Keywords:
Cancer, Screening, MR, Oncology, Breast
Authors:
V. S. Vilar1, A. I. Gomes2, J. V. Cubo1, A. C. S. Racy3, R. L. D. M. Ribeiro4, E. Francolin1; 1Sao Paulo/BR, 2Sao Paulo, Sao Paulo/BR, 3Sao Paulo, SP/BR, 4000/BR
DOI:
10.26044/ecr2019/C-1490
Results
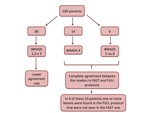
From the 100 patients,
4 were classified as BI-RADS 5 or 6 and 16 as BI-RADS 4.
In these 20 patients there were complete agreement between the readers regarding the final BI-RADS categorization in FAST and FULL protocols.
Despite the BI-RADS agreement,
in 4 of these 20 patients we found one or more lesions in the FULL protocol that were not seen in the FAST one.
In relation to BI-RADS 1,
2 and 3 the agreement was smaller mainly due to the presence or absence of purely small cystic lesions (Fig.
4).
Interpretation time was significantly lower with the abbreviated protocol.
The mean time of interpretation of the abbreviated protocol (FAST) was about 21 minutes,
while the mean time of interpretation of the complete protocol (FULL) was about 59 minutes.
Clinical relevance / application
Mammography is the gold standard method for screening of breast cancer.
However,
it has some limitations,
generating overdiagnostics of prognostically unimportant cancers,
as well as subdiagnostics of cancers that are indeed relevant (1).
MRI has emerged as an essential tool in the diagnosis and staging of breast cancer (2,3).
Breast MRI with abbreviated protocol was proposed with the objective of reducing the cost associated with this method,
reducing the exam time (up to 3 minutes),
but especially due to the reduction of the reading time and interpretation of images by the radiologist (4,5).
Given this current situation,
our study investigated whether the use of an abbreviated breast MRI protocol would be effective for the screening of breast cancer in random women.
Although our data predicted good results,
there were some limitations: our sample was small and our study was retrospective.
A larger sample of patients is needed to ensure that the applicability of the FAST protocol is effective for screening of breast cancer.
Maybe it would be also interesting to include T2-weighted sequence to avoid false positives regarding cystic lesions.
In the literature there are few (mostly retrospective) studies comparing the FAST protocol with the FULL protocol to screen a specific populaion,
such as women with dense breasts or high risk for breast cancer,
testing the sensitivity and specitivity of each.
These will be crucial for the evaluation and validation of this new protocol.