Clinical applications: we review various neurovascular and neurodegenerative conditions in which SWI can help in diagnosis.
- Cerebral amyloid angiopathy: It is characteristic in this microangiopathy the deposition of amyloid β protein in the cerebral small arteries leading to fibrinoid necrosis and vessel fragility.
They predominate in the frontal and parietal cortical and subcortical regions.
It causes arteriolar microhemorrhages extending into the parenchyma.
Also it is the main cause of primary lobar hemorrhage.
It is important the differential diagnosis with microhemorrhages resulting from hypertensive or atherosclerotic microangiopathy,
the last one showing predilection for deep gray matter / basal ganglia and infratentorial location.
Routine MRI and CT are much less sensitive than SWI,
CT is usually not able to detect cerebral microbleeds.
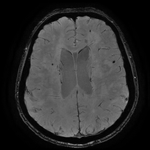
Amyloid deposition is manifested by white matter hyperintensities on T2 sequences (Fig. 5) and may be accompanied by clinical significant cognitive impairment.
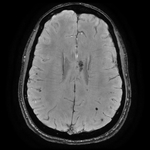
There is highly suspicion of cerebral amyloid angiopathy if we detect cerebral microbleeds in frontoparietal subcortical regions,
hyperintensities in the white matter and sometimes lobar hemorrhages [4],
(Fig. 6).
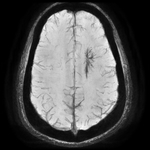
As as consequence of chronic changes in some cases we can detect hemosiderosis on the pial surface of the brain from older repetitive superficial hemorrhages (Fig. 7).

Fig. 5: Cerebral amyloid angiopathy.
T2 Flair with extense white matter lesions.

Fig. 6: Cerebral amyloid angiopathy.
Microbleeds on SWI.
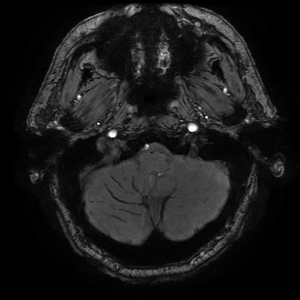
Fig. 7: Cerebral amyloid angiopathy.
Hemosiderosis from older subarachnoid bleeding on SWI.
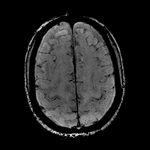
- Hypertensive cerebral angiopathy: In this disease we can detect silent cerebral microhemorrhages but associated with chronic arterial hypertension and in a different distribution from what we see in amyloid deposit helping in the differential diagnosis.
It mainly affects thalamus,
basal ganglia,
cerebellum and pons (Fig. 8).
It is also a risk factor to develop lobar hemorrhages and the number of cerebral microbleeds correlates with blood pressure levels.
SWI sequence is more sensitive to demonstrate these acute deoxyhemoglobin or chronic hemosiderin microbleeds than conventional T2*GRE imaging.

Fig. 8: Typical distribution of hypertensive cerebral angiopathy involving thalamus and basal ganglia. T2GRE.
- Traumatic brain injuries: In this clinical setting SWI sequence has prognostic value.
Diffuse Axonal Injury (DAI) is a type of traumatic brain injury in which torsional forces generated by extreme acceleration or deceleration forces cause hemorrhagic lesions.
The most vulnerable areas are the cerebral gray-white matter junction,
splenium of corpus callosum,
basal ganglia and dorsolateral brainstem.
DAI lesions are seen as small punctate hemorrhagic foci (Fig. 9),
(Fig. 10),
SWI being more sensitive than any other MRI sequence or CT scan in detecting number,
size,
volume and distribution of the lesions [4].
The extent of the axonal injury shows correlation with a poorer prognosis.
Brain stem involvement is a very important predictor of the long term outcome of the patients.
It can show also small intraventricular and subarachnoid hemorrhage not visible on CT [4].

Fig. 9: SWI of Diffuse Axonal Injury (DAI) showing hemorrhagic lesions in gray-white matter junction and corpus callosum.

Fig. 10: MinIP SWI of Diffuse Axonal Injury (DAI) showing hemorrhagic lesion in brain stem.
- CNS vascular malformations: The arteriovenous malformations (AVMs) are high-flow lesions and therefore we can actually detect them on conventional MRI or angiography MRI techniques (Fig. 11).
On the other hand we have low-flow vascular malformations like cavernomas / cerebral cavernous malformations (CCMs),
developmental venous anomalies (DVAs) and capillary telangiectasias.
Sometimes T2*GRE conventional sequence is capable of detecting small venous structures and hemosiderin deposition but SWI sequence is superior in detection and characterization of these lesions adding the information of the magnitude and phase reconstructions.
SWI is the indicated ideal sequence in the study of patients with clinical suspicion of low-flow vascular malformations,
sometimes manifesting with epilepsy,
unexplained recurrent subarachnoid bleeding or hemosiderosis.
Cavernomas or CCMs are enlarged capillary cavities with a single layer of endothelium.
They typically show a popcorn-like aspect and a peripheral rim of hemosiderin,
but we can have different MRI findings depending on the presence of calcification,
acute or chronic blood within the lesion (Fig. 12).
If they grow or are symptomatic sometimes surgical resection is needed.
DVAs are very common and many times discovered incidentally on routine MRI.
They are venous structures in normal brain parenchyma converging to a draining main venous trunk,
usually asymptomatic and very rare bleeding.
SWI shows very well the “head of Medusa” and converging medullary veins,
specially on MinIP reconstruction (Fig. 13).
Capillary telangiectasias are small vascular malformations,
mainly located in the pons and usually some millimeters to 2 cm size.
They are asymptomatic and sporadic,
but sometimes we find them in hereditary hemorrhagic telangiectasia or after radiation therapy.
On T1 image after gadolinium they can enhance faintly and are easily missed on unenhanced studies.
SWI sequence increase the sensibility for detection of these lesions (Fig. 14),
(Fig. 15).

Fig. 11: High flow lesion compatible with AVM in SWI.
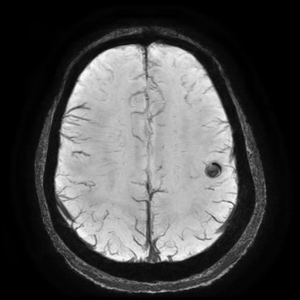
Fig. 12: MinIP SWI with hypointense lesion with hemosiderin deposit compatible with cavernous malformation.

Fig. 13: MinIP SWI showing venous structures ("head of medusa") converging to a draining main venous trunk compatible with DVA.

Fig. 14: SWI show small hipoyntense vascular lesion located in the pons.
Capillary telangiectasia.

Fig. 15: FSPGR T1 with gadolinium show faint enhancement of the small lesion located in the pons.
Capillary telangiectasia.
- Arterial Stroke: The vascular occlusion causes low arterial flow,
and increases deoxygenated blood resulting in deoxyhemoglobin that is paramagnetic.
The draining veins can be seen prominent and hypointense,
some authors propose the identification of a mismatch with diffusion image representing penumbral brain tissue in a different way than current perfusion weighted imaging techniques [4].
In the setting of stroke SWI also can help detecting hemorrhagic infarct or hemorrhagic transformation in the ischemic area.
The acute intra-arterial thrombus is showed as the presence of dark signal from acute deoxyhemoglobin in the vessel,
with diameter larger than the contralateral vessel,
similar to the dense thrombus sign seen on unenhanced CT.
SWI can detect subacute and chronic subarachnoid hemorrhages that sometimes are missed by CT.
- Cerebral venous sinus thrombosis (CVST) and venous infarction: Clinically patients may present with non-specific symptoms and headache due to the increased intracranial pressure.
In an acute CVST we can see prominent hypointense signal with “blooming” artifact within the involved veins.
It is due to the presence of deoxyhemoglobin.
SWI sequence can show also engorgement of the venous system as an early sign of CVST.
Sometimes are demonstrated the frequent hemorrhagic changes associated to venous infarction in the underlying brain parenchyma.
- Neurodegenerative diseases: Iron deposition in the basal ganglia increases with normal aging,
but abnormal iron levels in the CNS structures can be found in a variety of neurodegenerative diseases.
It is encountered in Parkinson disease,
Huntington disease,
Alzheimer,
Multiple Sclerosis (MS),
Amyotrophic lateral sclerosis,
Wilson disease or Pantothenate kinase-associated neurodegeneration (PKAN),
formerly known as Hallervorden-Spatz syndrome [5].
- MS is studied typically with FLAIR and gadolinium enhanced T1.
SWI can improve the sensibility by detecting the perivenular distribution of the demyelinating lesion showing the plaque around the small medullary veins (Fig. 18).
In MS SWI can additionally help to detect iron deposition in demyelinating lesions (Fig. 16),
(Fig. 17).
The level of iron deposition in patients with MS correlates better with disability than the MS plaque volume.
It is also described increased iron content in patients with clinically isolated syndrome (CIS) [6].

Fig. 16: Multiple Sclerosis. SWI. Hypointense iron deposition lesions with two frontal right subcortical lesions, one of them nodular and another one ring shaped.

Fig. 17: Multiple Sclerosis. Phase image. Hypointense iron deposition lesions with two frontal right subcortical lesions, one of them nodular and another one ring shaped.

Fig. 18: Multiple Sclerosis. SWI. Perivenular white matter lesion.
- In Amyotrophic lateral sclerosis (ALS) SWI sequence can detect iron deposition in the cortex,
most notably in the precentral gyrus,
known as the "motor band sign" [7],
(Fig. 19).

Fig. 19: Amyotrophic lateral sclerosis (ALS). SWI. Bilateral iron deposition in the precentral gyrus known as the "motor band sign".
- Brain tumor characterization: Providing identification on hemorrhagic and calcification inside the lesions SWI can help in the grading of brain tumors.
High grade tumors like glioblastomas usually show a very proliferating vascular pattern with hemorrhagic component.
SWI can determine also the calcification in brain tumors like oligodendrogliomas.
Calcification is diamagnetic in opposition to hemorrhage that is paramagnetic,
resulting in different signal intensities on phase images.
Hemorrhage in a tumor can be differentiated from veins if SWI is used both before and after gadolinium,
blood vessels changing their signal intensity but not the regions of inactive hemorrhage.
It serves as an additional tool in grading the tumor by showing the vasculature within and around the tumor.
High rCBV on perfusion weighted imaging has a good correlation with evidence of blood products demonstrated within the tumor using SWI [5,8].
Future directions: SWI is rapidly increasing its clinical use.
Actually the strongest indication are in neurovascular and neurodegenerative disease.
There are several research lines in progress in aging,
atherosclerosis,
MS,
occult vascular disease,
trauma and tumors that will reveal new applications in the near future [4].