Lymphangioleiomyomatosis
Lymphangioleiomyomatosis (LAM) is a rare and progressive disease that is almost exclusive to women of child bearing age and generally leads to respiratory failure 1,4,5,6 Prevalence in the UK is approximately 1 per million,
although the incidence increases in patients with tuberous sclerosis complex to 1% 1,4,6.
LAM is caused due to proliferation of atypical smooth muscle cells (LAM cells) particularly in pulmonary lymphatics,
small pulmonary vessels and airways,
causing obstruction and leading to the formation of cysts 4,5.
Other respiratory complications caused include recurrent pneumothoraces in 50% of patients,
chylous effusions and less commonly haemoptysis1,5,6.
Patients often initially present with pneumothorax 1,5,6,7.
Along with respiratory features LAM is associated with: formation of angiomyolipomas (AML) 53% of which are renal; lymphadenopathy,
lymphangioleiomyomas (large lobulated masses) and chylous ascites1,5.
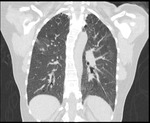
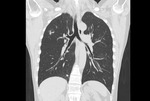
A CXR will usually appear normal in the early stages of disease,
but as it progresses lung volumes may appear hyper-expanded and course reticular shadowing (due to cyst walls),
cystic changes if over 1cm and Kirley B lines may become evident1,5,6.
Pneumothorax and pleural effusions may also be seen.
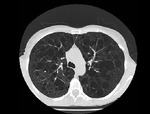
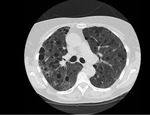
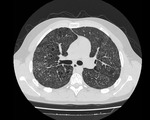
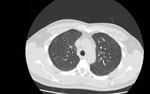
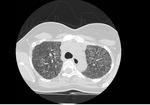
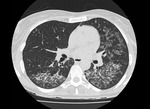
HRCT demonstrates characteristic diffuse distribution of multiple thin walled rounded cysts with surrounding ‘normal’ parenchyma,
without zonal preference 1,4,5. As the disease progresses cysts may amalgamate5.
Broncho-vascular bundles can be seen at periphery of cyst walls,
helping to differentiate it from emphysema,
where the bundle is central1.
Pneumothorices and chylothorax suggested by low attenuation may be present1,6. Occasionally patchy ground glass appearance due to haemorrhage,
interlobular septal thickening due to dilatation of lymphatics,
enlargement of thoracic duct and mediastinal adenopathy may be seen1,,5,6.
On a V/Q scan speckling and well defined hot spots may be seen due to altered diffusion1.
Fig. 1 Fig. 2
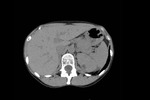
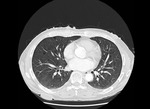
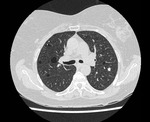
Abdominal findings are also present in up to 70% cases with over half of patients having AMLs present in kidneys1,4,6.
AML’s can be suggested on CT by areas of fat density <10 HU.
Fig. 3
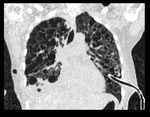
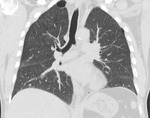
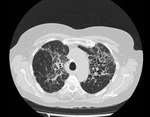
The presentation of LAM on HRCT is classical and hence may prevent the need for biopsy if classical cystic appearance is seen in conjunction with supportive features such as AML and a compatible history5.
Fig. 4
Langerhans cell histocytosis
Langerhans cell histocytosis (LCH),
is a rare granulomatous disorder that predominantly occurs in young adult smokers1,5,6.
Mean age of onset is 28-38 years with a shift over years from male dominance to equal distribution amongst females and males,
likely due to a shift in smoking habits5,7.
90% of patients diagnosed with it have a smoking history4.
The disease is rare in the African-American or Asian population5.
LCH is a spectrum of disease ranging for single organ to more aggressive multi-organ disease including bone,
lung (pulmonary Langerhans histocytosis – PCLH),
pituitary gland,
mucous membranes,
skin,
lymph nodes,
kidney and liver 5,7,8.
Langerhans cells are dendritic cells that function as antigen presenting cells to stimulate lymphocyte proliferation.
Smoking is believed to cause activation of Langerhan cells leading to granulomatous infiltration in bronchiole epithelium and cavity formation,
with occasional interstitial involvement4,5,6.
A typical sequence of nodule formation followed by cavity formation,
thick walled cyst and finally thin walled cysts can be appreciated on imaging 1,6.
As with LAM these patients often present with recurrent pneumothorax and dyspnoea.
In 15-30% of cases they may also have systemic symptoms such as fever,
cystic bone disease and diabetes insipidus 5.
Up to 25% are asymptomatic with disease only being found incidentally on CT7.
Plain chest imaging usually shows upper and mid zone diffuse reticulo-nodular opacities with superimposed small cysts/ring shadows although cavitation may be difficult to appreciate on a CXR1,5.
Initially nodules predominate with a cystic pattern seen in later stages.
This change is usually symmetrical bilaterally with sparing of the costophrenic angle5,6.
HRCT is more sensitive and shows characteristic upper and mid zone irregular shaped cystic structures with variable thickness walls and various sized interstitial nodules with variable degrees of cavitation 1,4,5,6,8. Nodules are usually smaller than 1cm,
spare the costophrenic sulci and may be only sign of disease in early stages 5.
This combination is so characteristic it can be used to provide a diagnosis in correlation with a young smoker7.
Lymphadenopathy and consolidation are rarely seen which help differentiate this from sarcoidosis1,4.
As with LAM cysts may decrease in size on expiratory phase images due to continued communication with airways4.Serial imaging may also be useful in suggesting a diagnosis where a progression of nodular and cyst formation can be appreciated4,5.
Occasionally rib involvement with lytic lesions may be seen4.
Fig. 5
However where there is an atypical pattern such as ground glass attenuation,
adenopathy or changes involving lower lobes further evaluation may be required.
In these cases bronchioalviolar lavage (BAL) or lung biopsy will be required to confirm diagnosis.
Tuberous Sclerosis
Tuberous sclerosis (TSc) is an autosomal dominant germline mutation in tumour suppressor genes leading to hamartoma formation,
cortical tubers and benign neoplastic lesions that affect multiple organs5,,9.
Patients normally have a classic triad of mental retardation,
facial angiofibromas and epilepsy3,4,5.
Radiology criteria require presence of calcified subpendymal nodules or non- calcified nodules with cortical tubers,
on CT head.
Imaging may also demonstrate cardiac rhabdomyoma,
renal AML,
retinal harmatomas and subpendymal giant cell astrocytomas9.
Lung involvement of TSc has a cystic appearance identical to LAM.
Bronchopneumonia may also be present.
However it can be found in both sexes and you would also expect the patient to have the classical triad of symptoms and other radiological features,3,4,5,9.
Neurofibromatosis
Neurofibromatosis is a rare autosomal dominant multisystem disease.
Neurofibroma’s are tumours which develop from the nerve sheath.
Clinically it requires a combination of café au lait spots,
and neurofibroma’s for diagnosis1,5,9.
In addition axillary freckling,
optic lesions,
lisch nodules,
skeletal abnormalities such as sphenoid dysplasia,
CNS lesions (cerebral glioma),
vascular lesions (stenosis/aneurysms) and lung pathology may also be found1,5,9 .
Cranial abnormalities are often appreciated better on MRI.
Chest imaging may show large thin walled cysts with asymmetric apical predominance1,5.
Multiple subcutaneous neurofibromas will be visible on the chest wall1. Also visible may be rib abnormalities,
mediastinal/ intrathoracic masses,
peripheral pulmonary nodules and progressive interstitial fibrosis of lower lung predominance1,9.
Fig. 6
Other common cystic presentations
Desquamative interstitial pneumonitis (DIP)
This is an idiopathic interstitial pneumonia that primarily affects cigarette smokers1.
It makes up less than 3% of ILD4.
Mean age of onset is 45 with a male to female ratio of 2:15,7.
Prognosis is usually good with a mean survival of 12years 1,5.
Histologically DIP is represented by a large number of pigmented macrophages within the airspaces with mild interstitial fibrosis and inflamation1,5,7.
It has a diffuse and uniform involvement unlike respiratory bronchiolitis-ILD which has a patchier and bronchiolocentric distribution5.
Patients usually present with a background of smoking and a history of Dyspnoea or dry cough7.
Chest radiographs can be normal 3-22% of the time 1,7.
However lower lung ground glass opacities with peripheral predominance may be seen,
sparing costophrenic angle in 33% of cases1,7.
The principal abnormality on HRCT is bilateral,
middle and lower zone peripheral ground-glass opacity,
which is often symetrical1,7.
Cystic lesions may develop within the areas of ground-glass opacity7. Irregular linear opacities,
honeycombing and traction bronchiectasis may also be appreciated,
with lower lung fibrosis being found in later stages1,7.
Emphysema may exist in combination with DIP.
Smoking cessation is the main treatment for DIP along with oral corticosteroids,
despite the lack of randomized trials demonstrating their efficacy7.
Fig. 7 Fig. 8
Lymphocytic interstitial pneumonitis
Lymphocytic interstitial pneumonitis (LIP) is a benign lymphoproliferative condition that commonly affects women around the age of 50 1,5,9. Histologically infiltration by lymphocytes and plasma cells into bronchovascular bundles,
interlobular septa and pleura is found1,5,8.
Aetiology is unclear,
although Epstein Barr Virus has been a common factor in some patient 5.
It is a disease that rarely occurs in isolation and is commonly associated with other conditions such as Sjogrens,
autoimmune conditions and AIDs1,5,9.
HRCT demonstrates fine reticular/reticulonodular shadowing bilaterally across all areas1,5.
Diffuse areas of bilateral ground glass opacities,
poorly defined centrolobular nodules,
thickened bronchovascular bundles and interlobular septa,
subpleural nodules,
consolidation,
mediastinal lymph node enlargement and parenchymal cysts can be visulised1,5,8.
Cysts are found in nearly 70% of patients and are due to partial airway obstruction.
They are distributed diffusely throughout affected lungs1,2.
It is generally steroid responsive and hence follow up scans should show improvement.
Fig. 9 Fig. 10
Diagnosis is generally via biopsy or BAL5.
Amyloid associated cystic lung disease
Amyloidosis is a systemic disease characterised by extracellular deposition of abnormal amyloid fibrils.
Lung involvement is seen in up to 70% primary amyloidosis1. It has a poor prognosis with mean age of onset being 655.
Radiological pulmonary findings include diffuse reticulo/ reticulonodular infiltrates with enlarged hilar/mediastinal lymph nodes that are often calcified 5.It can also present as cystic lung disease,
with varying sized cysts usually located along periphery of lung parencyma2.
Interlobular septal thickening,
honeycombing and ground-glass opacities may also be visulised2.
Fig. 11
Cystic sarcoidosis
Sarcoidosis is an immune mediated multiple organ granulomatous disease of unknown aetiology that is more common in females and afro-Caribbean patients 1,4.
The age of onset is 20-40 years with females having a further incidence peak aged 501,5.
The hallmark of sarcoidosis is non-caseating epithelioid granuloma located along the course of lymphatic vessels,
although fibrosis may develop alongside them1,5.
These form primarily in the lungs 5.Patients may present with symptoms of fatigue,
dry cough,
erythema nodosum,
night sweats,
weight loss and dyspnoea5,8.
Sarcoid usually presents on CXR with symmetrical hilar and right paratracheal lymphadenopathy dubbed the 1,
2,
3 sign or Garland triad4 .
Over 90% patients are initially diagnosed on CXR,
50% as a result of incidental findings5,8.
Clinical staging is also often based on CXR interpretation.
HRCT demonstrates both adenopathy and parenchymal disease.
Irregular septal thickening,
perilymphatic nodules along bronchovasculature,
subpleural and interseptal lymphatics,
traction bronchiectasis and progressive mid to lower zone fibrosis are common1.
Also seen is ground glass opacities,
irregular bronchial wall thickening and air trapping,1,5,8. Less commonly pleural effusions,
solitary nodules,
bronchostenosis,
and in 0.6% cases cavitation of nodules/ cysts will be appreciated1.
Other common signs such as hepatosplenomegaly,
lymphadenopathy and skeletal abnormalities may be seen1.
Fig. 12
Pneumocytis jiroveci
Pneumocytis jiroveci (PCJ),
previously called pneumocystis is an opportunistic fungal infection more prevalent in HIV when CD4 count less than 200 and is a common cause of death in AIDS4,5.
CXR may show diffuse symmetrical fine reticular changes with a characteristic central distribution 1.
Pneumatoceles or thin walled cysts are seen in 10% of patients within the first few days of disease,
most resolve after a few months but a small number can persist despite treatment9.
The benefit of CT given the cost is questionable however on CT thorax bilateral ground glass opacities,
representing acute pneumonitis can be seen in central portion of lungs 2,4,9, Associated cystic changes may also be appreciated5.
Fig. 13
Lepidic type adenocarcinomas.
A new classification of adenocarcinoma of the lung was established in 2011 to include invasive lepidic –predominant adenocarcinomas (LPA) 10,11.
They display a lepidic (scale like) growth pattern and arise from alveloli and adjacent small airways9,10. Along with the mass/ nodule commonly associated with ground glass opacity seen on CT there may also be: pseudocavitation; Hilar and mediatinal lymphadenopathy; metastases; and air containing cysts9,11.
It is also important to remember as many of these patients are smokers emphysematous changes may also be present.
Fig. 14