Once reviewed the key concepts of gastric cancer,
the next step is to approach its imaging features.
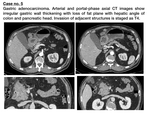
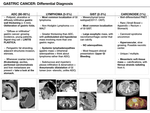
Gastric cancer consists of four main histopathologic subtypes,
each of them presenting different biological behaviour and distinct imaging appearance on which radiologists can rely on to reach an accurate diagnosis.
a.
GASTRIC ADENOCARCINOMA
It is the dominant subtype of gastric cancer (accounting for almost 90-95% of all cases).
General concepts
- Gastric ADC presents high incidence in Asia,
South-American Andine region and Eastern Europe.
- It mostly affects to >60-70 y.o.
patients (rare in < 40 y.o.) with male predominance.
- The disease is anodine at early stages.
Symptoms,
such as,
consitutional syndrome,
persistent abdominal pain,
disphagia,
GI bleeding,
vomiting or bowel obstruction,
usually occur once the malingancy is disseminated.
- Many factors play important roles in the development of gastric ADC.
Among these,
the following ones are well-established risk factors linked to gastric ADC:
- Inherited genetic and hereditary factors.
- H.
pylori chronic infection.
This gram-negative bacteria is considered a class I carcinogen of gastric cancer by the WHO.
It is also associated to MALT - Lymphoma.
- Dietary factors,
specially high-salt diet,
N-nitroso compounds (present in processed meat) and smoked food.
- Lifestyle factors,
with special mention to smoking and alcohol consumption.
- Others: radiation exposure,
older age,
male sex,
lack of physical activity and low socioeconomic status.
- Certain entities have been described as premalignant conditions: chronic inflammatory conditions and hypochlorhydria (chronic gastritis,
pernicious anemia),
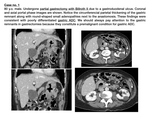
gastrectomy (specially Billroth-II, Fig. 3),
adenomatous polyps (> 2cm),
gastroesophageal reflux and Barrett's esophagus,
etc.
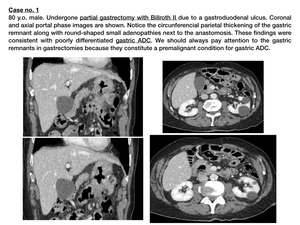
Fig. 3
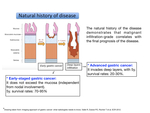
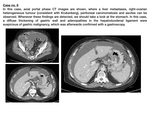
Natural history of disease
Gastric carcinoma is a slow-developing malignancy.
On the basis of the depth of infiltration of the gastric wall,
the disease is described as early-staged (when the tumour does not exceed the mucosa) or advanced (when invasion of deep layers occurs). Fig. 4
Malignant degeneration to early carcinoma is supposed to happen within 10-15 years,
while,
in general,
it takes 2-3 years for an early-staged cancer to degenerate into an advanced one.
Malignant infiltration-grade correlates with final prognosis of the disease,
as depending on the stage of the disease,
survival rates change significantly.
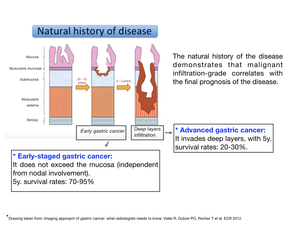
Fig. 4
Classification of gastric ADC ( Fig. 5 )
1.
Macroscopic anatomy
Advanced gastric carcinomas can be defined as exophytic,
ulcerated,
infiltrative or combined after the gross appearance of the tumor.
This classification is known as Borrmann's.
- Type I.
Polypoid growth or exophytic tumour (approx.
44%)
- Type II.
Fungative growth or ulcerative (but well circumscribed) tumour (approx.
7%).
- Type III.
Ulcerating infiltrative growth (approx.
40%).
- Type IV.
Diffusely infiltrative growth (approx.
9%); also referred to as linitis plastica.
2.
Microscopic anatomy
There are three main well-established classification systems based on microscopic features.
- WHO 2010 classification: It recognizes four major histologic patterns: tubular,
papillary,
mucinous and poorly cohesive (including signet ring cell carcinoma),
plus uncommon histologic variants.
- Lauren classification: It subdivides gastric adenocarcinoma into intestinal (55%),
diffuse (32%) or mixed (13%) subtypes. Fig. 6
- Ming classification correlates with Lauren classification and recognizes two main subtypes: expansive (38%,
equivalent to Lauren's intestinal),
infiltrative (46%,
equivalent to Lauren's diffuse) and mixed (16%).
Fig. 6
| INTESTINAL or EXPANSIVE |
DIFFUSE or INFILTRATIVE |
|
- Elder patients (> 60 y.o.)
- Male predominance
- Related to premalignant conditions
- Mucinous and tubular histology
|
- Young patients (30-40 y.o.)
- Growing incidence
- Male = female
- No related to premalignancy.
- Poorly cohesive histology
- Signet-ring cells; linitis plastica.
|
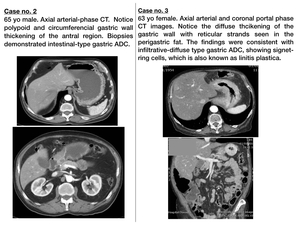
Fig. 6
Linitis plastica
Gastric linitis plastica is a diffuse or infiltrative type of carcinoma characterized by proliferation of signet-ring cells. It implies poor prognosis.
It is related to E-Cadherin mutation,
which provokes poor celular cohesion with consequent desmoplastic reaction and strong peritumoral fibrosis.
This results in rigidity of the gastric wall (also described as "leather-bottle stomach") as well as in tubular morphology of the gastric lumen. Fig. 7
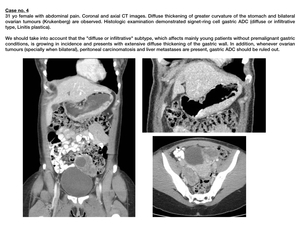
Fig. 7
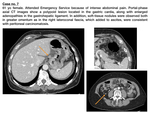
Imaging features
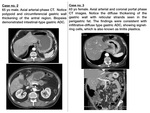
Gastric adenocarcinoma's radiological appearance includes a wide spectrum of findings.
The following imaging findings should draw the attention of radiologists,
being necessary to rule out gastric malignancy when present:
- Focal or diffuse wall thickenings (≥5 mm),
excluding false "thickenings" secondary to collapsed stomach or suboptimal gastric distension.
- Polypoid or ulcerative lesion with invasion of deep layers.
- Locorregional enlarged lymph nodes,
paying special attention to those located in the hepatogastric ligament.
- Stranding of perigastric fat-tissue.
- Loss of fat planes between stomach and adjacent organs (pancreas,
duodenum,
transverse colon,
spleen or liver).
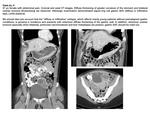
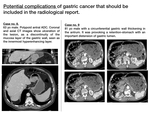
TNM classification
T: depth of the parietal infiltration
In our centre,
we don't have experience in staging gastric cancer's T by imaging techniques.
However,
imaging features for each of the T categories have been widely described in recent articles.
- T1.
Invasion of lamina propria,
muscularis mucosae or submucosa.
This stage is usually imperceptible.
- T2.
Invasion of muscularis propia.
It is the earliest noticeable stage for radiologists,
seen as transmural enhancement,
focal gastric wall thickening or small linear strands into the fat plane.
- T3.
Adventitia infiltration.
Its radiological traduction is perigastric fat infiltration,
seen as reticular or irregular outer border of the lesion.
Fat plane around the lesion is blurred.
- T4.
Adjacent structures invasion,
seen as obliteration of the fat plane (T3-T4) or gross infiltration (T4) of the involved organ. Fig. 8
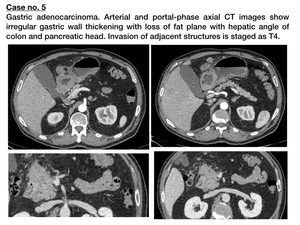
Fig. 8
N: affected lymph nodes
On the basis of regional lymph nodes infiltration,
N category is determined as follows:
- N1: metastases in 1 - 2 regional lymph nodes.
- N2: metastases in 3 - 6 regional lymph nodes.
- N3: metastases in >7 regional lymph nodes.
Regional lymph nodes include 16 perigastric lymph-stations. In the radiological report,
it should be included whether it exists infiltration of irresecable lymph-nodes,
which are considered as distant metastases (M1),
and include:
- Retro-portal lymph nodes
- Retro-pancreatic lymph nodes
- Lymph nodes adjacent to Superior Mesenteric and Middle Colic vessels,
as well as paraaortic lymph nodes.
Radiological criteria for malignangt lymph-node enlargement are: size (short axis >10 mm),
focal necrosis,
loss of fatty hilum,
round morphology,
heterogeneous & increased enhancement.
M: distant metastases
Distant disease (M1) is determined when:
- Liver,
lung,
adrenal,
renal,
bone or any other solid-organ metastases exist.
- Retro-portal or retro-pancreatic,
Superior Mesenteric and Middle Colic vessels or paraaortic lymph-stations infiltration exists.
- Peritoneal carcinomatosis or Krukenberg tumours are present.
Fig. 9
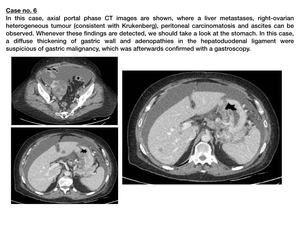
Fig. 9
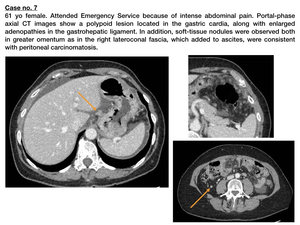
Fig. 10
*Cancer of esophagogastric junction:
In the 8th edition of TNM classification for gastric cancer,
the definition of esophagogastric junction is revised such that cancers involving the esophagogastric junction with the epicenter of it:
- Within the proximal 2 cm of gastric cardia are staged as adenocarcinomas of the esophagus.
- Epicenter distal to the proximal 2 cm of gastric cardia are staged as stomach cancers.
Tumors not affecting the EGJ should be staged as gastric cancer even though located in the proximal 2 cm of gastric cardia.
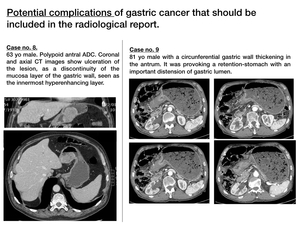
Fig. 11
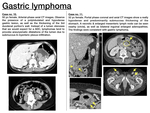
B. GASTRIC LYMPHOMA
Primary gastric lymphoma is the most common extranodal non-Hodgkin lymphoma and represents a wide spectrum of disease,
ranging from indolent low-grade marginal zone lymphoma or mucosa-associated lymphoid tissue lymphoma (MALT) to aggresive diffuse large B-cell lymphoma.
MALT lymphomas are rare,
but present an indolent course and have an overall better prognosis.
Although rare (it accounts for less than 5% of primary gastric neoplams),
stomach is the most common gastrointestinal site for this malignancy,
with almost 35% of all gastrointestinal lymphomas being gastric.
Imaging features (Fig. 12)
- Compared to ADC,
lymphomas produce a more diffuse and extensive parietal infiltration,
usually involving more than one gastric region.
In addition,
they produce a thicker & more hypodense polylobulated gastric wall enlargement,
arising from the submucosa.
- Lymphomas produce a denervation of submucous and myenteric plexus,
with a subsequent lumen dilatation of the affected region.
Therefore,
in contrast with adenocarcinomas,
which tend to be obstructive,
lymphomas are non-obstructive and produce the so-called aneurysmatic dilatations of GI tract.
- Systemc lymphadenopathies orientate to lymphomas.
In fact,
when a gastric lesion and infrarrenal and/or inguinal pathologic adenopathies coexist,
lymphoma must be ruled out.
As a conclusion,
gastric lymphoma has to be excluded when an extensive and polylobulated gastric lesion is detected,
specially when the lumen is dilated instead of reduced (as we would expect for ADC) and systemic adenopathies are present.
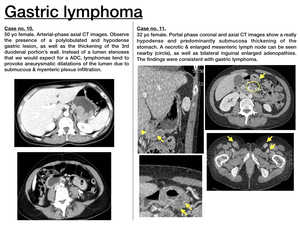
Fig. 12
C.
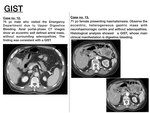
GASTRIC GIST
Gastrointestinal stromal tumours (GISTs) are the most frequent mesenchymal tumour subtype,
derived from Cajal's interstitial cells and characteryzed by C-KIT mutation and subsequent CD117 expression.
As the rest of mesenchymal tumours,
GISTs are submucous tumours; thus,
endoscopic ultrasound plays an important role for its detection.
Stomach is the most common localization for GIST tumours (60-70% of all),
followed by the small bowel.
However,
GISTs constitute a rare primary gastric cancer subtype,
accounting for approx.
2-3% of all.
The vast majority (70-90%) of GISTs are benignant.
The aggressivity-grade has direct prognostic implications and is determined on the basis of mitotic index (Ki67) as well as size of the tumour.
However,
low-grade GISTs may also present with metastases.
Therefore,
it is really difficult to assign potential malignancy depending on radio-histological criteria.
Imaging features ( Fig. 13 )
- Large size (>5cm),
adjacent structures invasion or distant metastases are suggestive of aggressive tumour.
- The majority of GISTs present as large exophytic masses with peripheral contrast-enhancement and/or central necrohaemorrhagic foci or ulcer,
which explains that the most usual clinical form of presentation is digestive haemorrhage.
These necrohaemorrhagic foci may calcify.
- Unlike ADC and lymphoma,
GISTs don't disseminate via lymphatic system and thus,
pathologic adenopathies will not be present.
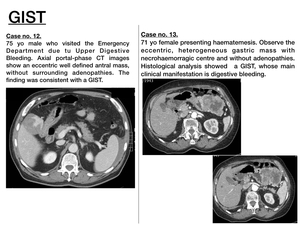
Fig. 13
D.
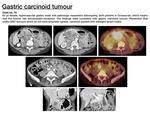
GASTRIC CARCINOID TUMOUR
Carcinoid tumours are derived from Kultsichtsky cells and constitute a well-differentiated subtype of neuroendocrine tumours (NET).
Most of carcinoid tumours (70-80%) are gastrointestinal but unlike lymphoma and GISTs,
whose most common localization is stomach,
gastric carcinoid tumours are really rare,
accounting for < 1% of all primary gastric neoplasms.
Carcinoid syndrome (including a variety of symptoms such as flushing,
diarrhoea,
heart failure,
bronchoconstriction and emesis) is rare in this localization.
These tumours are slow-growing neoplasms with an overal good prognosis (5-year survival rates vary from 70% to 80%).
Imaging features ( Fig. 14 )
- Carcinoid tumours are solitary or multiple submucous and hypervascular tumours,
sometimes presenting a necrotic centre.
- Their most characterisitc feature is the remarkable desmoplastic reaction they provoke in the adjacent mesentery,
causing a soft-tissue mesenteric mass with fibrous strands radiating from it (stellate-pattern).
- Octreoscan results useful for detection of primary tumour as well as distant metastases.
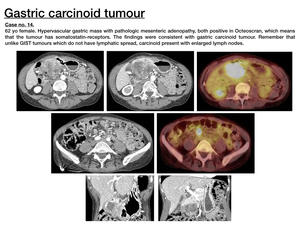
Fig. 14
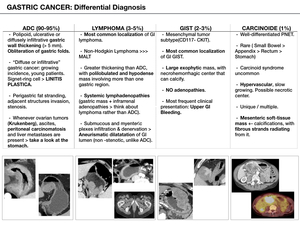
Fig. 15