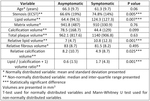
In total 54 patients (67.7% male,
32.3% female,
median age of patients: 61 years) and 65 internal carotid artery plaques were included in this study.
Diametric stenosis of the internal carotid artery (based on ECST criteria) was significantly higher in symptomatic plaques (median 74.8% vs 66.6%,
p<0.01).
Regarding plaque composition analysis,
symptomatic plaques were found to have significantly higher lipid volume (median mm3 124.3 vs 64.4,
p<0.01),
relative lipid volume (median 10.87 vs 7,
p=0.001),
lower relative calcification volume (median 4.9 vs 8.2,
p<0.05) and higher L/C ratio (1.7 vs 0.6,
p=0.001) compared with asymptomatic.
On the contrary,
no statistically significant difference was detected between symptomatic and asymptomatic plaques for fibrous matrix and calcification volume,
the sum of three components volume (lipid,
fibrous matrix and calcification) and relative fibrous volume.
The exact values of every variable and the statistical tests results can be found in Table 1.
The boxplots of these comparisons are presented in Fig. 1.
Based on these results,
it appears that some plaque components are significantly associated with the occurrence of stroke,
while others not.
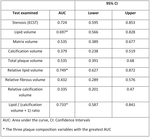
On ROC analysis,
the areas under the curve (AUC) of all these parameters were assessed and were as follows: stenosis (ECST) 0.724,
lipid volume 0.697,
calcification volume 0.379,
fibrous matrix volume 0.533,
total plaque volume 0.535,
relative lipid volume 0.749,
relative calcification volume 0.335,
relative fibrous volume 0.432,
ratio of lipid volume divided by calcification volume plus one 0.733.
The three variables with the greatest AUC were: i) the relative lipid volume (0.749),
followed by ii) L/C ratio (0.733) and iii) lipid volume (0.697).
The 95% confidence intervals of ROC analysis can be found in Table 2.
The AUC of the variables can be found in Fig. 2.
Screening,
optimal and diagnostic cut-off values were defined for the relative lipid volume,
L/C ratio and lipid volume to achieve superior sensitivity,
balanced sensitivity and specificity and superior specificity respectively.
For lipid volume,
the screening cut-off value was 19.75 mm3 and the resulting sensitivity and specificity was 96.7% and 21.2% respectively.
The optimal cut off value was 76.25 mm3 and the respective values were 70% and 63.6%.
The diagnostic cut-off value was 264.35 mm3 and the respective measures were 16.7% and 97%.
For relative lipid volume the screening,
optimal and diagnostic cut-off values were 2.67 mm3,
8.02 mm3 and 15.65 mm3 respectively.
The respective resulting sensitivities and specificities are 96.7% and 21.2%,
70% and 72.7%,
33.3% and 97%.
For L/C ratio the screening,
optimal and diagnostic cut-off values were 0.36,
1.18 and 12.5 respectively.
The respective resulting sensitivities and specificities are 96.7% and 27.3%,
70% and 69.7%,
20% and 97%.
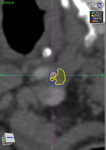
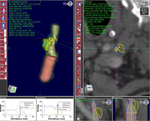
Examples of cases analyzed with the software can be found in Fig. 3, Fig. 4,
Fig. 5, Fig. 6.
The limitations of the study include the relatively small number of patients and carotid plaques examined as well as a potential spectrum bias as only plaques with moderate or severe stenosis have been recruited.
Further studies are needed to confirm the value of this technology and investigate the implications of these results in terms of patient management.