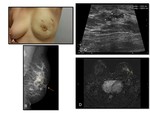
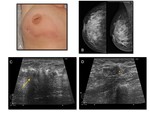
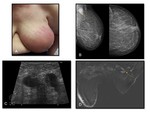
At imaging,
granulomatous mastitis can manifest with a variety of nonspecific findings,
which often mimic the appearances of malignancy.
A standard diagnostic protocol has not yet been established.
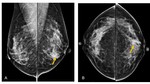
The first step for diagnosis is usually the mammography.
If diagnosis is unclear,
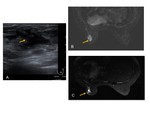
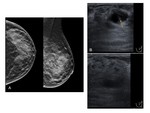
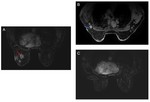
ultrasound and MRI are performed ( Fig. 1 ,
Fig. 2 ,
Fig. 3 ,
Fig. 4 Fig. 5 ).
In patients younger than 40 years who present with a palpable abnormality,
mammography is often omitted and the initial evaluation is performed with ultrasound (2).
Ultrasound enables further characterization of mammographic findings,
consequently,
it is nearly always performed.
Mammography and ultrasound are often sufficient for the approach to IGM,
but MRI may be indicated when ultrasound and mammographic evaluation cannot visualize lesions due to parenchymal edema.
In addition,
as IGM generally affects younger females,
MRI may also be useful in cases where mammography or ultrasound is inconclusive due to the character of the breast parenchyma (6).
It can also be recommended for the follow-up of cases with histological confirmation,
to evaluate the success of conservative treatment,
to see the extent of inflammatory process,
to evaluate the contralateral breast,
to evaluate possible residual disease after treatment and to diagnose coexisting inflammatory carcinoma.
Excluding these indications,
MRI does not provide additional information and probably will not be helpful in differential diagnosis (2,
7).
Mammography: The most frequent mammographic features are focal asymmetry with or without skin thickening and architectural distortion; diffuse asymmetric opacity with trabecular thickening or an irregular focal mass (2,
3,
4,
6,
7,
8).
Additionally,
findings sometimes include axillary adenopathy (3,
6).
Suspicious microcalcifications are hardly ever present in association with IGM (9).
Often,
there are no mammographic findings,
especially in the setting of heterogeneously or dense breasts.
Therefore,
normal mammographic reports are detected up to the half of the patients (7).
In conclusion,
mammography alone cannot be used to an accurate diagnosis because of the non-specific nature of the mammographic features and particularly in the young population that is affected,
as the above mentioned findings may not be detected in women with dense breast tissue (6).
Ultrasound: hypo-echoic or heterogeneous ill-defined lesions with or without tubular extension are the most common manifestations.
The tubular extension demonstrates how IGM insinuates around the breast lobules rather than destroys them (4).
It can be associated with parenchymal heterogeneity and abscess formation.
These findings are detected in almost 60% to 70% of the patients (3,
6,
8).
However,
we can see only parenchymal edema and generalised soft tissue thickening without any focal lesion.
Although irregularity has been thought to be a marker for malignancy,
there are no clear association between irregularity of the lesions detected via ultrasound and the absence of IGM.
The presence of a mass with irregular borders requires the histopathological analysis for differentiation of IGM from malignant pathologies (7).
Detection of fistula tract formation extending to the skin surface and skin thickening via ultrasound is another important imaging finding for IGM,
present in a high number of patients (7).
Doppler ultrasound examination highlights the hypervascularity of the lesion in up to 75% of the patients (9,
10).
It has been reported that unilateral axillary adenopathy with smooth reactive cortical thickening and preserved fatty hilum can be seen (3,
8,
10).
Also,
some studies reported an increased parenchymal heterogeneity or focally decreased echogeneity,
with both posterior acoustic enhancement and shadowing having been described,
but these findings are not specifically associated with IGM (3,
4).
Evaluation of elastic features by using sonoelastography has also been studied to reach more accurate diagnosis.
A statistically significative difference has been shown between IGM and malignant breast tissues based on the shear wave velocity marginal and internal values (7).
MRI: the MR imaging manifestations of IGM,
in the same way that ultrasound and mammographic appearances,
vary greatly.
Focal or diffuse asymmetrical hypointense signal on T1 images and hyperintense on T2 images (representing edema) without significant mass effect are the most frequent findings.
During contrast enhanced,
IGM is more frequently presented as heterogeneously enhancing mass,
lesions with rim enhancement or non-mass lesions enhancement with segmental distribution rather than regional distribution (6,
7,
9,
10,
11).
Although the majority of cases are presented with progressive or plateau patterns of enhancement,
the enhancement kinetic properties are nonspecific.
Due to this variability,
MR imaging cannot be used to reliably differentiate inflammatory breast cancer from granulomatous mastitis (2,
6,
9,
11,
12).
Some small lesions demonstrating confluency or well-defined borders,
T2 hyperintensity,
and rim enhancement at MR imaging have been presumed to represent microabscesses (4,
6,
9).
Additional MR imaging findings include axillary lymphadenopathy,
nipple or skin thickening,
nipple retraction,
sinus tracts,
and parenchymal distortion (4,
9,
12).
Ductal ectasia and periductal enhancement are commonly additional findings.
It is a valuable finding for distinguishing IGM from invasive cancer.
It has been also reported that IGM is seen as non-masslike lesions with restricted diffusion,
so Diffusion-weighted MRI is not helpful in the differentiation with malignant lesions (13).
The role of the breast imaging in the surveillance,
presurgical,
and posttreatment evaluation of confirmed IGM is to establish the multiplicity and location of IGM lesions,
measure the size of the lesion,
identify abscess formation and the related possibility for intervention,
evaluate the stability of the lesion,
evaluate the treatment response and,
finally,
identify metachronous disease and local recurrence (2).
The most commonly detected imaging findings,
in addition to the clinical examination,
can be used for the diagnosis and follow-up of IGM with acceptable safety margins.
Nevertheless,
in the presence of any suspicious sign a biopsy is required.
Main differential diagnosis
The main differential diagnoses for IGM include malignancy and other benign inflammatory and granulomatous conditions of the breast,
including infective mastitis,
tuberculosis,
systemic lupus erythematosus mastitis,
Wegener's granulomatosis and sarcoidosis.
The most important thing is to know how to differentiate breast carcinoma,
especially Inflammatory Breast Cancer (IBC),
from other benignant pathologies.
The differential diagnostic will be based on mammography and ultrasound characteristics in conjunction with clinical history,
fundamentally; and,
in case of doubt,
the final diagnosis will be achieve by histopathology.
Focusing on the main differential diagnosis,
IBC is an aggressive form of breast cancer that typically occurs in women between the 4th to 5th decades,
compared to IGM,
which affects more frequently women in the 3rd decade.
Thus,
age is a useful differential characteristic to take into account.
According to clinical presentation,
in contrast to IGM,
which rarely involves the skin extensively,
IBC characteristically involves more than one-third of the breast skin (14,
15).
Peau d’orange,
warm breast with or without an underlying palpable mass,
asymmetric breast engorgement,
axillary adenopathy and rapid onset to manifestation of symptoms less than 3 months of the initial presentation are other typical characteristics (16).
Mammographic features include inflammatory changes such as extensive skin edema and trabecular thickening,
which are less common with IGM.
Tumor mass,
asymmetry,
architectural distortion and increased breast density may be present in both cases (2).
Ultrasound may also show characteristically extensive skin thickening (the most common and obvious finding on ultrasound),
breast edema and dilated lymphatics.
We can also find pectoral muscle invasion and axillary involvement.
Other findings described previously are not so characteristic.
( Fig. 6 ).
At MR imaging we can see breast enlargement,
extensive skin thickening,
axillary adenopathy,
dilated lymphatics,
ductal ectasia and periductal enhancement,
T2 hyperintensity images,
abnormal parenchymal enhancement and irregular breast mass (which are frequently multiple,
small,
and confluent) with rapid enhancement and washout kinetics,
which are more suggestive of IBC (2,
17,
18). There is another entity that clinically can simulate the IBC,
it is the block lymphatic drainage pathways due to axillary adenopathies.
( Fig. 7 ).