ADPKD is a monogenic disease.
The main systemic manifestation occurs in the kidney.
It can also affect the liver,
pancreas or blood vessels.
This disease is the most common hereditary kidney disease; affecting about 300 thousand to 600 thousand Americans and between 1:400-1000 live births in worldwide.
There is no predominance of sex.
The son of an affected father has a 50% chance of inheriting the gene mutation with full penetrance.
The mutation occurs spontaneously from 5 to 10%.
The mutation of PKD1 is more common than PKD2 (85% of cases) and it is associated with greater formation of renal cysts that eventually culminates in renal failure.
It represents 5% of the population in end-stage renal disease (ESRD).
About 50% of patients with ADPKD will develop ESRD at around 60 years of age and will require a renal replacement therapy either dialysis or transplantation.
Systemic arterial hypertension in patients with ADPKD is present in up to 50% between 20 and 30 years old,
with functional normal kidney,
and debut 10 years earlier than the general population.
PKD-1
An 85% of cases occur due to mutations in the gene PKD1 (MIM 601313) located on the short arm of chromosome 16 (16p13) that encodes the protein polycystin 1 ,
is characterized by an earlier clinical presentation,
with higher morbidity and mortality.
PKD-2
The remaining 15% occurs due to mutations in the PKD2 gene (MIM 173910) located on the long arm of chromosome 4 (4q21) that encodes the protein polycystin 2,
the clinical presentation will have a less early and milder course.
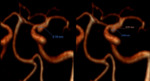
People who have ADPKD at the age of 63 will have a PKD2 gene up to 40% and increase to 50-70% if the age of presentation is after 70 years (Fig. 1 ).
Polycystin 1 is a transmembrane protein and primary cilia where it interacts with polycystin 2. Polycystin is a member of the transient receptor potential channel family,
found in the endoplasmic reticulum and in primary cilia,
(Table 1).
These plays an important role in tissue development and homeostasis by regulating intracellular calcium transport.
In addition,
they are related in the cell-cell and matrix-cell interactions that determine tubular morphogenesis.
The alteration of the function of these proteins leads to epithelial-tubular proliferation,
increased intratubular fluid secretion and alters the interaction between basal membrane and extracellular matrix.
All these changes eventually progress to tubular ectasia and cyst formation.
Also,
polycystins are expresed (in less proportion) in many tissues including:
- Tubular epithelia.
- Endothelial and vascular smooth muscle cells.
- Cardiomyocytes.
This explains the multiple extrarenal manifestations.
Mutations of PKD1 and PKD2 produces identical renal and extrarenal manifestations,
although as already mentioned the presentation and progression to ESRD is at earlier ages in the PKD1 mutation.
RENAL
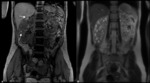
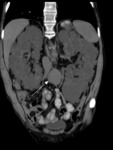
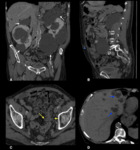
The signs and symptoms are due the growth of the kidneys and cysts,
as well as the complications of the cysts (Fig. 2):
• Systemic arterial hypertension in up to 50% of cases
• Increase in abdominal volume
• Chronic pain in flanks
• Macroscopic hematuria
• Urinary tract infections
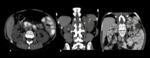
• Urolithiasis (Fig. 3)
ADPKD does not increase the risk of renal cell carcinoma.
EXTRARENAL
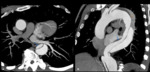
The extrarenal manifestations are also characterized by the formation of cystic cavities,
which include:
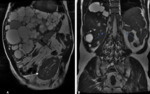
- Liver (94%) (Fig. 4)
- Seminal vesicles (40%)
- Pancreas (9%)
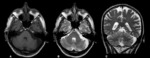
- Arachnoid space (8%) (Fig. 5)
Polycystic liver disease is the most common extrarenal manifestation and its frequency increases with age.
The volume of the cyst is higher in women,
mainly in those with multiple pregnancies and contraceptive use.
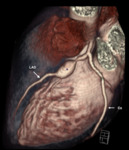
There is a wide variety of cardiovascular abnormalities and include:
- Dolichoectasias.
- Dissection of the aorta (Fig. 9) and other arteries such as coronary arteries.
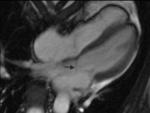
- Miltral valve propapse (25%) (Fig. 10)
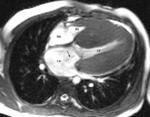
- Left ventricular hypertrophy (Fig. 11)
- Pericardial effusion.
These are the entities that represent the greatest risk for life.
The risk of developing IA is 9 to 12% (up to 30%) if there is a family history of IA,
compared with the general population that is 1-4%.
The rupture of IA´s represents 4 to 7% of deaths recorded in patients with ADPKD,
with a high morbidity and mortality (40% mortality in the first 30 days after rupture of the aneurysm).
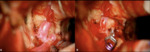
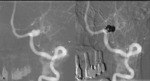
The treatment can be surgical or endovascular ( Fig. 12, Fig. 13).
Other connective tissue abnormalities (Fig. 14) :
- Diverticular disease
- Abdominal hernia
- Bronchiectasis
ADPKD DIAGNOSIS.
There is a tendency by consensus that molecular and genetic study is the ideal method for the diagnosis of ADPKD however,
the renal US should be the first diagnostic study in patients with clinical suspicion (Evidence 1B),
(Table 2), compare the diverse imaging method for diagnosis ADPKD.
Caution must be exerted if IV iodine contrast is to be used.
As this patients generally have or will developed renal insuffiency.
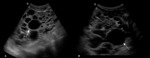
ULTRASOUND
It is characterized by its low cost,
non-invasive,
easy availability,
and not using ionizing radiation.
It has shown that its advanced stage sensitivity of the disease is 100%,
while in early stages,
due to the low velocity of growth of the cyst,
the sensitivity can decrease to 88.5%.
Recently the US high definition can detect 2 to 3 mm lesions.
Facilitates identification of urinary tract obstruction,
lithiasis,
and complicated cysts.
It is not very sensitive to characterize the volume in kidneys greater than 16 cm and does not have enough precision to obtain exact measurements,
due to the intraobserver and interobserver variability and the limited field of vision.
Another disadvantage is that it is a operator dependent.
COMPUTED TOMOGRAPHY.
This image modality offers a high spatial resolution,
facilitating the detection of smaller cysts.
It is highy sensitive to distinguish lithiasis,
images suggestive of intracystic hemorrhage and sometimes suggestive of infection (gas,
intracystic level,
increased density of nearby fat) can also be observed.
Some disadvantages are exposure to radiation and the use of intravenous contrast agents are eliminated by the kidney and in patients with ADPKD it can be an absolute contraindication for the risk of nephrotoxicity.
MAGNETIC RESONANCE
This imaging method is not regularly used for diagnosis.
Its advantages are that is does not subject the patient to ionizing radiation,
it has great spatial resolution and can detect very small lesions and cyst complications, with a high sensitivity and specificity of 86-95% and 100% respectively.
The most commonly used contrast medium is gadolinium,
which has low nephrotoxicity,
is generally safe in patients with an almost normal renal filtration rate.
The prevalence of allergy or anaphylactic reaction is lower than that of other contrast media such as iodine.
Among the disadvantages of MRI is the duration of the study,
this is greater than the US and CT,
little accessibility in some centers and a higher cost.
In addition,
there are some contraindications to its use,
for example:
- Patients with claustrophobia.
- Those who have devices such as pacemakers,
implantable cardioverter defibrillators,
some brain clips that are contraindicated to enter the resonator according to the RM safety guidelines.
RENAL SCINTIGRAPHY
This study facilitates the functional evaluation of the kidney and it is not used to establish the diagnosis.
The most widely used radiopharmaceuticals are Tc-99 DTPA and Tc-99 MAG3,
they do not have side effects,
allergies and there is not risk of nephrotoxicity.
The Tc-99 MAG3 offers better image quality and renal clearance.
The study is carried out in three phases: blood flow phase,
concentration phase and phase of clearance or elimination.
Scintigraphy may be useful in the resection of native kidneys (the more functional kidney could be preserved) (Fig. 15).
DIAGNOSTIC CRITERIA
• Bilateral kidney cysts (Fig. 16)
• Cysts in other organs
• Extrarenal manifestations (previously mentioned)
• Absence of manifestations that suggest another cystic disease.
In an individual with a positive ADPKD history:
• Increase in kidney or liver size in the physical examination is highly suggestive for diagnosis.
• Presence of hypertension,
prolapse of the mitral valve or abdominal hernia are suggestive findings for the diagnosis.
The absence of a family history of ADPKD:
• Increase in bilateral renal size and cysts with or without the presence of hepatic cysts
• Absence of manifestations that suggest another cystic disease
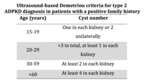
The US diagnosis of ADPKD type PKD1 is based on the Ravine criteria (Table 3),
the sensitivity is close to 100% in both patients older than 30 years and in younger patients.
The application of the Ravine criteria in patients with PKD2 mutation is lower,
particularly in the group younger than 30 years (in this case,
the diagnostic sensitivity is around 67%).
This is also true in patients with no family history of the disease,
therefore,
more precise diagnostic criteria developed by Demetriou et al.
(Table 4) and are recommended in suspected cases of ADPKD type PKD2.
MOLECULAR CRITERIA
Up to 10% of patients may have sporadic mutations and although the diagnosis must be clinical and radiological,
in some cases it will be necessary to carry out a genetic study especially in the early stages (Table 5).
According to the Spanish clinical guidelines there are specific situations where the genetic diagnosis is indicated:
• Potential living donor with family relationship.
• Patients with suspected disease with no family history of ADPKD
• Patients with very early onset of disease.
• Patients with or without a family history who want a future pre-implantation or prenatal diagnosis.
• Families with multiple relatives with renal cysts with an atypical pattern of ADPKD,
candidates for differential diagnosis of other renal cystic diseases.
TREATMENT AND PROGNOSIS
ADPKD is characterized by the formation and progressive growth of renal cystic cavities that eventually causes associated ESRD ,
abdominal pain,
hemorrhage within the cyst,
hematuria,
nephrolithiasis and infection of the cyst.
There is progressive destruction of the renal parenchyma,
with an initial compensatory phase of hyperfiltration in the remaining glomeruli,
maintaining a normal renal function for decades.
Around the 4th decade of life,
renal function decreases with a direct relationship of nephron loss.
According to the Kidney Disease Improving Global Outcomes (KDIGO)2012,
CKD is defined as abnormalities of renal structure or function,
present for> 3 months,
with implications for health.
Peritoneal dialysis and hemodialysis are valid renal replacement therapy modalities for patients with ESRD.
DIALYSIS
This type of substitution treatment offers better prognosis and quality of life to patients with ADPKD than those who are not carriers of the disease.
Some patients with hepatomegaly restricts the area available for peritoneal exchang and increases the possibilities of hydrothorax and abdominal hernias; in these cases hemodialysis should be considered as the best option.
NEPHRECTOMY
About 1/5 of patients who will undergo to kidney transplantation will require a unilateral or bilateral nephrectomy (up to 35.4%).
The time to contemplate this procedure consists in the presence of persistent pain,
space for a future transplant,
hematuria,
recurrent infection or gastrointestinal symptoms due to the intra-abdominal mass effect (Fig. 17) .
KIDNEY TRANSPLANT
It is considered the treatment of choice for patients diagnosed with ADPKD.
The main difference between the population that ESRD by other etiologies lies in the need to assess the existence of IA and decide whether to perform nephrectomy of native kidneys to make room for a kidney transplant,
there is not enough evidence about whether the native kidney nephrectomy must be performed pre-transplant or simultaneous in the act of transplant.