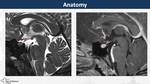
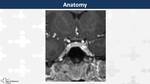
Sellar region is a complex anatomical space with several structures from different embryological origin (Fig. 1, Fig. 2)
Normal size and shape of pituitary gland depends on the age.
It is 6 mm with upper surface flat or slightly concave in children under 12 years-old and 10 mm with upper surface convex in puberty.
It should be homogeneously isointense on T1WI with a posterior “bright spot” representing neurohypophysis.
Pituitary stalk and neurohypophysis enhance before (20 seconds) than adenohypophysis (80 seconds)
Pathologies can be classified according to their location.
Additional radiological (calcification,
cystic content,
enhancement pattern) and clinical features or age narrow the diagnosis (Table 1, Table 2)
Anatomical variants:
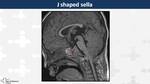
J-shaped sella (Fig. 3)
Variant morphology of the sella turcica present in 5% of children,
due to a flattened tuberculum sellae that form the straight edge of the "J".
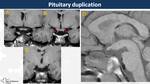
Pituitary Duplication (Fig. 4)
Rare condition where two pituitary glands and osseous sella are normal in morphology but laterally located,
associated with two pituitary stalks,
a thick tuber cinereum on midline and,
sometimes,
a basisphenoid cleft or frontonasal dysplasia.
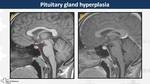
Pituitary Gland Hyperplasia (Fig. 5)
Physiologic condition present in neonates,
young menstruating females or pregnant/lactating women.
It may also be found in patients with end-organ failure (e.g.
Addison disease)
On MRI,
pituitary gland enlarge diffusely between 10-15 mm with convex superior margin and it shows homogeneous contrast-enhancement,
although it may also be nodular mimicking an adenoma.
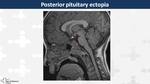
Posterior pituitary ectopia (Fig. 6)
Normal variant incidentally found in 1:4,000-20,000 live births.
On MRI,
it presents as a nodule between 3-8 mm,
hyperintense on T1WI,
located at the floor of the third ventricle.
It is associated with small adenohypophysis and sella turcica and even persistent sphenopharyngeal foramen.
In symptomatic cases,
other malformations may be present,
such as heterotopia,
optic nerve hypoplasia or callosal dysgenesis.
Posterior pituitary absence
Normal variant incidentally found in 10% of neonates,
associated with normal infundibulum and adenohypophysis.
Sellar conditions:
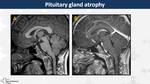
Pituitary atrophy (Fig. 7)
In children,
radiotherapy is the main cause of pituitary atrophy.
Atrophy evolves with time due to impaired secretion of hypothalamic trophic factors and/or direct radiation-induced damage.
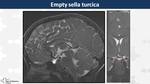
Empty sella turcica (Fig. 8)
It may be idiopathic or secondary to prior tumours,
radiotherapy,
surgery or haemorrhage.
On MRI,
there is a enlarged sella turcica filled with CSF.
The infundibulum passing through the space excludes a cystic mass.
In patients with benign intracranial hypertension,
it is associated with flattened ocular globes and nerve sheath prominence.
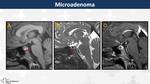
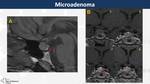
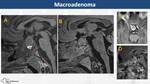
Adenoma (Fig. 9, Fig. 10, Fig. 11)
Benign intrasellar tumour (WHO I) detected in middle-aged patients but also in adolescents.
Microadenomas (<10 mm) are frequently asymptomatic,
except for microprolactinomas,
whereas macroadenomas (>10 mm) present with symptoms secondary to mass effect (hydrocephalus,
cranial pairs palsies and visual disturbances) or to hormonal deficiencies.
On MRI,
they are well-circumscribed masses surrounded by crescentic rim of compressed adenohypophysis associated with suprasellar extension,
especially in macroadenomas (snowman sign).
Neurohypophysis "bright spot" is displaced above diaphragma sellae (80%) or absent (20%)
They are isointense on T1WI and hypointense on T2WI.
Cystic and necrotic degeneration may be present,
especially in macroadenomas.
Microadenomas show delayed and homogeneous contrast-enhancement while macroadenomas,
avid and heterogeneous contrast-enhancement.
Pituitary carcinoma is extremely rare,
similar of macroadenomas,
except for the metastases.
Pituitary Apoplexy is an acute clinical syndrome caused by hemorrhagic necrosis of a macroadenoma (60-90%) but also of a normal adenohypophysis in patients with prior radiotherapy or surgery or Sheehan Syndrome.
On MRI,
it presents hyperintensity on T1WI due to haemorrhage (85%) and restricted diffusion in infarcted areas.
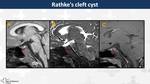
Rathke's cleft cyst (Fig. 12)
They are well-circumscribed midline cyst between 5-15 mm,
with variable T1WI signal depending of its proteinaceous content,
hyperintense on T2WI without contrast-enhancement.
A millimetric non-enhancing intracystic nodule (75%) and small curvilinear calcifications (10-15%) may be present.
There is suprasellar extension over the posterior lobe in 60% of cases (posterior ledge sign)
Craniopharyngioma should be considered if recurrence or progression,
since they share a similar embryological origin.
Pituicytoma
Benign glial tumour (WHO I) incidentally found in middle-aged men,
although cases have been reported in children.
They are well-circumscribed tumours located in the posterior pituitary or the infundibulum,
isointense on T1WI and heterogeneously hypointense on T2WI,
with avid and homogeneous contrast-enhancement.
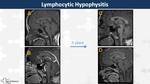
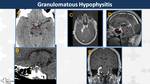
Hypophysitis (Fig. 13, Fig. 14)
Uncommon inflammatory condition of the adenohypophysis in children.
Lymphocytic hypophysitis is a self-limiting autoimmune condition present in pregnant women during third trimester and puerperium and granulomatous hypophysitis is a chronic inflammation in young women with systemic sarcoidosis.
They present an enlarged adenohypophysis hypointense on T2WI with heterogeneous contrast-enhancement and without the “bright spot” of neurohypophysis.
Thickened infundibulum (>2 mm without normal “top to bottom” tapering),
dural thickening and sphenoid sinus mucosal thickening may be present.
Suprasellar conditions:
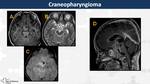
Craniopharyngiomas (Fig. 15)
Most common pediatric intracranial tumor of non-glial origin (WHO I) (1-5%).
Two main subtypes: Adamantinomatous in adolescents and papillary in adults.
Adamantinomatous subtype is a suprasellar tumour with intrasellar extension (75%) and multiple cysts with variable signal intensity on T1WI depending on content (protein,
blood products and cholesterol) Calcifications are present in 90% of cases.
Unlike papillary subtype,
solid portion of the tumor is minimal and enhances avidly,
although wet keratin nodules may be present.
Hyperintensity on T2WI of brain parenchyma may be present secondary to gliosis,
tumor infiltration or irritation from leaking cyst fluid.
Complete resection is difficult in adamantinomatous subtype and recurrence is common (33%)
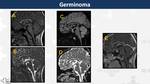
Germinoma (Fig. 16)
Benign tumour (WHO I) located in the pineal region (50-65%) but also in the suprasellar region (6-13%) Patients usually present with diabetes insipidus.
They are lobulated masses slightly hyperintense both on T1WI (fat content) and T2WI,
located at the floor and lateral walls of the third ventricular and anterior columns of fornices.
Cystic and necrotic degeneration and central calcification may be present.
They show restriction on diffusion and avid contrast-enhancement.
Leptomeningeal seeding of the lesion is possible,
so imaging of the entire neuraxis is necessary.
Germinal tumours
Spectrum of lesions: epidermoid cysts (ectoderm) in middle-age men,
dermoid cysts (ectoderm with skin appendages) in women under 30 years-old and mature teratomas (three layers with additional adipose tissue) in childhood.
Epidermoid and dermoid cysts are lobulated lesions that fill and expand CSF spaces and encase nerves and vessels,
with similar signal intensity than CSF.
Restriction on diffusion and presence calcifications (10-25%) are characteristic of epidermoid cysts,
while hyperintense droplets in the subarachnoid space are typical of ruptured dermoid cysts.
Diffuse pial enhancement secondary to recurrent aseptic meningitis has been described in both type of cysts.
Teratomas present as solid-cystic lesions that appear hyperintense on T1WI,
with contrast-enhancement of the nodular portions and coarse calcification.
Lipoma
Tuber cinereum is the third most common location (0.4%) and they are usually asymptomatic.
On MRI,
they appear as solid lesions that follow fat signal on all sequences,
without calcification or contrast-enhancement nodules.
Differential diagnosis with posterior pituitary ectopia should be considered.
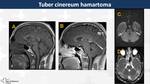
Hamartoma (Fig. 17)
Benign heterotopia presented in children with seizures and progressive cognitive and functional disability.
On MRI,
they appear as sessile or pedunculated tumours arising from the tuber cinereum,
isointense on T1WI and slightly hyperintense on T2WI,
without contrast-enhancement.
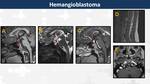
Hemangioblastoma (Fig. 18)
Suprasellar location is extremely rare for hemangioblastomas,
with only sixteen cases described in literature,
all of them in patients with von Hippel-Lindau disease.
On MRI,
they appear as a thickened infundibulum with avid and homogeneous contrast-enhancement.
There is no cystic degeneration unlike those in the cerebellum.
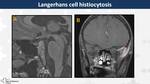
Langerhans cell histiocytosis (Fig. 19)
It is a multisystemic granulomatosis presented in children with pituitary gland involvement in 6% of cases.
On MRI,
they appear as thickened and enhanced infundibulum with absence of the “bright spot” of the neurohypophysis.
There are also symmetric hyperintensities on both T1WI and T2WI at dentate nuclei of cerebellum and basal ganglia and masses arising from the meninges,
pineal gland and choroid plexus.
It is usually associated with bilateral symmetric neurodegeneration of the cerebellum (especially the dentate nuclei) and basal ganglia and mass lesions arising from the meninges,
pineal gland,
choroid plexus,
hypothalamus,
or ependyma
It is also associated with multiple skull lytic lesions without sclerotic rim and with asymmetrical involvement of the inner and outer tables.
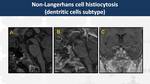
Non-Langerhans cell histiocytosis (Fig. 20)
Group of non-Langerhans cell multisystemic granulomatosis which include Erdheim-Chester disease,
Rosai-Dorfman disease,
juvenile xanthogranuloma and dendritic cell histiocytosis.
They usually present in middle-age men with diabetes insipidus,
although cases have been described in children.
On MRI,
they also appear as thickened and enhanced infundibulum with additional extension to the optic nerve and the hypothalamus.
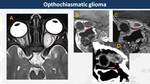
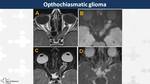
Hypothalamic-optochiasmatic glioma
They are pilocytic astrocytomas (WHO I) that account for 10-15% of supratentorial tumours in children,
especially those with type 1 neurofibromatosis (30-40%)
On MRI,
optochiasmatic gliomas appear as fusiform and tortuous enlargement of the optic nerves,
hyperintense on T2WI with an hypointense halo of arachnoidal gliomatosis.
Contrast-enhancement is variable,
usually more avid in sporadic cases (Fig. 21) than in syndromic ones (Fig. 22).
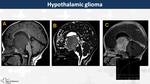
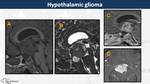
Hypothalamic gliomas usually appear as a voluminose suprasellar mass hyperintense on T2WI,
with micro and macrocysts and avid contrast-enhancement (Fig. 23, Fig. 24)
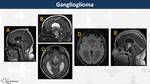
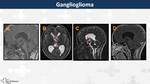
Ganglioglioma (Fig. 25, Fig. 26)
They are rare tumours (95% WHO I,
5% anaplastic WHO III) presented in children and young adults.
On MRI,
they are found more frequently in temporal lobes (70%) than suprasellar region and they appear as cystic masses with mural contrast-enhanced nodule.
Calcification may be present (35%)
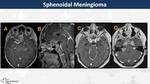
Meningioma (Fig. 27, Fig. 28)
Most common tumour of the meninges (70% WHO I,
30% WHO II,
1% WHO III),
presented in patients over 40 years-old,
but also in adolescents with type 2 neurofibromatosis.
On CT,
they appear as well-circumscribed and broad dural-based masses with dural tail (60-72%) and CSF vascular cleft.
Vessels adopt an spoke-wheel pattern inside the tumour.
There are calcifications (20-30%) and hyperostosis (5%) Enlargement of paranasal sinuses (pneumosinus dilatans) may be present.
Characteristics on MRI are similar to CT.
Fibrous and psammomatous variants usually are hypointense on T1WI and T2WI,
whereas microcystic,
secretory,
cartilaginous,
chordoid and angiomatous variants are hyperintense on T2WI.
All of them usually present avid contrast-enhancement.
Restriction on diffusion and volume transfer constant (K-trans) on perfusion show a good correlation with histological grade.
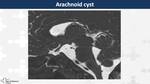
Arachnoid cyst (Fig. 29)
Suprasellar is a rare location of arachnoid cysts and,
unlike the rest of locations,
they usually are symptomatic because of the compression of optic chiasm and infundibulum.
On MRI,
they appear as thin-walled cysts relatively less intense than the surrounding CSF,
without restriction on diffusion neither contrast-enhancement.
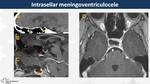
Third ventricle ventriculocele (Fig. 30)
Cephaloceles are less common than mieloceles and they sometimes are associated with ventriculoceles in which ventricles protrude into a hernia sac in association with herniating choroid plexus.
They usually involve the IV ventricle in posterior fossa but III ventricle ventriculocele has not been described in literature.
Cavernous sinus conditions:
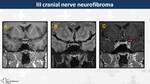
Nerve sheath tumors (schwannomas and neurofibromas) (Fig. 31)
They are presented in middle-aged women,
although multiple schwannomas have been described in children with type 2 neurofibromatosis.
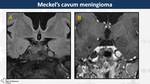
Trigeminal schwannoma is the second most common intracranial schwannoma,
usually located in Meckel's cave but also in cavernous sinus (10%).
III,
IV and VI cranial nerves neurofibromas in their cavernous portion are extremely rare.
On MRI,
schwannomas appear as well-circumscribed lesions,
isointense on T1WI and hyperintense on T2WI with cystic degeneration and avid and heterogeneous contrast-enhancement.
Neurofibromas are similar to schwannomas since target sign of neurofibromas is virtually never encountered intracranially.
They are slow-growing tumours and bone remodeling,
best depict on CT,
may be present.
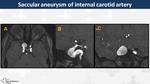
Internal Carotid Artery (ICA) Aneurysm (Fig. 32)
Supraclinoid ICA and ICA/PCoA junction are the second most common location (30%)
On MRI,
they appear as a flow void on T2WI,
but if they are thrombosed,
signal intensity on T1WI is variable depending on the age of the clot.
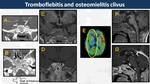
Cavernous sinus thrombosis (Fig. 33)
Rare condition in children (4.5/1,000,000/year) presented with headaches and III cranial nerve palsy.
It is secondary to contiguous spread of paranasal sinus or dental infection or orbital cellulitis.
Coalescent mastoiditis is a very rare source of cavernous sinus thrombosis.
On CT,
they appear as a distended cavernous sinus with non-fat density filling defect.
Hyperdense thrombus is detected in 25% of cases.
Indirect carotid-cavernous fistulas may be present in chronic cases.
On MRI,
absent flow void of ICA is detected both on T1WI and T2WI.
Sphenoidal sinus conditions:
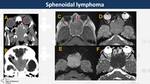
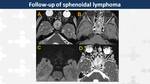
Sphenoidal lymphoma (Fig. 34, Fig. 35)
Primary or secondary non-Hodgkin lymphomas (DLBCL or Burkitt) are rarely located in paranasal sinuses but they entail a high incidence of visual disturbance and also facial swelling,
nasal obstruction,
rhinorrhea and epistaxis.
On CT,
they appear as soft-tissue masses with bone destruction.
On MR,
they show high signal intensity on T2WI with variable contrast-enhancement.
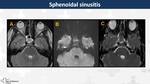
Sphenoidal sinusitis (Fig. 36)
Acute inflammation secondary to an upper respiratory tract infection presented in young adults with fever,
headache and nasal congestion.
On MRI,
they appear as mucosal thickening isointense on T1WI,
hyperintense on T2WI,
with contrast-enhancement.
Fluid shows marked restriction on diffusion.
Sphenoid sinus mucocele
They account for only 1-2% of all paranasal sinus mucoceles.
On CT,
they appear as sinus opacification with bone expansion and erosion.