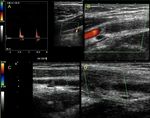
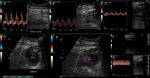
The 90% of the vascular injuries are found in the lower extremities.
Penetrating neck trauma leads to vascular injuries in 25% of casualties.
Arterial,
venous,
neural,
muscular and bony lesions can coexist. They may have an obvious or hidden hemorrhagic site.
1.
Ethiology: The most frequent mechanisms of vascular injury are penetrating wounds (bullets,
knives or foreign bodies), crushing, bone fractures and iatrogenic.
This last cause represents 10% of all the cases in some series7. They are secondary to endovascular procedures such as angioplasty and cardiac catheterisation,
interventionism,
surgeries,
etc.
2.
Clinical presentation: Vascular injuries represent a great challenge to the sonographer because it requires urgent diagnosis to prevent loss of life or limb.
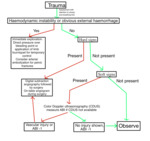
Hard and soft clinical signs are very useful to evaluate the severity of the vascular injury (Fig.1). The bleeding can be obviuos or not.
It is important to define if it is an open or closed wound.
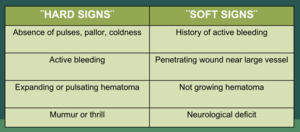
Fig. 1: Clinical signs used for detection of severity of vascular lesions. It can be classified in hard or soft signs.
3.
Diagnostic: Most of the time,
diagnosis is obvious (hard signs of vascular injury).
However,
in doubtful cases,
diagnostic procedures such as hand held Doppler,
Color Doppler,
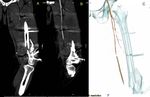
CT-angiography,
and MR-angiography can aid the diagnosis. Angiography is the historical gold standard method.
It localizes the lesion,
defining type and extention.
It can demonstrate active hemorrhage vs occlusion and allows treatment planning.
However,
it has risks for the patient,
as radiation,
renal insult or anaphylaxis.
It can also cause iatrogenic vessel injury.
It is expensive and delays operative intervention.
Angio CT scan is the actual replacing modality for angiography as standard of care.
It has a 95% sensitivity and 87% specificity for the detection of vascular injuries in trauma patients.
It is a fast and effective costwise procedure but requires an hemodynamically stable patient,
cannot exclude all arterial dissections,
and angiography may still be required.
It has limited resolution in presence of foreign bodies and vascular calcifications.
Color Doppler ultrasound (CDUS) is a fast,
inexpensive, innocuous and most of the times,
accurate tool for the diagnosis of the most frequent vascular pathologies.
It needs a trained operator with a slow learning curve and have several limitating factors such as air,
bone fragments,
foreign bodies,
cutaneous wounds,
edema,
large hematomas, casts and bandages. CDUS is presently considered the first-line examination method for the evaluation of vascular injuries,
with 95-97% of sensitivity and 95-98% of accuracy4-7-8-13-14.
4.
Ultrasound technique: In our institution protocols,
the Doppler US exam can only be performed in an hemodynamically stable patient.
During the exploration,
a complete and careful vascular study of the extremity must be done,
following the arterial and venous anatomy in a comparative way with the opposite side.
It is important to seek for the presence of flow and leaks, verifying the indemnity of the arterial wall and the permeability of the vessel.
We have to keep in mind the signs of specific vascular lesions,
such as arteriovenous fistula (AVF) or pseudoaneurysms,
and,
if it is possible,
finding the exact location of the injury.
Some publications have shown some increased peak systolic velocity and end diastolic velocity found at the bleeding site,
as compared to a normal 12.
It is described in the literature that the ankle / brachial index (ABI) is useful in the trauma patient,
to exclude larger vascular injuries (Fig.2).
This is not true for patients with peripheral arterial disease.
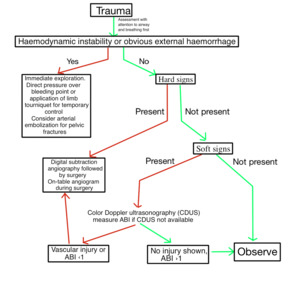
Fig. 2: Action protocol in trauma related vascular injuries.
5.0 Types of vascular injuries: Sometimes,
the sonographer can determine exactly what type of vascular injury the patient has.
Other times,
it is not so easy,
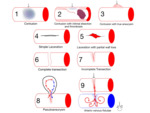
and only a leak or alteration of the flow can be detected and the lesion should be tipified later by another diagnostic method or in the same surgical act. Figure 3 and 4 illustrate the different types of vascular lesion that can be found.
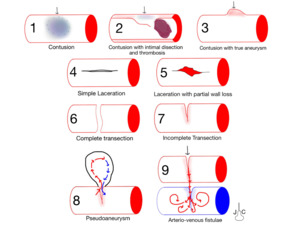
Fig. 3: Types of vascular injuries. 1. Contusion. 2. Contusion with intimal dissection and secondary lumen thrombosis. 3. Contusion with post-traumatic true aneurysm. 4. Simple laceration. 5. Laceration with partial wall loss. 6. Complete transection. 7. Partial transection. 8. False aneurysm or pseudoaneurysm. 9. Arteriovenous fistula.
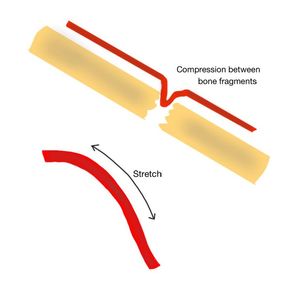
Fig. 4: Types of arterial lesions. Compression between bone fragments and stretching of the artery.
5.A.
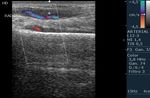
Hematomas: A perivascular hematoma is the most frequent complication at the puncture site.
By ultrasound,
it appears as a complex solid and cystic soft tissue mass adjacent to the injured vessel,
without blood flow on Doppler interrogation.
It is important that if the hematoma has several perisisting liquid anechoic areas,
CDUS must be directed to these sectors,
in order to exclude pseudoaneuryms or pulsatile hematoma (Fig.5).
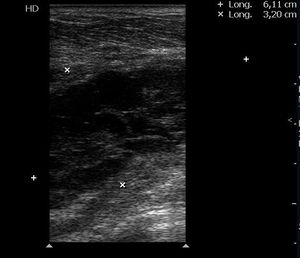
Fig. 5: Hematomas: complex solid and cystic soft tissue mass adjacent to the injured vessel, without blood flow on Doppler interrogation. It is important that if the hematoma has several perisisting liquid anechoic areas, CDUS must be directed to these sectors, in order to exclude pseudoaneuryms or pulsatile hematoma.
5.B.
Contusions: A high impact hit on the artery can cause a contusion of the arterial wall,
damaging the structure of it,
affecting or not the wall indemnity.
It can cause dissection by detachment of the intima and associated thrombosis of the lumen. It can also cause weakness of the arterial wall with subsequent dilatation as focal true aneurysms.
5.C.
Lacerations: The lacerations can be a simple cut in the arterial wall or it can be accompanied by a partial loss of the wall.
From the fissure,
bleeding occurs,
forming periarterial haematomas,
pseudoaneurysms and AVF.
Fig. 6: Types of complications associated with transections
5.D.
Transections: The transections can be partial or complete (Fig.6).
From the fissure,
immediate or delayed hemorrhage can occur (Fig.7) and hematomas,
pseudoaneurysms and AVF can also be formed.
The lumen can also be compromised by thrombus or embolism. Complete transections are responsible for the interruption of flow to the distal areas,
leading to acute ischemia.
Also the active bleeding of a transected vessel stimulates the vasospasm of it provoking also distal ischemia (Fig.8-9).

Fig. 7: Incomplete or complete transections.
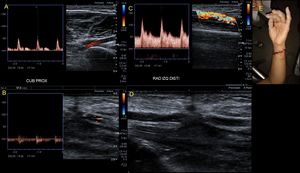
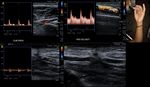
Fig. 8: 21-year-old woman who was wounded with a glass in the medial area of the wrist. The proximal ulnar artery, near the elbow, has low velocity flows. Distally, the velocities continue to decrease until a partial transection of the artery is identified, with critical distal flows and signs of vasospasm. The radial artery presents compensatory hyperflow. The case continues in Figure 9.
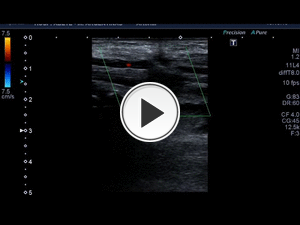
Fig. 9: This figure is a continuation of the case in Figure 8. A 21-year-old woman who was wounded with a glass in the medial area of the wrist. The proximal ulnar artery, near the elbow, has low velocity flows. Distally, the rates continue to decrease until a partial transection of the artery is identified, with critical distal flows and signs of vasospasm, seen in this video. The radial artery presents compensatory hyperflow.
5.E.
Arterial Thrombosis: Arterial thrombosis is a frequent complication.
Varying degrees of thrombus echogenicity may be detected depending on the thrombus age (Fig.10-11).
A partially occluding thrombus causes alteration in the color flow pattern,
waveform,
and velocities.
A totally occluding thrombus causes an abrupt cutoff of color flow and retrograde flow in a collateral pathway3.
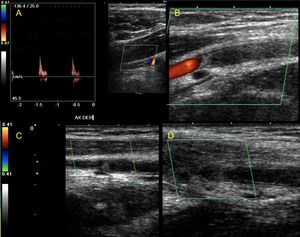
Fig. 10: A 37-year-old male patient with a bullet wound in the axillary region. A complete transection of the axillary artery is observed, with thrombosis of the proximal and distal ends of the same. It presents spontaneous collateral circulation that recanalizes at the humeral level.
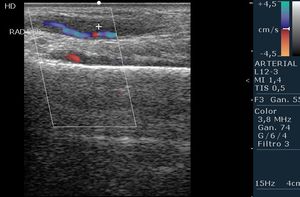
Fig. 11: Radial Artery post cardiac catheters. The artery has signs of focal post-traumatic arteritis, with asymmetric thickening of the arterial wall and partial thrombosis of the lumen.
5.F.
Pseudoaneurysms or false aneurysms:They happen when there is a total disruption in the arterial wall,
the blood can leak from the lumen,
and be contained by the external surrounding soft tissues and compressed thrombus.
By this way,
a pulsatile hematoma is formed,
that fills in systole through a channel (neck) that communicates with the injured artery, and empties in diastole.
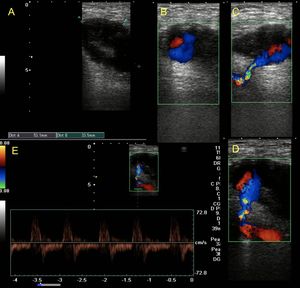
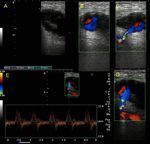
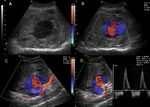
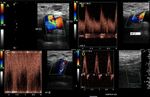
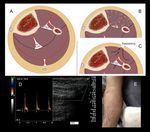
Fig. 12: Post-traumatic femoral pseudo-aneurysm. It presents as and heterogeneous image, partially liquid, with the presence of flow with color Doppler (sign of yin-yang) in those areas. You can also see the thin neck that connects with the arterial lumen. The flow at the level of the neck is bidirectional ¨to and fro¨.
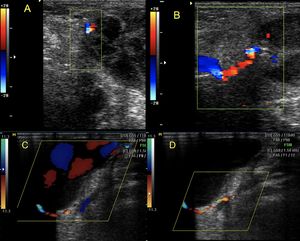
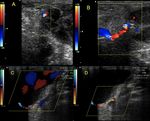
Fig. 13: A and B: partially thrombosed pseudoaneurysm. Some peripheral liquid areas persist, which present flow with color Doppler.
C and D: Pseudoaneurysm. History of a puncture wound with a branch at the right infrapatellar level. It can be identified the long and thin neck that connects with the artery that originates the seudoaneurysm
They may involve surgical or puncture sites such as femoral or radial arteries,
but they can be present in any trauma related vascular injury.
Clinically,
a pulsatile mass with a palpable thrill and murmur is detected.
Ultrasound reveals anechoic or hypoechoic areas resembling fluid collections located adjacent to the arterial wall.
CDUS can delineate the size and limits of the cavity,
the degree of clotting,
the neck localization,
and the blood flow pattern. The lumen of the pseudoaneurysm has swirling turbulent flow,
that is seeing in CDUS as a “yin-yang” sign (Fig.12-13-14-15-16-17-18).
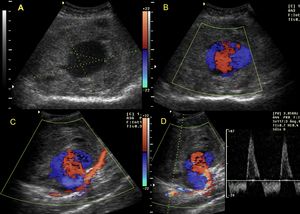
Fig. 14: A 32-year-old female patient that went through a glass door, breaking it and sticking a splinter in the distal thigh at the level of Hunter's canal. Post-traumatic pseudoaneurysm is observed (A) with the sign of yin-yang (B) and the neck that communicates with the femoral artery (C) with bidirectional flows (D).
The neck typically has a bidirectional ¨to-and-fro¨ waveform due to flow entering during systole and exiting during diastole. Smaller diameter and long necks are more suitable for percutaneous interventions than pseudoaneurysms with short (less than 1 cm) and wide necks (larger than 2–3 cm),
and location must be considered19.
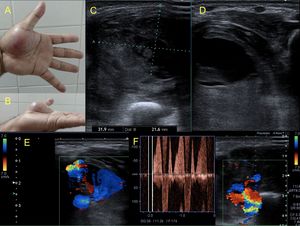
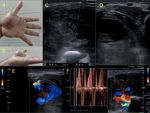
Fig. 15: Male patient of 43 years, cameraman, who reports having fallen with the camera in his hand, receiving a heavy blow. The hand persists swollen in the thenar eminence and with pain.
A heterogeneous image is observed, with hypoechoic and anechoic areas, liquid zones. It presents evidence of arterial flow within the liquid zones, confirming the diagnosis of pseudoaneurysm. It is possible to identify the neck with bidirectional flows, as well as the artery from which the leak comes, behind the pseudoaneurysm. This case continues with the video in figure 16.
Fig. 16: This video is a continuation from figure 15. Male patient of 43 years, cameraman, who reports having fallen with the camera in his hand, receiving a heavy blow. The hand persists swollen in the thenar eminence and with pain.
A heterogeneous image is observed, with hypoechoic and anechoic areas, liquid zones. It presents evidence of arterial flow within the liquid zones, confirming the diagnosis of pseudoaneurysm. It is possible to identify the neck with bidirectional flows, as well as the artery from which the leak comes, behind the pseudoaneurysm.
The natural history is varied.
Most pseudoaneurysms spontaneously thrombose.
The dreaded complication of a pseudoaneurysm is continued expansion and bleeding into the thigh or retrograde bleeding into the pelvis.
Expanding pseudoaneurysms can cause limb ischemia through compression19.
Sonographically guided thrombin injection is the treatment of choice for large pseudoaneurysms that do not clot spontaneously,
with a 93% to 100% success rate.
Hypoechoic hypervascular lymph nodes,
dilated varicose veins,
and fluid containing femoral or inguinal hernias,
may mimic pseudoaneurysms.
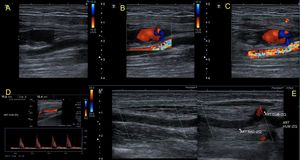
Fig. 17: Patient with history of cardiac catheterization 1 month ago, which enter through the humeral artery. It presents a pseudoaneurysm of the artery with the ¨yin yang¨ sign and a short and wide neck difficult to compress. It also evidences a thrombosis of the radial artery with compensatory hyperflow in the ulnar artery. This case continues in figure 18.
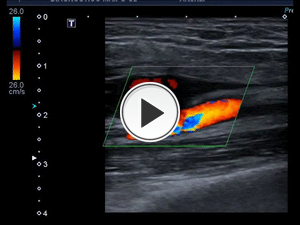
Fig. 18: This video is a continuation from the same case of figure 17. Patient with history of cardiac catheterization 1 month ago, which enter through the humeral artery. It presents a pseudoaneurysm of the artery with the ¨yin yang¨ sign and a short and wide neck difficult to compress.
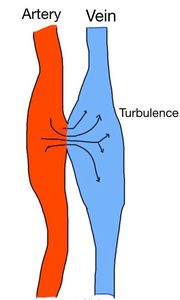
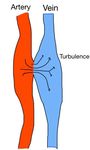
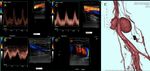
Fig. 19: Arterio-venous fistulae. The artery is a high pressure compartment, from which the blood is expelled to the vein, causing turbulence and bruits. The vein becomes pulsatile.
5.G.
Arteriovenous fistula: It is a false vascular channel between an artery and the adjacent vein (Fig.19).
A palpable thrill and a bruit on auscultation are often present. Color Doppler duplex sonography shows a mosaic color pattern or ¨aliasing¨ due to high turbulent flow in the fistula.
Spectral findings are low-resistance arterial flow with increased diastolic flow in the feeding artery,
and a high-velocity,
chaotic waveform in the draining vein.
Extravascular color signals represent perivascular tissue vibration due to transmitted pulsation of turbulent continuous blood flow between the artery and the vein (Fig.20-21-22-23-24).
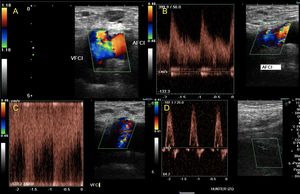
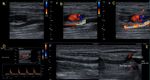
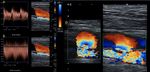
Fig. 20: Patient with a history of gunshot wound 5 years ago in the inguinal region. He presents intermittent claudication of the left side. They request an arterial Doppler of lower limbs in which it is possible to observe: arterial flows of low resistance and high velocities in the common Femoral artery (B) that are not observed at the level of the Hunter channel (D). It is also possible to check pulsatility in the femoral venous flow (C). These findings suggest the presence of an arteriovenous fistula which is located between the common Femoral artery and its vein, which present a lateral communication (A).
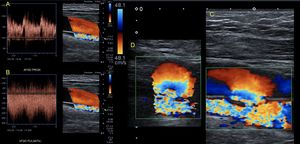
Fig. 21: Patient with gunshot wound on right thigh, middle third 1 year ago. It presents pain, claudication and thrill. Femoral artery is observed with decreased flow resistance and increased velocities (A), Femoral vein with turbulent and pulsatile flow (B) and arteriovenous fistula between the artery and vein (C and D). This case continues in figure 22 (video)
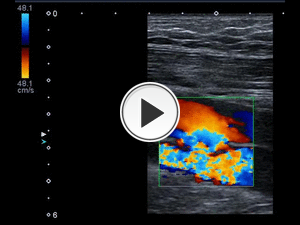
Fig. 22: Patient with gunshot wound on right thigh, middle third 1 year ago. It presents pain, claudication and thrill. Arteriovenous fistula between the artery and vein. Color mosaic or aliasing and turbulence can be seen. There are very high velocities In the fistulas.
Arteriovenous fistulas are often asymptomatic, but when significant can contribute to high-output cardiac failure. They can also cause symptoms of distal ischemia.
Small arteriovenous fistulas can spontaneously remit. Percutaneous placement of a covered stent or surgical repair is often indicated19.
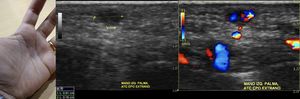
Fig. 23: Patient who stuck his hand with a rose thorn, 1 month ago. Trying to remove the thorn, he autoexplored it with a needle several times.
A blue spot persists in the palm of the hand, for which an ultrasonographic study is requested. A small liquid area is observed, which shows flow with color Doppler which comes from the depth. It presents aliasing and high velocity flows compatible with arteriovenous fistula that is confirmed with angioResonance
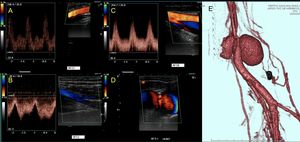
Fig. 24: Patient with history of gunshot wound three months ago. After that, pain and swelling in the knee and calf persist. It is observed Femoral arterial flow of low resistance and high velocities and in the vein pulsatile flow.
At the level of Hunter's canal, two pulsatile masses are found at periarterial level, with turbulent flow and venous pulsatility. An angioCT scan shows a complex vascular lesion, composed of two pseudoaneurysms lateral to the femoral artery and a concomitant arteriovenous fistula.
5.H.
Arterial compression between bone fragments: In some cases,
the artery may become trapped between bone fragments of a fracture.
This can compromise the distal arterial flow and cause ischemia (Fig.25-26).
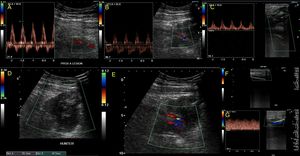
Fig. 25: Comminuted fracture of the distal third of Femur, which generates a large hematoma at that level (D). The osseous fragments have lacerated the artery, which shows flows (A and B) up to that level (E). Distally, the patient presents scarce collateral circulation with postobstructive flows in popliteal artery and posterior tibial artery(C and G). No flow in the anterior tibial is detected (F). Distal vasospasm is not ruled out by arterial transection or extrinsic compression by the hematoma. AngioCT scan is performed demonstrating the interruption of the flow at the level of the lesion (the case continues in Figure 26).
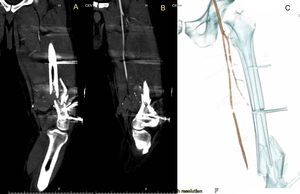
Fig. 26: This case continues from imagen 25. Comminuted fracture of the distal third of Femur, which generates a large hematoma at that level. The osseous fragments have lacerated the artery, which shows flows up to that level. Distal vasospasm is not ruled out by arterial transection or extrinsic compression by the hematoma. AngioCT scan is performed demonstrating the interruption of the flow at the level of the lesion.
5.I.
Stretching: can produce that the innermost layer of the artery may tear,
causing dissection,
embolism,
thrombosis and occlusion of the artery. This can compromise the irrigation of the distal arterial branches.
5.J.
Compartment syndrome: It is an increase in pressure against the fixed fascial or skeletal boundaries that compromises perfusions to the tissue within that compartment. It can be caused by a variety of injuries: crush injuries,
large soft tissue injury with bony fractures, ischemia–reperfusion,
and hemorrhage into a compartment among other less common causes. The diagnosis relies on a high index of suspicion and vigilant surveillance postinjury16 (Fig.27).
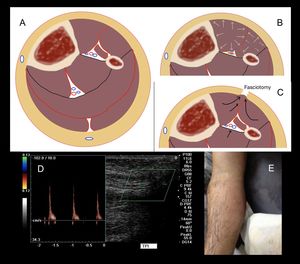
Fig. 27: The compartment syndrome is the increase in pressure within an aponeurotic compartment, which produces muscle ischemia. First the veins collapse, aggravating the picture with edema, and then the arterial flow is compromised giving ischemia and necrosis (B). The fasciotomy decompresses the compartment, lowers the pressure and allows a new flow in the artery and vein, preventing ischemia (C). With color Doppler it is very difficult to make the diagnosis. There are no veins.
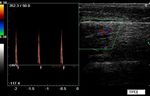
Arterial flows can be of high resistance showing spectral patterns called needle or spike (D).
Regarding the ultrasonographic diagnosis,
no venous flow can be found within the compartment because of the high pressure that colapses the veins.The espectral Doppler evaluation of the compartment arteries can show high resistence waveforms,
call as ¨spike¨ or ¨needle¨ waveforms (Fig.28).
But they are not always present.
In our experience,
CDUS is not a good method for confirmation of compartment syndrome.
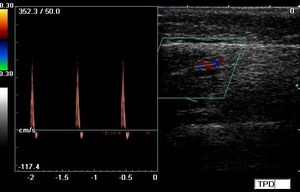
Fig. 28: Arterial flows spectral patterns called needle or spike, that can be seen in compartmental syndrome
5.K.
Vein lacerations: Isolated venous injuries (Fig.29) are less common than combined arterial and venous injuries. Colour flow duplex is a valuable tool with the ability to identify not only occult arterial injuries,
but also venous injuries15.
The diagnosis is not easy.
Venous injuries may require 12 to 24 hours to become symptomatic,
with swelling, edema,
or cyanosis.
In cases of proximal venous injury, swelling may be massive with limb threatening and phlegmasia cerulea dolens. Recognition of venous injuries in patients with non-life or limb threatening trauma can be more challenging18.
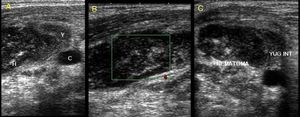
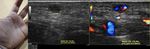
Fig. 29: Parajugular hematoma after attempt to place a central line. Heterogeneous image, hypo-echoic, that collapses the Yugular vein which is not seen.
5.L.
Deep vein thrombosis (DVT): The incidence of acute DVT secondary to penetrating trauma to the thigh in proximity to a major vascular structure,
can occur in 4 to 16%.
The loss of spontaneous venous flow,
respiratory variation,
and compressibility confirm the diagnosis18(Fig.30).
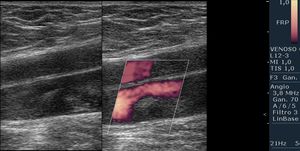
Fig. 30: Post-traumatic deep vein thrombosis. It is observed inside the vein, thrombotic material inside the lumen of acute type, central, not adhered.