Pancreatic neuroendocrine tumors (pNETs) are rare neoplasms that arise in the endocrine tissues of the pancreas.
They have an incidence of ≤1 case per 100,000 individuals per year and account for 1 to 2 percent of all pancreatic tumors.
Pancreatic NETs represent less than 3 percent of primary pancreatic neoplasms.
Incidence rates have been increasing and elsewhere over the last two decades,
but it is likely that this is mainly related to increased detection of asymptomatic disease on cross-sectional imaging and endoscopy done for other reasons.
Overall,
there is no significant gender predilection.
Although they may manifest at any age,
they most often occur in the fourth to sixth decades of life.
Most pancreatic NETs are sporadic,
but they can be associated with hereditary endocrinopathies,
including multiple endocrine neoplasia type I (MEN1),
von Hippel Lindau (VHL) syndrome,
neurofibromatosis type I (NF1),
and tuberous sclerosis (TE).
Patients who develop a pancreatic NET in the context of an inherited syndrome must be considered separately with regard to prognosis,
as these tumors tend to be associated with a more indolent course than are sporadic tumors.
Other potential risk factors for pancreatic NETs include smoking,
diabetes and a previous history of chronic pancreatitis.
-Clinical presentation:
They can secrete a variety of peptide hormones,
including insulin,
gastrin,
glucagon,
and vasoactive intestinal peptide (VIP),
resulting in countless clinical syndromes.
In the past,
they have been categorized as either functioning or non- functioning.
However,
because all of the neoplasms are hormonally active,
it is more accurate to classify them as either syndromic or nonsyndromic,
on the basis of clinical and laboratory findings.
Recent clinical series describe the majority (50-80%) of pancreatic NETs as nonsyndromic.
In general,
syndromic tumors tend to manifest earlier and have a smaller size than other nonsyndromic endocrine tumors,
findings likely due to the dramatic clinical syndrome.
Symptoms such as abdominal pain,
weight loss,
an abdominal mass,
and,
rarely,
jaundice are a result of mass effect.
The frequency of metastatic disease at the time of diagnosis has been reported to be as high as 60%–80%.
-Pathophysiologic features:
Measures of proliferative index,
Ki-67 and mitotic index,
are used to assign histologic grade for pancreatic NENs.
Prior to 2017,
well-differentiated pancreatic NETs were separated into low-grade (G1,
Ki-67 index <3 percent) and intermediate-grade (G2,
Ki-67 index 3 to 20 percent) categories according to proliferative rate.
High-grade tumors (G3,
Ki-67 index >20 percent) were considered equivalent to poorly differentiated carcinomas.
However,
it became apparent that not all G3 tumors were poorly differentiated and that some tumors,
particularly with a ki-67 index in the 20 to 55 percent range,
had relatively well-differentiated histology,
relatively good prognosis (compared to poorly differentiated carcinomas),
and relatively poor response to platinum-based chemotherapy.
Therefore,
the 2017 WHO classification of pancreatic NENs includes a NET G3 category (ki-67 >20 percent) that must be distinguished from the poorly differentiated pancreatic NEC category.
Most pNETs are composed of well-differentiated endocrine cells.
These cells typically form nests,
trabeculae,
or ribbons.
The neoplastic cells are small to medium in size and characteristically contain uniform round nuclei with “salt-and-pepper” chromatin.
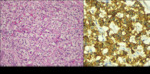
The endocrine differentiation can be confirmed with a Grimelius silver stain or with immunohistochemical labeling for chromogranin or synaptophysin (Fig. 1).
Specific hormone production by the neoplasms can also be confirmed with immunolabeling.
Predicting the clinical behavior of a well-differentiated endocrine neoplasm of the pancreas can be extremely difficult.
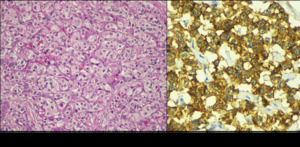
Fig. 1: (a) Epitelial cells nests and trabeculae, with granular cytoplasm and central nucleus with size variability and fine chromatin separated by thin septa. (b) Cytoplasm positivity to synaptophysin.
The only unequivocal criteria for malignancy are gross invasion of adjacent organs,
large vessel invasion,
and metastases to lymph nodes or distant organs.
Histologic differentiation between benign and malignant tumors is difficult because tumors that metastasize are not necessarily histologically different from those that do not.
It is not usually possible to distinguish syndromic from nonsyndromic tumors with immunohistochemical staining alone.
Tumor size is an important factor in predicting the behavior of nonsyndromic pNETs.
They vary ranging from smaller than 1 cm to larger than 20 cm,
with most tumors being 1-5 cm in diameter.
Smaller tumors tend to be more homogeneous,
whereas larger tumors more commonly are heterogeneous with areas of cystic change,
necrosis,
and calcification.
They are solitary tumors except when they are associated with familial syndromes.
Larger tumors also tend to behave more aggressively and are more likely to metastasize.
The presence or absence of metastases is the major predictor of survival.
The most common site of metastatic disease involvement for pancreatic NET is the liver.
Well-differentiated pNETs don´t have a defined capsule but they are characteristically well circumscribed (unlike adenocarcinomas).
They displace,
rather than invade,
adjacent structures,
although larger neoplasms may have a multinodular border and demonstrate macroscopic invasion.
In contrast,
poorly differentiated pNETs have ill-defined borders and abundant necrosis.
-General Imaging features:
Imaging is used for localization of known syndromic pNETs,
diagnosis of nonsyndromic pNETs and surgical planning.
1.-US
Pancreatic NETs appear as well-circumscribed round or oval hypoechoic masses with smooth margins.
They may be heterogeneous or homogeneous.
The sensitivity of transabdominal US for detection of pNETs varies from less than 20% to as high as 80%.
2.-CT
Most NETs are highly vascular.
They show peak contrast enhancement in the early arterial phase (25-35 secs) rather than in the late arterial phase (35-45 secs) which is normally used for pancreatic imaging.
This is particularly important when considering that small lesions may be missed in the late arterial phase when tumor will appear isointense with enhancing pancreatic parenchyma.
Several studies have shown promising results in the preoperative detection of pNETs with dual phase CT (early arterial phase and portal venous phase),
which typically demonstrates a sensitivity exceeding 80%.
Tumors as small as 4 mm have been visualized with CT scanning; however,
sensitivity is decreased for tumors smaller than 2 cm in diameter compared to larger tumors.
Small tumors can often appear as rounded,
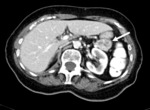
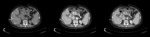
enhancing vascular lesions (Fig. 2, Fig. 3).
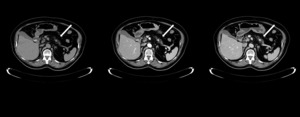
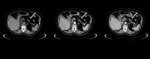
Fig. 2: Typical appearance of pancreatic NET in a 73-year-old woman. (a) Nonenhanced CT scan shows a rounded, well- defined 1 cm lesion at the junction of the pancreatic body and tail (arrow) with avid enhancing in the arterial phase (b) and late venous phase (c).
Larger lesions are more likely to demonstrate heterogeneous enhancement,
a finding due to areas of cystic degeneration,
necrosis,
fibrosis,
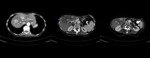
and calcification (Fig. 4, Fig. 5).
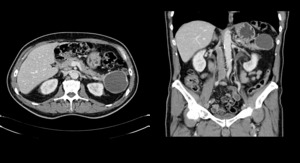
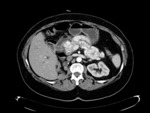
Fig. 4: A 68 year old man with a pancreatic NET with cystic appearance. Late venous phase contast-enhanced CT axial (a) and coronal scan (b) shows a 5 cm cystic lesión with peripheral calcification in the pancreatic tail. Note the hypervascular rim, which is seen in 90% of cystic NETs, that allows them to be distinguished from other cystic neoplasms of the pancreas.
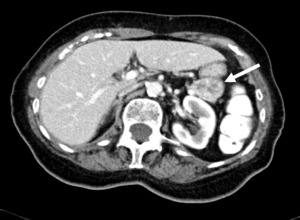
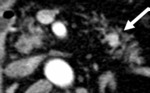
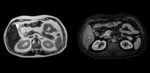
Fig. 5: Pancreatic NET in a 77-year-old woman with a constitutional syndrome and loss of weight. Arterial phase contrast-enhanced CT scan with oral contrast demonstrates a 4 cm well-defined heterogeneous pancreatic tail mass (arrow) with intense peripheral enhancement and central degeneration.
Cystic pancreatic NETs typically have a hypervascular rim,
a finding that allows them to be distinguished from other cystic neoplasms and that,
according to several studies,
is seen in 90% of cystic NETs.
Lymph node and liver metastases also are hypervascular and therefore more conspicuous on arterial phase images.
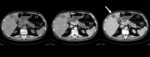
Hepatic metastases often demonstrate ringlike enhancement (Fig. 6, Fig. 7).
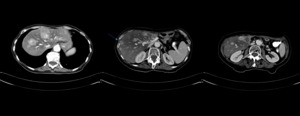
Fig. 6: Hypervascular hepatic metastases in a 68-year-old woman with a pNET excised 10 years ago. Arterial phase contrast-enhanced CT scan reveals multiple enhancing lesions in the liver, some of them are with ring-enhancing (arrow).
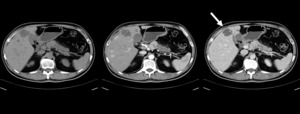
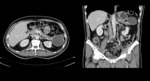
Fig. 7: Pancreatic NET in a 47-year-old man with hepatic metastases. (a) Non contrast, (b) arterial phase and (c) late venous phase contrast-enhanced CT scan showing a well defined enhancing mass in the pancreatic body with dilatation of the pancreatic duct and multiple enhancing lesions in the liver, some of them are ring-enhancing (arrow).
3.-RM
MR imaging has sensitivity similar to that of multiphasic CT for the detection of pNETs.
Pancreatic NETs appear as relatively hypointense,
circumscribed masses on T1-weighted images and most pNETs demonstrate signal intensity much higher than that of a normal pancreas on T2-weighted images.
They enhance more than normal pancreas on contrast-enhanced arterial and venous phase images (Fig. 8, Fig. 9).
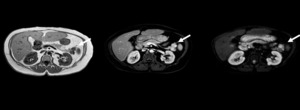
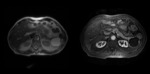
Fig. 8: Pancreatic NET in a 55-year-old woman. MRI shows (a) a bilobulated hipointense lesión in the pancreatic tail (arrow) in T1 without fat-suppresion, (b) which enhances in the arterial phase contrast T1 with fat-suppresion and (c) it has restiction on diffusion sequence. There is also a cyst in the left kidney.
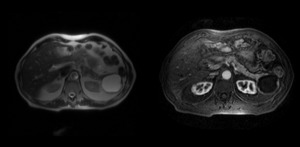
Fig. 9: Same patient as figure 4. (a) T2 MRI shows a cystic leison in the pancreatic tail with hyperintense content . (b) Arterial phase enhanced T1 with fat-suppresion MRI reveals an image with hypointense content and hypervascular rim.
As with CT scans,
early arterial phase imaging following the injection of gadolinium contrast is critical for the detection of small hypervascular liver metastases.
4.-Nuclear medicine
Diagnostic imaging using radiolabeled somatostatin analogs is recommended for most patients because many pancreatic NETs express high levels of somatostatin receptors.
Uptake of radiolabeled somatostatin analogs is predictive of clinical response to therapy with somatostatin analogs and peptide receptor radionuclide therapy,
and it can assist in identifying an otherwise occult primary site.
111-In pentetreotide has proven effective for visualizing gastrinomas,
glucagonomas,
and nonfunctioning pancreatic tumors.
Because insulinomas and poorly differentiated neuroendocrine carcinomas express relatively scant levels of subtype 2 somatostatin receptors,
they are less likely to be detected on 111-In pentetreotide scanning.
Where available,
functional imaging with Gallium Ga-68 DOTATE (68-Ga DOTATATE) positron emission tomography (PET)/CT is preferred over indium-111 (111-In) pentetreotide (OctreoScan) due to its greater sensitivity.
5.-Occult hormonally-functioning p NET.
Occasionally,
patients present with a hormonal syndrome suggestive of a pancreatic NET,
but lack evidence of disease on conventional imaging.
The combined use of conventional imaging and endoscopic studies for an occult hormonally-functioning pancreatic NET has improved the sensitivity of preoperative detection to nearly 100 percent.
Endoscopic ultrasonography (EUS) provides high-resolution imaging of the pancreas,
and it can detect lesions as small as 2 to 3 mm in diameter.
Another benefit of EUS is that EUS-guided fine-needle aspiration biopsy can often provide a non-operative histologic diagnosis of pancreatic NET.
EUS is limited by the requirement of a highly skilled endoscopist and by its inability to consistently visualize the pancreatic tail.
In rare cases where there is strong clinical and biochemical evidence for an occult pancreatic NET that is undetectable on EUS,
we recommend arterial stimulation with transhepatic portal venous sampling (THPVS) if expertise in this technique is available.
In THPVS small peripancreatic veins are accessed and tested for levels of hormones such as insulin.
A more recent innovation,
arterial stimulation with hepatic venous sampling (ASVS),
involves selective injection of a stimulating secretagogue (secretin for gastrinomas and calcium gluconate for insulinomas) into arteries supplying the pancreas with subsequent sampling of the hepatic venous effluent.
-Specific clinical and imaging features (Table 1).

Table 1: Specific clinical and imaging features
-Differential diagnosis:
The appearance of pancreatic neuroendocrine tumors is relatively distinct,
characterized by their hypervascularity and conspicuity on arterial phase images.
As imaging technology has improved over the last decade,
it has become increasingly common to incidentally discover hypervascular lesions in the pancreas,
which overwhelmingly represent small pancreatic neuroendocrine tumors.
However,
we have encountered a number of incidentally discovered hypervascular lesions in and around the pancreas that can mimic the imaging appearance of pancreatic neuroendocrine tumors including:
-Pancreatic metastases (renal cell carcinoma as the most frequent primary tumor) (Fig. 10,
Fig. 11).
-Intrapancreatic splenules (Fig. 12).
-Peripancreatic paragangliomas.
-Solitary fibrous tumor.
-Pancreatic hamartoma.
-Exophytic gastrointestinal stromal tumors.
-Serous adenoma.
-Pancreatoblastoma.
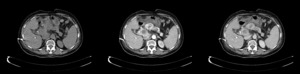
Fig. 10: Pancreatic metastases that mimics pNETs in a patient with a right nefrectomy due to a RCC 10 years ago. (a) Non enhanced, (b) arterial phase and (c) late venous phase contrast-enhanced CT scan demonstrate hyperenhancing lesions in the pancreatic head, body and tail. The two first are larger and heterogeneous with central necrosis.
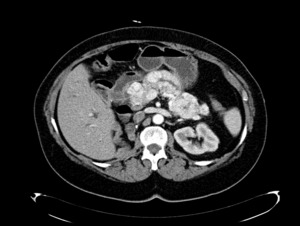
Fig. 11: Pancreatic metastases in an other patient with a right nefrectomy due to a RCC 20 years ago. Arterial phase contrast-enhanced CT scan demonstrate heterogeneous lesions in the pancreatic head, body and tail.
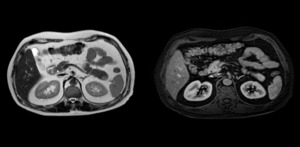
Fig. 12: Intrapancreatic splenule in the pancreatic tail diagnosed after surgery. (a) T2 MRI and (b) T1 contrast-enhanced arterial phase MRI show a well defined nodule isointense with the spleen in all two sequences.
-Treatment:
Surgery is the only curative modality for sporadic pNETs,
and resection of the primary tumor in localized,
regional,
and even metastatic disease can improve patient survival.