Considering the evaluation of neuroblastomas and Wilms tumors,
in some atypical cases it may be a great challenge to distinguish them by CT evaluation,
giving the various possibilities of evolution.
Suggestive examples are the extension of the neuroblastoma into the renal hilum and invasion of the kidney parenchyma,
or atypical appearance of Wilms tumor with calcifications,
exophytic growth,
crossing the midline,
or large lymph node metastases.
Nevertheless,
another issue that may cause diagnosis difficulties is the close contact of the tumoral mass with adjacent structure,
the undetermined interface making hard to differentiate from true invasion.
The CT findings of 15 children with abdominal neuroblastoma and 10 with Wilms tumor,
diagnosed in the Emergency Clinical Hospital for Children from Iasi,
Romania,
between July 2012 and March 2018,
were retrospectively reviewed and compared.
The imaging evaluation was part of the preoperative or pretreatment workup,
and the diagnosis was histologically proven.
The scans were performed on a Siemens Somatom Emotion 16 Slice CT scanner,
both before and after intravenous contrast medium administration.
The characteristics that have been followed were the site of the tumor mass,
tumor composition and margins,
presence of calcifications,
extension of the tumor and growth pattern,
with great regard on the midline crossing,
the displacement of the great vessels and the distortion of the kidney,
and not eventually,
the metastatic sites.
Results for nephroblastoma
The ages of patients with Wilms tumors (6 girls and 4 boys) ranged from 7 months to 14 years,
with an average of approximately 3.5 years.
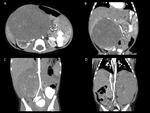
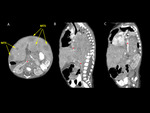
In 7 of 10 cases,
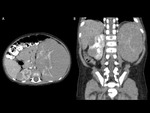
the tumor arose from the right kidney (one of which was a horseshoe kidney – fig.
2),
two cases from the left kidney,
and one having multiple tumors in both kidneys,
with larger diameters on the left side (fig.
3).
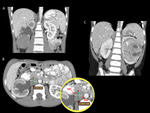
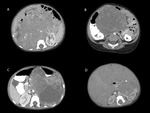
The renal mass had soft tissue densities,
only one case with hypodense areas due to necrosis and/or hemorrhage,
with moderate inhomogeneous (8 of 10 cases) or homogenous (2 of 10 cases) enhancement,
rounded,
regular shape and regular margins (6 of 10 cases),
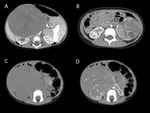
or with no evident interface with adjacent structures (4 of 10 cases),
such as right hepatic lobe,
cephalic pancreas,
psoas and quadratus lumborum muscles (fig.
4,
5).
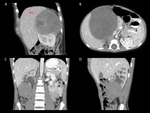
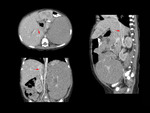
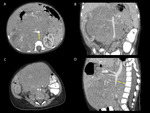
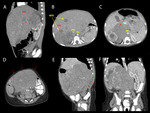
In 2 of 10 cases the tumor included fine calcifications (fig.
6 B) and one case presented evident neoformation vessels (fig.
4 C,D).
In 3 of 10 cases the tumor crossed the midline, displacing the bowels,
with slightly displacement of the aorta,
or the superior mesenteric artery,
but with no compression or encasement of the vessels (fig.
7).
In one case the inferior vena cava (IVC) was displaced to left and comprised,
and one case included IVC thrombosis (fig.
8).
The renal mass caused the distortion of the collecting system (8 of 10 cases),
in some cases occupying quasi-completely the kidney (2 of these 8),
leaving only a fine edge of parenchyma.
One case involved an adenopatic block that encased the renal hilum,
causing hydronephrosis and delayed secretion and excretion (fig.
9).
4 of 10 cases involved metastatic sites,
represented by pulmonary nodules (fig.
10).
Results for neuroblastoma
Considering the patients diagnosed with neuroblastoma (7 girls and 8 boys),
the age ranged from 9 days to 4.5 years,
with an average age of approximately 2 years.
The tumoral mass was located at retroperitoneal level,
on the topography of the left adrenal gland in 8 cases,
while 6 cases arose on the topography of the right adrenal gland.
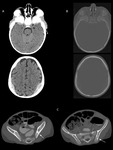
One case involved both the retroperitoneum and posterior mediastinum,
suggestively along the sympathetic chain,
with thoracic extension,
which was eventually diagnosed as ganglioneuroblastoma (fig.
11).
The tumor appeared as a solid mass,
with heterogeneous structure,
in 6 cases having mixed densities,
with areas of necrosis or hemorrhage,
cystic areas or fatty-densities,
the majority (9 out of 15) including calcifications,
either coarse or fine.
Commonly with irregular shape and margins (11 of 15 cases),
some of the tumors had round-oval shape,
well circumscribed,
with polycyclic outline (4 out of 15).
Following intravenous contrast medium administration,
all tumors had either discrete,
moderate,
or intense,
heterogeneous or even homogenous enhancement.
The tumors with mixed structure showed important enhancement in the solid component (fig.
12).
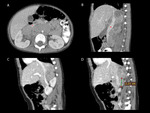
In 7 of 15 cases the tumor crossed the midline,
with mass effect on major vessels and their branches (fig.
13,
16B).
Three of these involved the encasement of the aorta,
without compression,
associated with anterior displacement away from the vertebral bodies and encasement of both the renal arteries (one case),
or with extension on left common iliac artery (one case),
one of the cases also involving the encasement and compression on the IVC.
The effect on the aorta was not correlated with the site of the origin,
giving the extension beyond the midline.
Nonetheless,
the tumor occurring on the right adrenal gland manifested an evident encasement and compression of the IVC,
regardless the midline crossover.
The ipsilateral kidney was only displaced in 8 cases,
one of which involved the renal hilum,
causing ureteral encasement and consequently,
hydronephrosis,
and also renal artery thrombosis; another one revealed an adenopathic block which encased the renal hilum,
but without significant consequences.
One case revealed renal atrophy with architectural changes,
possibly due to hilum infiltration and renal vessels thrombosis.
In other 4 cases,
the tumor grew toward the kidney and showed no demarcation between the renal parenchyma and the retroperitoneal mass,
2 of them associating hilum infiltration (fig.
14).
Metastatic sites were present in 9 cases,
5 of them located in bones: scapula,
sacrum,
iliac bone,
vertebral bodies or skull,
with extradural infiltrates (fig.
15),
5 cases with hepatic disseminations (fig.
16B),
1 case with pulmonary nodules,
pleural effusion and diffuse consolidation.
Three cases showed retroperitoneal lymphadenopathy: one with contiguous extension of the primary tumor in the retroperitoneal space,
one with lymphadenopathy that encased celiac trunk and superior mesenteric pack and one with retrocaval adenopathy (fig.
17).
Discussion
Although both neuroblastoma and nephroblastoma occurred in early childhood,
nephroblastoma seem to have a peak incidence at a slightly older age.
Nephroblastomas usually have round well-circumscribed shape and smooth margins,
while neuroblastomas tend to appear lobulated,
with irregular shape,
poorly marginated with an invasive pattern (fig.
4,
12,
16).
Nonetheless,
none of these features are valuable in distinguishing these two conditions,
given the anteriorly illustrated possibilities that nephroblastoma may cross the midline or invade the adjacent structures,
or that neuroblastoma may be round,
with regular margins.
The internal structure and enhancement after contrast administration are non-specific findings,
both of the tumoral masses having inhomogeneous pattern.
Even though cystic,
hemorrhagic or necrotic areas are more common for neuroblastoma,
it is a feature with little help in differentiation,
with no proven specificity (fig.
4,
12).
However,
a distinguishing feature of neuroblastoma cases is the presence of calcifications,
while for nephroblastoma is less common.
Most of the tumoral masses in neuroblastoma presented multifocal calcifications,
rather coarse or pinpoint,
but numerous,
while nephroblastoma showed minimally,
fine calcifications (fig.
6).
A characteristic of neuroblastoma is the tendency to cross the midline,
with tumoral encasement and possible displacement of large vessels.
In accordance with previous studies,
this aspect,
with the elevation of the aorta away from the vertebral column,
is a reliable sign of differentiation from nephroblastoma,
and appeared in 3 of the 15 cases included in the present study (fig.
13).
The nephroblastoma cases that included the crossing of the midline did not associate encasement or compression of aorta,
even though a slightly lateral deviation was registered,
but no anterior displacement.
However,
nephroblastoma can invade and extend through the renal vein and IVC,
and a tumoral thrombus in these vessels is highly predictive,
although not diagnostic (fig.
7,
8).
In nephroblastoma,
tumors obviously occur within the kidney,
usually with calyceal distortion,
whereas neuroblastoma have extrarenal origin,
related to the kidney,
but causing renal displacement or possible hilum infiltration.
A challenging situation is renal invasion found in neuroblastoma,
as shown in 4 of the cases,
with no demarcation with the kidney parenchyma.
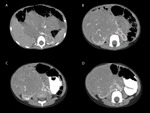
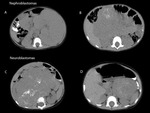
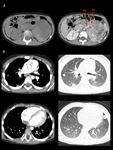
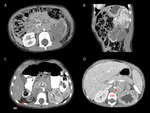
A very useful sign that can indicate the origin of the mass was the “claw sign”,
referring to the sharp angles with the surrounding normal parenchyma,
on either side of the tumor (fig.
1).
This sign is useful to determine if a mass emerges from a solid structure rather than being located adjacent to it and distorting the outlines and is a valuable tool in the diagnosis of nephroblastomas.
Another distinctive criterion is the pattern of metastases: nephroblastoma predominantly involved pulmonary sites (fig.
10),
while neuroblastoma,
more frequently metastatic when diagnosed,
evenly presented hepatic and bone metastasis (fig.
15),
followed by lymph node enlargement (fig.
17),
and only one pulmonary metastatic site,
the data being in accord with the previous studies from the literature.