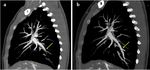
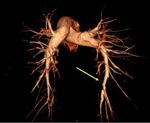
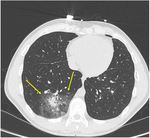
CTPA plays an essential role in evaluating patients with suspected CTEPH clearly depicting highly suggestive findings for CTEPH essentially divided into vascular and parenchymal signs10,11 (Figure 2).
Principal vascular signs are:
- vessel complete obstruction;
- vessel narrowing;
- intimal irregularities;
- bands and webs;
- thickening of arterial wall;
- collateral systemic supply.
Principal parenchymal signs are:
- scars from prior pulmonary infarction;
- mosaic pattern perfusion;
- focal ground glass opacities;
- cylindrical bronchial airway dilatation;
- systemic perfusion of the peripheral pulmonary arterial bed.
Moreover CT may allow visualization of course,
connections and size of the ectasic bronchial arteries and associated mediastinal or chest comorbidities (mediastinal carcinoma,
fibrous mediastinitis,
sarcoidosis,
and pulmonary arteritis) or other diseases (pulmonary angiosarcoma).
CTPA may also result pivotal for the selection of patients who will benefit from surgery providing a vascular roadmap for surgical planning and delineating the proximal extent of the organized thromboembolic material,
with good endarterectomy plane correlation12.
Patients are considered eligible for resection if the disease is present in the main,
lobar,
or proximal segmental arteries,
a finding easily evaluable at CTPA8.
CTPA in fact is excellent for assessment of central CTEPH lesions,
but not equally excellent for evaluation of distal subsegmental lesions8.
Operability assessment may be a complex process routinely requiring a multidisciplinary team of CTEPH experts who to assess surgical accessibility,
presence of hemodynamic or ventilatory impairments,
and eventual underlying comorbidities which represent contraindications for PEA.
Patients with CTEPH are considered not eligible for PEA in presence of:
- distal lesions;
- comorbidity;
- age;
- residual pulmonary hypertension after PEA;
- patient rejection of PEA.
BPA represent an alternative treatment option for this group of patients: the European Society of Cardiology/European Respiratory Society guidelines state that BPA may be considered for patients who are technically inoperable or who carry an unfavourable risk/benefit ratio for PEA2.
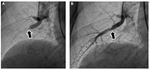
BPA is an interventional procedure based on the use of a balloon catheter to dilate pulmonary stenoses or obstructed lesions in the pulmonary artery (Figure 3) in order to improve haemodynamics and pulmonary perfusion.
During the planning of BPA,
each single artery is evaluated for indication of BPA according to the lesion type and distribution/location of the lesion13 based on a recent angiographic classification with a pivotal role in the identification of BPA target lesions6.
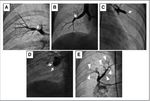
This classification identifies 5 different lesion types as follows (Figure 4):
-type A: ring-like stenosis lesion;
-type B: web lesion;
-type C: subtotal lesion;
-type D: total occlusion lesion;
-type E: tortuous lesion.
The main target vessel is usually a segmental or subsegmental pulmonary artery accessible with a minimum 1.5–2.0 mm balloon,
characterized by pouching defects,
webs or bands,
intimal irregularities,
abrupt vascular narrowing,
and complete vascular obstruction14 (Figure 5,
6).
Therefore BPA could potentially be performed in patients with9:
- distal distribution of vascular obstructions;
- unfavourable risk/benefit for PEA (i.e.
few lesions but severe haemodynamics,
or extreme surgical risk because of comorbidities,
etc.);
- persistent/recurrent pulmonary hypertension after PEA;
- need of rescue angioplasty after early failure of PEA;
- residual lesions after PEA;
- inoperable or persistent/recurrent CTEPH;
- inadequate response to medical therapy.
Conversely,
BPA is not recommended in presence of9:
- large amounts of organized thrombi in the proximal portion of the pulmonary artery as the thrombi is surgically easily accessible;
- complete obstructions at the orifices of the segmental arteries that do not show traces of distal arteries or pouching defects as it is extremely difficult for the guidewire to pass through these arteries;
- lesions located too distally in the subegmental pulmonary arteries for the ballon catheter to pass the lesion,
although BPA is mainly performed in the distal pulmonary arteries;
- severe renal failure and iodine allergy,
as the use of a contrast medium is essential in BPA.
The main BPA-related complications include vascular and lung injuries.
Vascular injuries,
secondary to wire perforation or angioplasty,
are in general characterized by dissection,
hemorrhage or rupture,
usually promptly diagnosed angiographically and treated with prolonged balloon inflation,
coil embolization,
covered stent placement8.
BPA-related lung injuries typically occur following the procedure and patients present respiratory symptoms (cough,
hemopstysis,
dyspnea,
hypoxia); the most common lung complication post-BPA is reperfusion oedema (Figure 7) characterized by airspace opacities in the territory of the angioplastied vessels on chest CT and usually managed conservately,
even if sometimes may require mechanical ventilation and percutaneous cardiopulmonary8,13.Although all recent BPA studies report remarkable improvements in term of haemodynamic effects (Figure 8) and symptoms14-17,
the outcome and the incidence of complication are higly dependent on the lesion types13; the success rate of BPA is almost 100% in case of ring-like stenosi,
web and abrupt narrowing with a low complication rate,
while is approximetely 90% in case of subtotal obstruction lesion and <50% in case of pouching defects13,18.Total occlusions lesions report the lowest success rate and tortuous lesions are associated with a high complication rate9.