, Casablanca/Morocco
o greater than 50% Fig. 17

Fig. 17: Axial CT image acquired 44 days from onset symptoms in a 48 years-old women: enlargement (1point) of the pancreas with a parenchymal necrotic collection >50% of the body of the pancreas (white arrow): these collection persisted, organized, enlarged and developed a well-defined wall and became a WON (walled-off necrosis). Left intrahepatic biliary dilatation (red arrow). A rare complication is associated: portal vein thrombosis (black arrows). Gallstones (yellow arrow) is the cause of the pancreatitis. It is classified using the CTSI as severe (score: 7).
References: Department of Radiology, Ibn Rochd Hospital, Casablanca/Morocco
When the extent of parenchymal involvement is less than 30%,
the low attenuation of necrosis may mimic the low attenuation of the gland seen with acute IEP,
making the diagnosis of necrosis less reliable.
In these cases,
follow-up CT may be required.
Involves only the peripancreatic fat,
has a better prognosis than parenchymal necrosis and a higher morbidity rate than IEP only.
Diagnosed when heterogeneous peripancreatic areas of nonenhancement are visualized commonly in the retroperitoneum and lesser sac Fig. 13.

Fig. 13: More than 2 ill-defined and heterogenous peripancreatic collections (white stars). Pancreatitis is classified Balthazar grade E. The diagnosis of ANC is easy because the CT is performed after 72h and the collections became heterogenous. The pancreatic volume is normal (black measurements) and there is no pancreatic necrosis: it is classified as moderate using the CTSI (score: 4).
References: Department of Radiology, Ibn Rochd Hospital, Casablanca/Morocco
Recognition of peripancreatic necrosis is difficult in the first week after onset because increased attenuation,
linear stranding,
and AFPC associated with IEP have a similar appearance.
After 1 week,
the heterogeneous peripancreatic fat and the liquefied components among the fat become more apparent,
so that peripancreatic necrosis can be diagnosed with greater confidence.
The most common morphologic subtype is the combination of the findings described for pancreatic parenchymal necrosis alone and peripancreatic necrosis alone.
Fig. 20 Fig. 18
Peripancreatic necrosis associated with full width necrosis of the pancreatic parenchyma may be connected to the main pancreatic duct

Fig. 18: Combined necrosis in a 48-year-old woman. (a) Axial contrast-enhanced CT image shows
an evolving, mildly heterogeneous pancreatic-peripancreatic necrotic collection involving the neck, body,
and tail of the pancreas (arrows). (b) Axial T2-weighted MR image shows a well-defined, walled-off,
heterogeneous pancreatic-peripancreatic collection, with a fluid component that appears hyperintense
(white arrows) and necrotic nonliquefied material that appears hypointense (black arrow). Hypointense
sludge is seen in the gallbladder (*).
References: Shyu et al. RadioGraphics 2014; 34:1218–1239
- Pancreatic and peripancreatic necrotic collections:
Terms such as pancreatic abscess,
fluid collection,
and phlegmon are no longer accepted owing to their ambiguity.
Collections associated with necrotizing pancreatitis are heterogenous and contain nonliquefied components:
o ANC
A collection developed within the first 4 weeks of onset of acute necrotizing pancreatitis,
without a well-defined wall.
It could be:
-a pancreatic collection that should never be called a pseudocyst (pseudocysts are only peripancreatic) Fig. 16
-an heterogenous peripancreatic collection Fig. 13
-an homogenous peripancreatic collection but since its association with pancreatic necrosis,
it could be distinguished on CT from APFC (that is only peripancreatic without pancreatic necrosis) Fig. 20.
Diagnosis of heterogenous appearance is confirmed on MRI.
Fig. 18
-an homogenous peripancreatic collection but with heterogenous appearance on MRI or on late CT images (within 2-6weeks).
In fact,
when the collection starts liquefying and becomes clearly heterogeneous,
the necrosis can be detected better.

Fig. 20: CT scan after 72h from onset symptoms revealed nonenhanced areas of the tail <30% (yellow arrow) and multiple ill-defined and heterogenous peripancreatic collections limited by the retroperitoneal. These collections are called ANC because pancreas necrosis is present. It is classified Balthazar grade E, and as moderate (score: 6) using the CTSI.
References: Department of Radiology, Ibn Rochd Hospital, Casablanca/Morocco
An ANC may or may not have a connection to the disrupted pancreatic ductal system within the necrosis.
o WON
After 4 weeks,
ANC generally evolves into WON,
which is heterogeneous or homogeneous with a well-defined wall Fig. 17.
It may be pancreatic,
peripancreatic or combined.
Any collection with a well-defined wall that replaces portions of the pancreatic parenchyma should be called a WON after 4 weeks from onset of necrotizing pancreatitis.
Both ANC and WON may be infected or not.

Fig. 8
References: Shyu et al. RadioGraphics 2014; 34:1218–1239
Diagnostic problems in AP
Both develop within 4 weeks and have no discernable walls.
When areas of non-enhancement are only peripancreatic,
distinction between APFC and ANC in the 1st week of AP may be difficult or impossible,
because both collections may appear as areas of homogeneous nonenhancement Fig. 19 .

Fig. 19: White stars: multiple ill-defined peripancreatic collections. Since there is no pancreatic necrosis, these collections are either fluid or necrotic (APFC or ANC) depending on the MRI appearance): the pancreatitis is classified Balthazar grade E. The pancreas volume is normal and the parenchyma is correctly enhanced, without necrosis (black arrow): it is a moderate pancreatitis as the CTSI scoring system (score: 4).
The retrogastric collection (yellow star) has a well-defined wall: pseudocyst or WON depending on the heterogenous (necrotic) or homogenous composition that could be defined only on MRI. The red star indicates the stomach.
References: Department of Radiology, Ibn Rochd Hospital, Casablanca/Morocco
Beyond 1 week after onset,
collections associated with peripancreatic necrosis become more heterogeneous and are more readily distinguished from APFC.
MR imaging is more helpful than CT in this regard: it can detect nonliquefied components that allow classification of a collection as an ANC.
When slightly heterogeneous appearance of the nonenhancing peripancreatic areas or if hemorrhage or fatty tissue are seen,
the diagnosis of ANC is suggested.
Such findings are not compatible with IEP.
The distinction is easy when pancreatic parenchymal necrosis is present: associated collections are then classified as an ANC.
Both develop 4 weeks after onset,
from nonnecrotic (APFC) and necrotic collections (ANC),
respectively and have well-defined enhancing walls.
Pseudocysts contain homogeneous fluid and are only peripancreatic.
They are more likely to be associated with main pancreatic ductal dilatation (>3 mm),
possibly as a result of the compression of pancreatic parenchyma.
WON contain necrotic material (fat and fluid),
and can involve both pancreatic and peripancreatic tissue. Its diagnosis is also favored when pancreatic collection grows,
extends to the paracolic space,
and has an irregular border.
However,
any pancreatic collection with well-defined wall is classified as WON,
regardless of its appearance.
Ductal dilatation is less likely to occur with WON because pancreatic fluid simply leaks into the collection.
MRI is more helpful than CT to detect liquefied from nonliquefied components that allow classification of a collection as an pseudocyst or WON.
Both pseudocysts and WON demonstrate a well-defined,
T2-hypointense enhancing wall but liquefied components of pseudocysts appear hyperintense and nonliquefied components of WON appear hypointense.
Complications of AP
The most common complications of AP are:
- infected acute collections: APFC or ANC
- extrahepatic biliary dilatation
- vascular complications (splenic/portal and mesenteric veinous thrombosis,
varices or arterial pseudoaneurysm)
- pleural effusion,
ascites
- other intraabdominal findings caused by pancreatic secretions,
in stomach,
duodenum,
small bowel,
colon,
spleen,
kidney,
ureters or liver.
1.
Infection
CT attenuation measurements cannot be used for distinction between sterile and infected collection.
Distinction is important because treatment and prognosis are different.
In the absence of intervention (percutaneous catheter drainage or surgical necrosectomy),
the presence of gas within pancreas or peripancreatic tissue at imaging suggests infection,
although gas is found in a minority of cases of confirmed infection.
Spontaneous drainage into the gastrointestinal tract can lead to an erroneous diagnosis of infected collection.
Careful analysis of the adjacent gastrointestinal walls can help prevent this diagnostic pitfall.

Fig. 21: (a) (b) Non-enhanced axial CT of the abdomen showing mottled appearance due to air pockets in the fundus of the stomach (right curved arrow) and pancreatic arenchyma (left curved arrow). The mottled appearance within the stomach and the pancreas are suggestive of walled of necrosis. Thickening of the left anterior renal fascia is seen (arrow head);
(c) Non-enhanced axial CT of the abdomen showing mottled appearance in the stomach(arrow head) and the pancreatic parenchyma (asterix) suggestive of fistulous communication between stomach and the pancreas(down arrow). Significant perigastric and peri pancreatic fat stranding is seen (left arrow).
References: R. Ramachandran et al., Surgical Science, 2014, 5, 237-241,
The absence of gas does not signify the absence of infection.
Gas can also be found in uninfected collections as a result of gastrointestinal inflammation and mass effect on adjacent organs.
2. Biliary obstruction
It can result from choledocholithiasis,
mass effect from pancreatic inflammation or a collection,
or biliary stricture from exposure to pancreatic proteolytic enzymes.
A strictured bile duct may appear tapered,
compressed,
or simply occluded with upstream biliary dilatation,
and it may or may not demonstrate mural enhancement.
MRI is particularly helpful for delineating the biliary system,
identifying the narrowed segment,
and identifying the cause of the obstruction.
3. Pancreatic Duct Stricture
Strictures may be single or multiple,
may result in upstream dilatation of the pancreatic duct
4. Disconnected Pancreatic Duct
Suggested when a large or growing collection involves the neck or body of the pancreas and a viable segment of upstream body or tail.
The duct in the upstream pancreas may or may not be dilated and may be seen communicating directly with the collection; disruption is suggested when the duct is oriented perpendicular to the collection.
5. Pseudoaneurysm
It develops when an arterial vessel wall is weakened by pancreatic proteolytic enzymes.
It appears as a focal outpouching of an arterial vessel within the necrotic region Fig. 23 .
A mural thrombus may also be seen.

Fig. 23: Pseudoaneurysm in a 32-year-old man
with necrotizing pancreatitis. Coronal reformatted
CT image demonstrates an outpouching from the
left gastric artery (arrow), a finding that is consistent
with pseudoaneurysm. The pseudoaneurysm
was treated surgically owing to the inability to
access the left gastric artery with a percutaneous
endovascular approach.
References: Shyu et al. RadioGraphics 2014; 34:1218–1239
It involves most frequently the splenic artery,
followed by the gastroduodenal,
pancreaticoduodenal,
hepatic,
and left gastric arteries.
6. Hemorrhage
The splenic artery,
portal vein,
splenic vein,
and other smaller peripancreatic vessels are the most common sources of bleeding.
Hemorrhage manifests at CT as a region of high attenuation,
typically in an area of necrosis.
MRI is more sensitive than CT for the detection of hemorrhage; subacute hemorrhage appears T1 and T2 hyperintense Fig. 22 .

Fig. 22: Combined necrosis with hemorrhage in a 48-year-old woman. (a) Axial contrast-enhanced
CT image obtained for worsening abdominal pain shows a well-defined, heterogeneous pancreaticperipancreatic
collection (arrows). (b) Axial fat-saturated T1-weighted MR image obtained 2 days later
for the evaluation of abdominal pain reveals hyperintense signal in the body and tail of the pancreas and
in the peripancreatic region (arrows), findings that are suggestive of hemorrhage.
References: Shyu et al. RadioGraphics 2014; 34:1218–1239
7. Venous Thrombosis
It results from local prothrombotic inflammatory factors,
reduced venous flow,
and mass effect on a venous structure.
It appears as focal or complete nonenhancement of an expanded venous structure Fig. 17 .
It involves most commonly the splenic vein; followed by the superior mesenteric and portal veins.
Etiologies
The two most common and important causes of AP are gallstones (40–70%) and alcohol (25–35%).
Obstructing stone in the pancreatic duct near the sphincter of Oddi leads to AP.
Fig. 10

Fig. 10: A focal enlargement of the head of the pancreas (black measurement) in a 65 years-old women with a lipasemia value at 900. The body and the tail measurements (red arrow) are normal. The pancreatitis is classified Balthazar grade B. The is no pancreas necrosis: it is classified as mild pancreatitis using the CTSI (score:1). The gall bladder and the common bile duct contain stones (red arrows), leading to a intrahepatic (yellow arrow) and extrahepatic biliary dilatation.
References: Department of Radiology, Ibn Rochd Hospital, Casablanca/Morocco
It is believed that the acinar cells of the pancreas are susceptible to damage by ethanol..
It may show hemorrhages,
disconnected duct or pancreatic fistulae Fig. 24

Fig. 24: Initial scan after trauma in a 26-year-old man with an initial lipase value at 1184 demonstrates linear low density at the junction of the body and tail of the pancreas, representing a pancreatic laceration (black arrows) which communicates with 2 peripancreatic collections (red stars). There is also a kidney contusion (white arrow) and pleural effusion (yellow star).
References: Department of Radiology, Ibn Rochd Hospital, Casablanca/Morocco
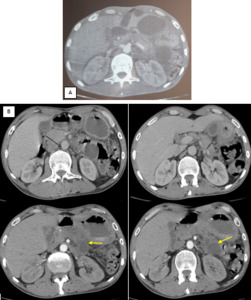
Fig. 25: A: Initial scan after trauma in a 19-year-old man with an initial lipase value at 800 demonstrates low density peripancreatic collections arround the tail of the pancreas (white arrows). The pancreas is normal without necrosis.
B. After 4 months, the two collections persisted, developed a well-defined wall and became a pseudocyst (yellow arrows).
References: Department of Radiology, Ibn Rochd Hospital, Casablanca/Morocco
Another common cause,
iatrogenic pancreatitis,
may occur after endoscopic retrograde cholangiopancreatography (ERCP) in up to 5% of patients Fig. 26
Other etiologies of AP include medications,
infections,
hereditary,
and autoimmune disease.