Patients and CTp study
This study included 46 patients randomly chosen from one Centre of the Project PIXEL,
one of the widest multicentre European study employing CTp for investigating the potential role of perfusion parameters in order to predict the development of liver metastases within three years from colorectal cancer diagnosis.
All patients underwent an axial CTp liver examination,
consisting of 60 CTp scans,
each of 8 slices of 5-mm thickness,
acquired within 2min (every 1s for the first 30s,
and every 3s after),
in which patients were asked to breathe shallowly.
Acquisition started contemporaneously with the injection of a bolus of Contrast-Agent (CA),
at a speed of 5ml/s and a concentration of 350mgI/ml,
followed by 20ml of physiological solution.
CTp protocol required fixed tube current (100mA) and voltage (80kV),
with 1s rotation time (100mAs exposure).
TCCs extraction and pre-processing steps
For each volume,
a central slice was selected and the Regions of Interest (ROIs) were contoured.
In particular,
two ROIs were drawn on the aorta and tissue,
and one more ROI outlined the portal vein in order to represent the dual-input hepatic blood supply.
Moreover,
the portal ROI was aligned over time in order to compensate for patient motion.
Hence,
two TCCs were attained for the aortic and portal inputs by averaging the CA enhancement within the ROIs,
while voxel-based TCCs were extracted from the organ.
Right after,
the TCCs were denoised through an edge-preserving spatial filter and a non-parametric model was employed to achieve a uniform sampling frequency of 1Hz.
Then,
the baseline was removed from the TCCs and the corresponding Time Attenuation Curves (TACs) were computed for the aortic (CA(t)),
portal (CP(t)),
and tissue (CT(t)) ROIs.
Finally,
a fitting procedure was performed on the achieved TACs by adopting the Lagged Normal model [8] for CA(t) and CP(t),
and an in-house algorithm directly exploiting the native TACs in order to achieve a patient-driven fitting of tissue signals at voxel-level,
by considering only samples belonging to CA first-pass.
The algorithm implemented provided us with two tissue components,
referred to the aortic (CTa(t)) and portal (CTp(t)) contribution,
respectively.
Computation of perfusion parameters
The MS method was adopted in order to compute tissue perfusion during the CA first-pass.
Hence,
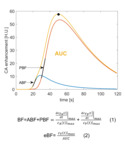
BF was estimated according to Eq.1 of Fig.1,
expressing the MS formulation applied to the dual-input model of the liver [9]
- Equilibrium Blood Flow (eBF)
eBF refers to the input-output balance occurring in the CT(t) maximum value,
where the first derivative of the signal is equal to 0.
Hence,
the peak value of CT(t),
measured in Hounsfield Unit (HU) and proportional to CA concentration,
was normalized by the Area Under the Curve (AUC),
corresponding to CA concentration-time product.
The eBF attained as a frequency measure was converted in [ml/min/100g],
by assuming a unit tissue density.
Eq.
2 of Fig.
1 reports the eBF formulation.
Assessment of results
Mean (m) and standard deviation (σ) of eBF and BF values are computed for each patient and,
globally,
for the whole cohort,
accordingly.
The Coefficient of Variation (CV) is considered in order to assess the repeatability of the two global mean values.
Moreover,
colorimetric maps of BF and eBF are submitted to readers’ evaluation and for each patient,
voxel-wise correlations are assessed trough the computation of the squared Pearson’s coefficient (R2).