IMAGING TECHNIQUES
Plain radiographs
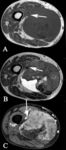
Initial assessment of soft-tissue sarcomas should begin with radiographic evaluation.
Often unrewarding,
but may reveal important diagnostic information
- Secondary bone involvement
- To distinguish myositis ossyficans and tumoral calcinosis from mineralization of soft-tissue sarcomas
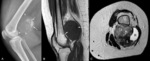
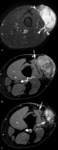
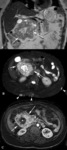
- Mineralization or fat that assists diagnosis [extraskeletal chondrosarcoma and osteosarcoma,
liposarcoma,
synovial sarcoma (Fig. 1)]
Ultrasound
Ultrasound (US) is the modality of choice for evaluation of superficial soft-tissue mass.
MRI should be performed when US features are not diagnostic of a benign tumor
Computed tomography
Current utility in soft-tissue sarcomas is limited due to radiation and less contrast resolution than MRI
It is the modality of choice for local staging when MRI is contraindicated or not available,
and to detect lung metastasis
MR imaging
It is the modality of choice for local staging
Protocol
- Patient position: Comfortable and with the lesion close to the isocentre of the magnet
- Cutaneous markers.
At least one anatomical landmark should be visible
- Coil selection should cover the entire lesion and the nearest joints both proximal and distal to the tumor
- Field of view (FOV) must be large enough to evaluate the entire lesion and allow appropriate local staging.
A reduced FOV focused on the lesion is preferred (increase the spatial resolution allowing markedly improved anatomic delineation of adjacent anatomic structure)
- Axial plane is the most useful primary.
Most neurovascular structures and muscles can be readily identified
- Long-axis planes.The best assessment of the entire lesion and its relationship to the important surrounding anatomic structures
- Conventional sequences.
Combination of T1 and T2-weighted sequences without and with selective fat-saturation.
Short tau inversion-recovery (STIR) sequence in areas where fat suppression is heterogeneous
- Diffusion-weighted imaging (DWI) evaluates random motion of water molecules (restricted diffusion implies higher cellularity).
Potential pitfalls: apparent diffusion coefficients (ADC) map values are elevated in benign and malignant myxoid tumors.
- Dynamic contrast-enhanced sequences. Evaluates enhancement patterns over time and identifies viable tumor (versus necrosis) for biopsy.
It is mandatory in myxoid sarcomas
CHARACTERIZATION
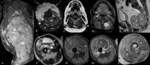
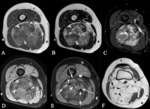
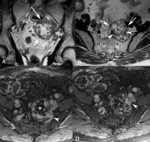
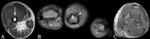
MRI features favoring malignancy (Fig. 2)
- Large size (> 5 cm)
- Infiltrative tumor growth [systemic inflammation markers (C-reactive protein) and infiltrative growth have been associated with poor prognosis and high-grade soft tissue sarcomas]
- Deep location
- Heterogeneous signal intensity
- Bone/neurovascular involvement
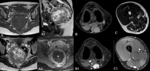
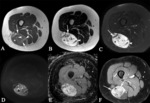
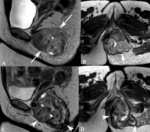
Pitfalls (Fig. 3):
- Both benign and malignant soft-tissue tumors often have well-defined margins (eg.,
well-differentiated,
dedifferentiated,
myxoid liposarcoma,
malignant solitary fibrous tumor,
synovial sarcoma)
- Superficial sarcomas often appear less “aggressive” than deep sarcomas (smaller).
MRI features suggestive of some subtype of soft-tissue sarcomas
Liposarcomas (Table 2)
Liposarcomas show high signal on T1-weighted sequence with areas of low-signal on T1 and T2-weighted sequences with selective fat-saturation.
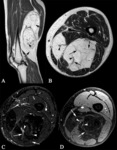
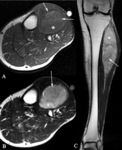
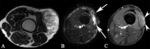
Well-differentiated liposarcoma (Fig. 4)
Most common subtype of liposarcoma.
Location.
Extremities (thigh),
retroperitoneum,
and paratesticular and inguinal regions
- Well-circumscribed
- Composed mostly of fatty tissue (75% of the lesion)
- Nonlipomatous components (thick septa > 2 mm and focal nodules < 1 cm
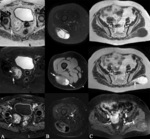
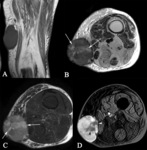
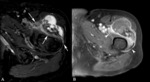
Dedifferentiated liposarcoma (Fig. 5)
Location.
Retroperitoneum and abdominopelvic region. Rare in extremities.
It shares imaging features with well-differentiated liposarcoma (always occurs within a well-differentiated liposarcoma)
- Well-circumscribed fat-containing masses with prominent soft tissue component (> 1 cm)
- Intense heterogeneous enhancement
- Necrosis and edema
- Thick septations
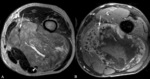
Myxoid liposarcoma (Fig. 6)
The second most common subtype of liposarcoma
Location.
Extremities
MRI features are diagnostic:
- Well-circumscribed,
and multilobulated lesions
- Low signal intensity on T1-weighted images and marked high signal intensity on T2-weighted MR images
- Diagnostic clues: lacy or linear fat in septa or small nodules and contrast-enhancement (peripheral,
nodular or diffuse)
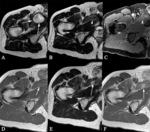
Pleomorphic liposarcoma (Fig. 7)
High-grade liposarcoma and less common subtype of liposarcoma.
Location.
Extremities
- Well-circumscribed or ill-defined mass containing little or no fat
- Heterogeneous signal intensity
Myxoid sarcomas (Table 3)
Myxoid tumors should be considered malignant when at least one of the
following criteria is present (Fig. 8):
- lll-defined margin
- Hemorrhagic component
- A tail sign (high signal intensity on T2-weighted images with contrast enhancement along fascial and vascular planes)
- Intra-tumoral fat (myxoid liposarcoma)
MRI features can suggest an accurate diagnosis in some myxoid sarcomas such
as myxoid liposarcoma (Fig. 6),
myxofibrosarcoma and low-grade fibromyxoid
sarcoma
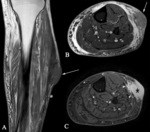
Myxofibrosarcoma (Fig. 9)
Common fibrous sarcoma of the extremities in the elderly
- Characteristic infiltrative growth pattern along fascial and vascular planes
- Tail sign (wispy streaks or “tails” of high signal intensity on T2-weighted images with contrast-enhancement).
This sign also can be seen on undifferentiated sarcomas
- These tails must be resected to fully remove the gross tumor and minimize the risk of local recurrence
Recurrences are common and of higher grade.
Metastases to sites atypical for most other soft-tissue sarcomas (pleura,
peritoneum,
adrenal glands,
and bone).
Low-grade fibromyxoid sarcoma (Fig. 10)
Extremities and trunk of young to middle-aged adults
Prolonged latency between diagnosis and local recurrent (usually as multiple lesions)
Imaging reveals features consistent with the fibrous and myxoid content
- Fibrous components are T1 and T2 hypointense with avid heterogeneous enhancement
- Myxoid component are T1 hypointense and T2 hyperintense with poor heterogeneous enhancement
Other useful MRI features
Location
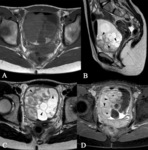
- Gastrointenstinal stromal tumor (GIST).
Origin from the wall of the stomach or intestine (rare extragastrointestinal)
- Leiomyosarcoma (large vessels)
- Dermatofibrosarcoma protuberans (Fig. 11).
Origin in the dermis but spreads locally into the deeper tissues
- Botryoid embryonal rhabdomyosarcoma (Fig. 12).
Mucosal epithelial lined viscera (bladder,
biliary tract,
vagina or upper respiratory tract).
Grape-like ("botryoid") growth pattern
Typical history
- Desmoid (Gardner's syndrome,
surgery,
pregnancy)
- Breast angiosarcoma (radiotherapy)
- Malignant peripheral nerve sheath tumors (Fig. 13) (neurofibromatosis)
Serpentine high-flow vascular channels
- Higher-grade vascular lesions (Fig. 14) (hemangioendothelioma,
hemangiopericytoma,
and angiosarcoma)
Peritumoral high signal intensity on T2-weighted images
- Common in high-grade soft-tissue sarcomas (Fig. 15)
- Suggests peritumoral tumor cell infiltration
LOCAL STAGING
Relation to the fascia
Tumor depth has been eliminated and excluded as a factor of staging the last eidition of the Cancer Staging Manual of the American Joint Committee on Cancer (AJCC).
However,
relation to the fascia should be recorded in the report because:
- Myxofibrosarcoma and undifferentiated sarcoma usually extends from the main mass for a substantial distance along fascial planes (Fig. 9).
It is important to plan their complete resection
- Skin thickening (Fig. 11).
Skin involvement need to be identified pre-operatively to enable adequate surgical planning and avoid wound breakdown
Tumor size
- The largest size and three-dimensional measurement
- In the new eight edition of AJCC,
tumor size categories include: T1: < 5 cm,
T2: >5 cm,
T3: 10-15 cm,
T4 >15 cm
Peritumoral edema
- The extent of the peritumoral edema should be clearly identified to be removed at surgery (Fig. 15)
- To differentiate between peritumoral edema and tumor tissue can be difficult.
Peritumoral edema usually no mass effect,
no distortion of soft- tissues and ill-defined margins
Neurovascular involvement
It is essential to determine the possibility of limb salvage
Neurovascular bundle is involved
- Contact between tumor and vascular or neural circumference >180º
- Complete encasement of an artery or vein (Fig. 16).
Limb-sparing surgery,
but arterial and/or venous reconstrucction is usually necessary
- Complete encasement of a major nerve.
Limb-sparing surgery with functional loss or amputation
- Contrast-enhanced tumoral thrombus.
Poor prognosis.
Limb-sparing surgery is possible,
but it needs arterial and/or venous reconstruction and survival decreases
Sparing of the neurovascular bundle
- Fat plane preserved with abutting or displacement of the neurovascular bundle by tumor (Fig. 17).
Limb-sparing is possible,
but may result in marginal resection requiring post-operative radiotherapy
Extension to adjacent bone or joint
Bone and joint involvement are rare.
Bone involvement is an independent poor prognostic and it is most common in synovial sarcoma
No bone involvement
- Preservation of a soft-tissue interface between the tumor and bone (fat plane on T1-weighted sequence without fat saturation)
Cortical involvement
- Signal alterations in the cortex on T1-weighted and T2-weighted sequences
Medullary involvement
- Medullary cavity signal alterations on T1-weighted without fat saturation
- Tumor abuts bone do not predict bone invasion (highly sensitive but not specific)
STRUCTURED RADIOLOGIC REPORT
Structured MRI reports of MRI for local staging of soft-tissue sarcomas should include:
Primary tumor
- Size (three-dimensional measurement)
- Location in compartment and relation to the fascia
- Margin (well or poorly defined)
- Signal characteristics,
suggesting fatty,
cystic,
solid,
myxoid tissue or hemosiderin
- Degree of necrosis
- Peri-tumoral edema (is considered to contain microscopic tumor)
- Prominent intra-tumoral vessels (may lead to pre-operative embolization)
Local extent
- Bone (cortex,
medulla) and joint involvement (capsule,
joint space)
- Neurovascular involvement (clear,
contact or partial or complete encasement).
Extent in the axial and longitudinal planes
- Extension through the fascia
- Skin involvement
- Presence and location of tumor tails and satellite lesions
Regional lymph node involvement
IMAGE GUIDED BIOPSY
Adequate tissue representing different components of the sarcoma is mandatory
for complete pathologic evaluation
Biopsy sample (core needle biopsy or open biopsy) comprising only or mostly of necrotic tissue is inadequate for complete evaluation
Fine needle aspiration biopsy has a limited role
US or CT guided biopsy depend:
- Radiologists preference
- Mass location and the potential highest-grade component
To determine the best biopsy approach:
- Biopsy approach should be indicated previously with an arrow on an axial MR image
- Always consult with an orthopedic surgeon (biopsy tract is resected en bloc along with the mass)
Biopsy should be done after obtaining all the necessary imaging techniques
- Hemorrhage and edema/inflammation after biopsy can alter the imaging appearance of the tumor
MONITORING OF RESPONSE TO TREATMENT BEFORE SURGERY
Increasement of tumor volume (hemorrhage and necrosis post-neoadjuvant therapy) is not associated with an adverse prognosis
Size of tumor not necessarily is a robust measure of treatment necrosis
- Stable size.
Unknown response
- Increase in size.
Nonresponse or necrosis or hemorrhage (Fig. 19)
Functional MR imaging should be used to improve the prediction of response to preoperative neoadjuvant therapy
- Dynamic contrast-enhanced sequences.
Use change in enhancement patterns (slope)
- DWI.
Increasement of ADC value after neoadjuvant therapy,
when compared to the pretreatment value,
suggests a positive response
LOCAL RECURRENCE
A marginal negative resection is the primary surgical aim with complete removal of the tumor.
Local recurrence (3-35%) occurs within the first 2–3 years after initial surgery (increased risk in high-grade tumors,
deep location,
and positive or close surgical margins)
The surgical margin status predicts the risk of local recurrence
- Peritumoral reactive edema is considered to contain microscopic tumor. Risk of local recurrence (40%) if the tumor alone is scooped out from the reactive edema around it
- Ideal surgical margins: 2 cm of skin,
fat or muscle and 1 mm of fascia,
periosteum or adventitia
Local recurrence of tumor needs to be differentiated from areas of granulation,
hemorrhage,
seromas and radiation-induced changes.
T2-weighted sequences
- No high signal intensity or mass.
Usually not tumor
- High signal intensity mass.
Tumor,
seroma,
hematoma, postoperative inflammation or radiation-induced change (Fig. 20)
- Extensive soft-tissue edema on MRI after unplanned surgical excision increases the likelihood of residual microscopic tumor and justify ample margins at tumor bed excision and consideration of adjuvant therapy
Contrast-enhanced images.
Smaller/subtle recurrences
- Difference between tumor and seroma (Fig. 21)
- The absence of nodular enhancement does not exclude microscopic residual tumor in the operative bed following unplanned excision
Postoperative inflammation/fibrosis may appear masslike
- DWI.
Characterize diffusion properties for confirmation.
Exclusion of recurrent tumor (high ADC).
Limited in myxoid sarcomatous tumor recurrences (high ADC values)
- Dynamic contrast-enhanced sequences: Tumor enhances early and rapidly,
while inflammation/fibrosis enhances gradually