We’ve performed a structured literature review across Medline and Pubmed databases,
with concept map of terms:pre-operative,
meningioma,
embolization,
complications,
embolizing agent,
.with literature articles from 1993 to 2018 were retrospectively reviewed.
Inclusion criteria included prospective and retrospective in the aforementioned period in English,
Exclusion criteria included case reports with small number of cases “ less than 20 cases”,
or non English language articles.
Studies included are tabulated in [table1] including 25 studies,
with different techniques and variable embolizing materials.
Articles were screened for advantages of preoperative embolization of meningioma,
complications happened in some cases,
the outcomes correlation with the embolizing agents used and nature of the meningioma including site and any special tumor characteristics,
and time between embolizations and surgery,
as well as patient type and age.
Cases from our practice
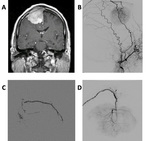
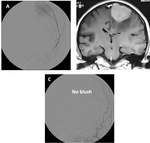
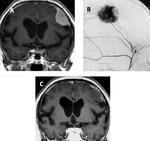
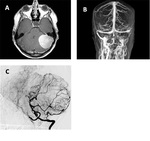
We present 4 cases of our practice,
the cases were performed in Egyptian military hospitals,
in first half of 2016.
[Table 2],
[Table 3] show the different embolizing material used and advantages,and disadvantages of each in[figures 1,
2,
3,
4].
The technique used was transarterial intravascular catheterization.
Embolizing material used were PVA particles 300-500 um and 250-300 um and OnyxⓇ.
Interdisciplinary risk-benefit weighing assessment was done for each and every case as general risk assessment couldn’t be applied.
Risks depend on Tumor localization,
size of the tumor,
vascular supply whether there is dangerous anastomoses,
and possibilities of peripheral vs deep supply,
in addition to pre-existing mass effect (post-embolization edema/swelling possibility).
Risk assessment is primarily indicated to balance surgery alone versus embolization and surgery.
The time from embolization to surgery was 3-10 days.
Discussion,
to embolize or not to embolize?
The real additive value of preoperative embolization for treatment of intracranial meningiomas remains contentious.
With growing number of literature suggests that this can be a valuable and safe adjunct to surgical excision in selected cases,
particularly those that are surgically challenging.[4],
[5],
[6]
The decision to embolize should be made following a risk-benefit discussion,
within a multidisciplinary team meetings and it’s worth mentioning that indications for treatment were not standardized.[7]
The complications experienced by [7] (Borg A.
et al.) were mostly falling into 2 principal categories: excessive ischemic tumoral necrosis leading to postprocedural tumoral hemorrhage and nontarget embolization causing neuroparenchymal or extracranial ischemia (ie,skin),
this was also demonstrated by (Gruber et al.) [5],[8],[9],[10]
Mass effect and other angiography related complications has been shown in other few studies.
It’s been shown that the neuroparenchymal ischemic complications are mostly related to dissemination of embolic material via arterio-arterial anastomosis between dural and neuroparenchymal structures.
Conversely,
a clear anatomic appreciation of these anastomosis is essential to prevent unintentional damage to the brain and cranial nerves.
The middle meningeal artery and its proximal branches gives anastomosis with arteries supplying cranial nerves at the skull base.
The sphenoidal branch of the middle meningeal artery supplies the oculomotor,
trochlear,
and
abducens nerves,
the cavernous branch,
and the trigeminal nerve,
with the petrosal branch supplying the facial and vestibulocochlear nerves.[11],[12]
Embolization causes histopathologic changes within meningioma,
including necrosis,
ischemic changes,
and microvascular fibrinoid changes.[13]
These changes may make histologic examination of embolized meningiomas more difficult because they may histopathologically resemble higher grade tumors.
[13],[14]
The leptomeningeal anastomoses developed through the tumor bed itself,
may lead to particular risks with ultrasmall particles,
thus connecting internal and external carotid causing parenchymal ischaemic infarction,
which was shown by (Benzodus et al.) [8].
This complication can be avoided as described by (kai Y.
et al.,
Horton JE.
et al.) [15],
[16] by provocation tests with Lignocaine and Amytal injections before progressing with embolization procedure.
Using liquid glue in distal catheter locations and particles in more proximal results in lower rate of complications; little more than 1% permanent complications.[7]
Time from embolization to the surgery is another factor.
Two to nine days are the best time as it allows necrosis to develop,
edema to lessens,
and tumor softening,
yet it seemed to have no significant effect on the number of blood units transfused even when other factors including “tumor location” and the “degree of devascularization” were taken into account as shown by (Borg A.
et al.) [7]
Hemorrhage happening in most cases is usually of subarachnoid origin [17] and may also be caused by obstruction of the venous outflow by embolic material,
resulting in high transmural intratumoral pressure.
Tumor infarction itself may result in hemorrhage due to edema and venous obstruction or as a result of the rupture of pathologically fragile tumor vessels.[18]
The greatest tumor softening occurred 7-9 days after embolization.
[19]
Surgery time and blood volume transfused were significantly lower in cases who had been embolized at least 7 days before definitive surgery.
[20]
Intraoperative blood loss was greater for the immediate group (<24h) than for the delayed group.
[21]
Tumors smaller than 6 cm,
the blood lost during the operation was significantly less in the embolized group.
In tumors larger than 6 cm,
there was not difference in blood lost.
[22]
Using this protocol (PVA) no embolization-related complications have occurred over the last 9 years.[5]
(Bendszus M.
et al.) [8] found that only complete embolization (>90%) had an actual effect on blood loss.
(Borg A.
et al.) [7] also stated that blood transfusion was significantly lower after embolization,
however,
he reported four patients had interventional complications.
Steve W.
Hetts commented on (Borg A.
et al.) [7],
stating that “The most significant complications in this series occurred during the use of liquid embolic agents in 2 relatively high-risk arterial territories: the first one the anterior choroidal artery and the second dural skull base of internal carotid artery branches.
This reminds us of the importance of tailoring the aggressiveness of embolization to the expected surgical risk of the specific tumor being addressed .
Overall,
however,
this report underscores the overall safety of preoperative embolization of presumed intracranial meningiomas,
with a trend toward reducing operative blood loss.”
Technical difficulties and anatomical considerations:
Although some studies proved the procedure would decrease blood loss and facilitates tumor resection,
it is difficult to draw definitive conclusions regarding the indications and usefulness of embolization because of the lack of randomized controlled trials.
The entire literature and standards of practice are drawn from level III evidence derived from case series.
The potential complications arising from embolization and the added cost of intervention have limited its use to a subset of meningiomas,
as stated by (Yoon N.
et al.)[23]
There situations that preoperative embolization may be beneficial,
like difficult vascular supply and access; edema that obfuscates the surgical plane,
involvement of dural sinuses,
scalp,
and calvaria; and location near the eloquent cortex.[18],
[5],
[19]
Preoperative embolization may even eliminate the necessity to surgically excise meningiomas,
as reported by (Bendszus et al.) [9],
who treated seven such patients with embolization without subsequent surgery.
They used trisacryl gelatin microspheres to embolize external carotid feeders.
They were able to achieve angiographic devascularization in the five patients whose tumors were supplied exclusively by the external carotid; the internal carotid fed part of the tumor in two patients and was not embolized.
(Campero A.
et al.) [24], demonstrated that the transzygomatic approach combined with mini-peeling of the anterior third of the middle fossa is effective for early devascularization of sphenoid wing meningiomas,
which also becomes handy during resection of large tumors.
(Yoon N.
et al.) concluded that subtotal embolization may not be associated with any reduction in operative blood loss,
whereas aggressive embolization may be associated with serious complications.
Thus,
the needs of thorough devascularization of the tumor must be balanced with the attendant risks of a more aggressive approach,
advocating adoption of a conservative approach that favors complication avoidance over more aggressive devascularization,
which means avoiding any feeders arising from the ICA and using different devices based on the
vascular anatomy,
such as ≥ 300-μm PVA particles when there is risk of CN palsy,
and NBCA or Onyx only when the microcatheter is sufficiently distal to any potentially hazardous anastomotic connections.[23]
Dangerous anastomosis and choice of embolizing material
Small PVA particles (45–150 mm) are more effective in preoperative devascularization than larger particles (150–250 mm) due to capability of more distal penetration into the capillaries feeding the tumor,
yet shows higher risk of complications such as cranial nerve palsies and postprocedural hemorrhage.
Larger PVA particles (> 400 mm) are associated with fewer complications,
but they result in less effective devascularization because they cause occlusion of larger vessels,
which may lead to revascularization via collateral blood supply.[25]
(Przybylowski CJ.
et al.) [26] states that use of Onyx should be reserved for dura matter based arterial pedicles without a distal supply to cranial nerves or extracranial-intracranial anastomoses.
There are major complications such as hemiparesis,
cranial nerve palsies,
tumoral swelling,
ischemia,
or hemorrhage.
Minor neurological complications included headache,
dizziness,
and vomiting.[Table 4] outlines the complications and material used in some of studies with considerable number of patients (∑=689).
Limitations and Potential
Till now there has been no reliable comparative control group.
Data about surgical outcomes of the previously embolized meningiomas are still limited,
extent of tumor resection and recurrence rates.
A randomized trial comparing preoperative embolization with surgery and a surgery only control would elucidate the potential benefits of preoperative embolization for intracranial meningiomas.
For now,
we must carefully select ideal cases for preoperative embolization,
in which the safety and clinical benefit of the procedure has been clearly demonstrated,
eg,
highly vascular large meningiomas.

![Table 1: [table1] including 25 most outstanding studies, with different techniques and variable embolizing materials. References: Sherif M. Shalaby, MSc & Wael Shawaf, MD](https://epos.myesr.org/posterimage/esr/ecr2019/147126/media/784851?maxheight=150&maxwidth=150)
![Table 2: 4 cases of our practice [Table 2] References: Wael Shawaf, MD](https://epos.myesr.org/posterimage/esr/ecr2019/147126/media/784852?maxheight=150&maxwidth=150)
![Table 3: [Table 3] shows the different embolizing material used and advantages,and disadvantages of each References: Sherif M. Shalaby, MSc & Wael Shawaf, MD](https://epos.myesr.org/posterimage/esr/ecr2019/147126/media/784853?maxheight=150&maxwidth=150)
![Table 4: [Table 4] outlines the complications and material used in some of studies with considerable number of patients ( References: Sherif M. Shalaby, MSc & Wael Shawaf, MD](https://epos.myesr.org/posterimage/esr/ecr2019/147126/media/784854?maxheight=150&maxwidth=150)