Role of radiologists in:
1. Localised disease
This section concerns tumour limited to kidney (T1-T2): TNM 2009 (7th edition) is the same in AJCC/TNM 2017 (8th edition).
Because of expanding indications of partial nephrectomy,
several authors [4,5,6] have tried to validate preoperative scoring to quantify tumour size,
location and depth based on preoperative imaging findings.
American Urological Association guidelines established that partial nephrectomy for management of T1 renal mass is possible when technically feasible [7].
Otherwise,
partial nephrectomy can be a surgical approach also for tumours larger than 7 cm but only for a strictly selected group of patients (solitary kidney,
pre-existing renal insufficiency) with an appropriate tumour location [8].
Infact,
partial nephrectomy preserves renal function (so called “nephron-sparing surgery”) but the risk of post-surgical complications is higher than after radical nephrectomy.
Therefore,
radiologists must provide morphological information which make nephron-sparing approach possible and identify anatomical variants which increase the risk of complications such as haemorrage,
fistulas.
What is the main problem?
Prolonged warm ischemia time (>30minutes): the standard technique of partial nephrectomy includes clamping of renal artery with or without renal vein and sharp excision of the renal mass.
The time a vessel is clamped is the warm ischemia time.
Can radiologists sort out this problem?
Yes.
Identification of renal tumour features which correlate with the risk of prolonged warm ischemia time or with other surgical complications is the aim of different scoring systems [4,5,6]:
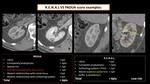
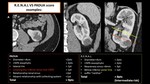
a) Padua-score (Preoperative Aspects and Dimensions Used for Anatomic Assessment),
Fig. 4;
b) RENAL score,
Fig. 5;
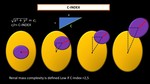
c) C-index (centrality),
Fig. 6 .
In Padua and RENAL-nephrometry scoring systems,
the final score is determined analysing several anatomic characteristics (remeber differences between sinus and polar lines,
Fig. 7).
Instead,
the C-index is calculated with measurements obtained from 2D-CT scan to determine the lengths of two sides of a right triangle.
The Pythagorean theorem is used to calculate the hypotenuse of the triangle.
The hypotenuse is indicative of the proximity of the center of the lesion to the center of the kidney.
This number is divided by the radius of the tumor to obtain the C index.
Presently,
literature is not able to state if one methods is superior to the others; some example cases are shown in Fig. 8,
Fig. 9,
Fig. 10
Additional significant anatomic considerations in planning partial nephrectomy are:
· proximity to ureter or ureters,
Fig. 10.
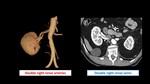
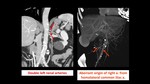
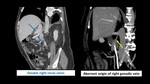
· number of renal arteries and veins present, Fig. 11 Fig. 13
· identification of collateral feeding vessels,
Fig. 14
2. Locally advanced disease (T3–4,
Tx-N1)
This section concerns tumours with different patterns of extrarenal extension (T3 and T4) and lesions with metastases into regional lymph nodes (N1).
I. Perirenal and/or renal sinus fat invasion (T3a):
Although prognostic significance of renal sinus fat invasion and perinephric fat invasion is not clear,
they are considered as predictors of recurrence and cancer-specific survival.
To develop a more precise risk-stratification system,
recent studies demonstrated that the compresence of multiple patterns of extrarenal extension (perinephric fat,
PF,
renal sinus fat,
SF,
and renal vein,
RV) is associated with an higher risk of disease progression after radical nephrectomy compared to isolated involvement of the PF,
renal SF,
or RV [9,10]).
Otherwise based on available data,
imaging preoperative assessment of fat invasion lacks to significantly influence surgical strategy.
II. Tumour Thrombus (T3a,
b,
c)
Invasion of the renal vein and inferior vena cava (IVC) is present in 4–23% of RCC due to its characteristic venotropism.
All the imaging modalities (ultrasound scan,
computed tomography and magnetic resonance imaging) can be used to tumour thrombus assessment: its extension is predicted with 96% accuracy with MDCT,
which is considered the most effective modality for preoperative evaluation and surgical planning [11].
Role of MRI and US are not clear and considered useful in selected and challenging patients.
Vascular involvement in RCC is an independent predictor of perioperative morbidity and mortality.
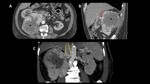
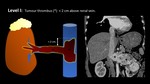
Extension of IVC involvement has been defined using five levels,
Fig. 15 :

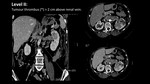
Fig. 15: Tumor Thrombus level
0 renal vein;
I,
< 2 cm above renal vein,
Fig. 16 and Fig. 17;
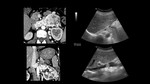
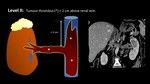
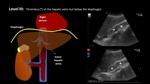
II,
> 2 cm above renal vein but below hepatic veins,
Fig. 18 and Fig. 19;
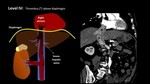
III,
at the hepatic veins but below the diaphragm,
Fig. 20;
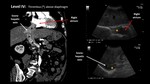
IV,
above the diaphragm,
Fig. 21 and Fig. 22.
A review showed how perioperative complication rates increased with tumour thrombus level (level 0,
12%; I,
18%; II,
20%; III,
26%; IV,
47%) [12].
For these reasons,
preoperative evaluation of a tumour thrombus and its extension are useful not only as prognostic factor (T3a vs T3b/c) but also to decide a lower risk surgical approach: level III/IV often require a midline incision which allows a major exposure of IVC/renal pedicles and access to the right atrium through extension of the incision via-sternotomy. Moreover,
for selected level III and most level IV tumour thrombi,
vascular bypass (cardiopulmonary or veno-venous) can be necessary.
We have already discussed that in the revision of AJCC/TNM classification,
Fig. 2
Another significant prognostic factor is invasion of the IVC wall (rather than luminal infiltration) by RCC tumour thrombus,
in this case preoperative MRI has 90% of sensitivity in estimation of wall invasion.
Also in this case,
preoperative identification of venous wall invasion may guide treatment choice (extention of IVC resection and reconstruction or neoadjuvant therapy).
III. Nodal Involvement (TX–3aN1)
Another critical element for determination of disease stage is the preoperative detection of lymph node involvement.
This is one of the most significant prognostic factors in RCC: N1 doubles patient's risk of distant metastasis and significantly reduces their 5-year survival [13].
The problem is that clinical node staging is inaccurate (only 42%) and the role of lymphadenectomy in the surgical treatment is controversial [14].
Several attempts were made to establish how select patients:
low-risk primary tumours, N0M0,
lymphadenectomy is unnecessary
vs
high risk tumour, M0,
lymphadenectomy for a correct staging but also to obtain therapeutic benefit.
Five high-risk features were proposed: tumour size larger than 10 cm,
grade 3 or 4,
category pT3 or pT4,
histologic tumour necrosis,
and presence of a sarcomatoid component.
Presence of 2 or more features identified high risk Patients.
Standard templates for nodal dissection are recommended [15] and typically include,
Fig. 23 Fig. 24:
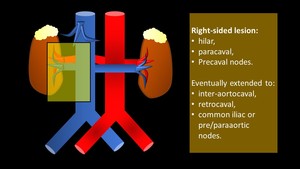
Fig. 23: Right-sided tumors nodal dissection: hilar, paracaval, and precaval nodes, with few authors extending the dissection to the inter-aortocaval, retrocaval, common iliac or pre/paraaortic nodes
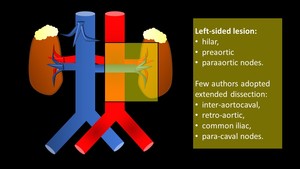
Fig. 24: Left-sided tumor nodal dissetion: hilar, preaortic and paraaortic nodes, with few authors reporting a systematic dissection of inter-aortocaval, retro-aortic, common iliac, or para-caval nodes
Preoperative identification of nodal disease is critical for surgical planning to reserve more aggressive resection only when necessary to obtain a radical attempt.
IV. Adrenal gland and adjacent structures (T4)
Contiguous extension into the ipsilateral adrenal gland is now considered T4 disease. Historically,
radical nephrectomy included surgical excision of the kidney and Gerota fascia,
ipsilateral adrenalectomy with lymphadenectomy from the crus of the diaphragm to the bifurcation of the aorta.
But also the necessity of adrenalectomy has been discussed because of the low incidence of adrenal invasion and the morbidity due to adrenal insufficiency.
In a recent review,
adrenalectomy is recommended if preoperative imaging findings demonstrate [16]:
-venous tumour extension,
-upper pole tumours,
-tumours larger than 7 cm.
Moreover,
RCC involving adjacent organs or structures (T4) has a poor prognosis [17,18].
But T4 lesions often are down-staged after surgical resection and for this reason surgical resection should be offer to all patients who are physically able to tolerate surgery and have lesions considered “resectable” (eventually requiring additional surgical teams).
Colon,
pancreas,
diaphragm,
liver,
spleen,
and bowel mesentery the most common involved structures.
The crucial point is the definition of surgical resectability: it is not univocal and depends on multiple factors (surgeon experience,
availability of multidisciplinary surgical teams and of ancillary services,
tumour and anatomy-specific factors).
However radical nephrectomy remains the standard of care for locally advanced RCC.
So,
When neoadjuvant therapies have a potential role?
1.
Tumour downsizing
2.
Downstaging IVC thrombus (lacks consistent benefit)
3.
Changing the unresectable to resectable
4.
Changing radical nephrectomy to partial nephrectomy (for patients with imperative indications for nephron sparing surgery).
A recent review did not recommend routine use of neoadjuvant-therapy in patients who otherwise have resectable disease [19].
Borregales et al suggested it in the setting of clinical trials or as a last chance in very selected and well-informed patients after multidisciplinary discussion.
3. Assessment of response to systemic therapies
In the last years,
there was a revolution in the management of advanced RCC.
Three main classes of anticancer agents are used:
1. antiangiogenic agents,
2. mTOR inhibitors,
3. immune checkpoint inhibitors.
Traditional Response Evaluation Criteria in Solid Tumors (RECIST) 1.0 and the revised RECIST (RECIST 1.1) are based on tumour size variation.
But in patients treated with antiangiogenic agents,
changes in tumour vascular density have been shown to correlate with treatment response and treatment resistance [20].
So purely size-based RECIST criteria are often inadequate,
sometimes even misleading,
for accurate response assessment with targeted therapies.
Criteria which incorporate both changes in tumour size and CT appearance must be used: Choi criteria,
revised Choi criteria,
or MASS,
Fig. 25.
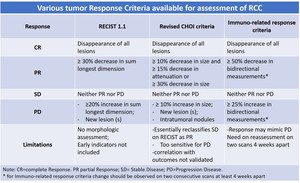
Fig. 25: Some tumor Response Criteria available for assessment of RCC
Also for response assessment in Immune Checkpoint Blockade,
new immune-related response criteria were developed [20]; the immune-related response criteria differ from RECIST.
It is important for the radiologists have to familiarize with different response patterns associated with these new therapeutic agents:Patient could be wrongly deprived of effective therapy.