Definition of early rectal cancer
Early rectal cancer is defined as a "rectal cancer with good prognostic features that might be safely removed preserving the rectum,
and that will have a very limited risk of relapse after local excision." (6).
This generally indicates T1 tumours with no local nodes (N0) and a low risk of lymph node metastases.
The risk of lymph node involvement increases with the presence of lymphovascular invasion and poor tumour differentiation.
Imaging of early rectal cancer:
MRI and endorectal ultrasound (ERUS) are complementary imaging modalities in the staging and pre-operative surgical planning of T1 tumours.
When used in conjunction,
they increase the accuracy of T-staging of early rectal cancers (7).
For staging T2 tumours or larger,
MRI is the modality of choice (6). Unlike ERUS,
MRI is also is able to identify lymph nodes within the mesorectum and within the pelvic side-wall (8).
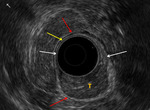
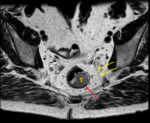
Case 1. Correlation of imaging in the same patient with a large rectal polyp on both ERUS ( Fig. 1 ) and on MRI ( Fig. 2 ),
suitable for TEO resection.
The he muscularis propria remins intact around the tumour,
which extends from approximately the 4 o'clock to 6 o'clock position.
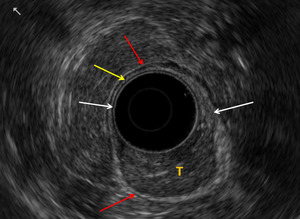
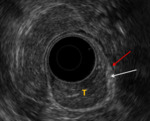
Fig. 1: Case 1. EUS images in a patient with a large rectal polyp. T = tumour. White arrow = Muscularis mucosa, hypoechoic – breached by the tumour. Yellow arrow = Submucosa, hyperechoic. Red arrow = Muscularis propria, hypoechoic – intact around the tumour
References: Truro/UK
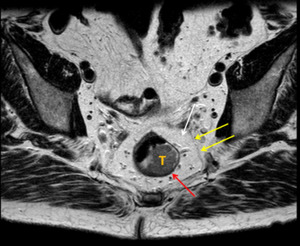
Fig. 2: Case 1. T2 axial images in the same patient with a large rectal polyp.
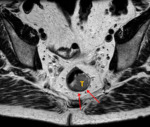
T = Tumour. Red arrow = hypoechoic muscularis propria, which is not breached by tumour. Yellow arrows = mesorectal fascia or circumferential resection margin. White arrow = mesorectal fat.
References: Truro/UK
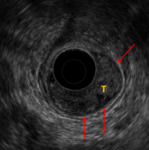
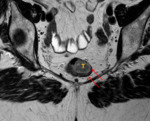
Case 2. Second patient with a large rectal polyp,
biopsy positive for adenocarcinoma: On both ERUS ( Fig. 3 ) and MRI ( Fig. 4 ) the muscularis propria is intact throughout with no evidence of invasion,
in keeping with a T1 lesion.
The patient underwent TEO resection and the final histopathology conirmed T1 tumour.
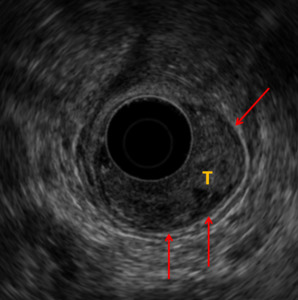
Fig. 3: Case 2. The low rectal polyp is seen on EUS. The hypoechoic muscularis propria (red arrow) is intact throughout with no evidence of tumour within it. If malignancy proven T1 at worst. T = tumour.
References: Truro/UK
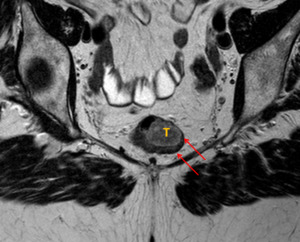
Fig. 4: Case 2. The invading edge of the tumour extends from 2 o'clock to 3 o'clock, but does not breach the hypoechoic muscularis propria (red arrow), making this a T1 cancer (at worst).
References: Truro/UK
Case 3. Third patient with a large rectal polyp and biopsy-proven adenocarcinoma: On ERUS ( Fig. 5 ) the muscularis propria is intact throughout with no involvement.
There is some irregularity of the submucosa in places.
On MRI,
the invading edge of the tumour extends from 4 o'clock to 6o'clock ( Fig. 6 ).
The tumour is confined to the muscularis propria.
The stage is therefore either T1 or benign,
the finl histopathology confirmed a T1 tumour.
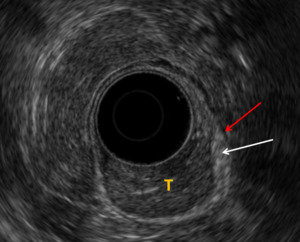
Fig. 5: Case 3. EUS demonstrates the polypoidal lesion in the rectum. The muscularis propria (red arrow) is intact. There is some irregularity of the submucosa (white arrow). Appearances are in keeping with a T1 lesion.
References: Truro/UK
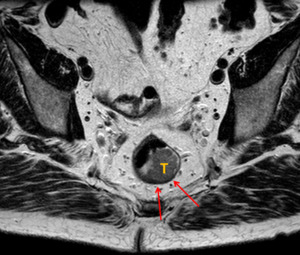
Fig. 6: Case 3. Axial T2 MRI images demonstrating that the invading edge of the tumour (T) extends from 4 o'clock to 6o'clock. The tumour is confined to the muscularis propria (red arrow). The T stage is therefore either T1 or benign. Final histopathology following TEO confirmed a T1 lesion.
References: Truro/UK
Case 4. Unfortuntely this patient was unable to have an MRI.
However,
ERUS ( Fig. 7 ) demonstrates T3 disease.
There is loss of the hypoechoic muscularis propria from the 9 o'clock to 11 o'clock position (red arrow) adjacent to the tumour (T).
This is in contrast to the presence of the normal hypoechoic muscularis propria seen elsewhere (green arrow).
This patient is not suitable for minimally invasive surgery.
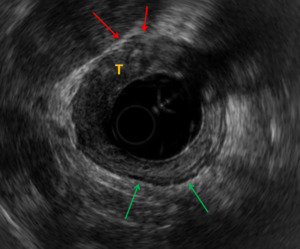
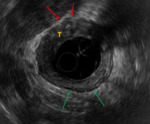
Fig. 7: Case 4. EUS demonstrates T3 disease. There is loss of the hypoechoic muscularis propria from the 9 o'clock to 11 o'clock position (red arrow) at the point of invasion of this semi-annular tumour (T). This is in contrast to the presence of the normal hypoechoic muscularis propria seen elsewhere (green arrow).
References: Truro/UK
Treatment Strategies
All patients deemed suitable for curative-intent resection of rectal cancer should receive a complete colonoscopy and digital rectal examination (6),
as the risk of synchronous colorectal cancers and colorectal polyps is approximately 3% and 30% respectively (9,
10).
There should be a multidisciplinary team meeting (MDT) discussion that involves the surgeon,
radiologist,
pathologist and oncologist when deciding what treatment method to proceed with.
This may involve a minimally invasive operation or a radical resection.
Discussion of the critical role of neoadjuvant chemotherapy is beyond the scope of this poster.
Evolving Surgical techniques
There is increasing utilisation of minimally invasive and sphincter preserving surgical techniques in early rectal cancer.
These minimally invasive techniques may be considered intraluminal or extra-luminal: Intra-luminal techniques include transanal endoscopic microsurgery (TEMS) or transanal endoscopic operations (TEO).
Extra-luminal minimally invasive techniques include the sphincter-preserving method of transanal total mesenteric excision (taTME) in curative-intent resection of rectal cancer.
Transanal endoscopic microsurgery (TEMS) or Transanal endoscopic operation (TEO)
Procedure Overview:
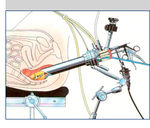
The patient is positioned in a lateral decubitus or supine position.
The scope is inserted via the anus with a three-dimensional viewing endoscopic system attached,
allowing passage of instruments and removal the polyp or T1 cancer via the anus ( Fig. 9 and Fig. 8 ).
TEO only differ from TEMS in that it involves the incorporation of high-definition vision.
The two procedures are similar,
and have no technical or clinical difference in outcome (11).
The resection margin is then marked and a full thickness incision is made,
which should include a sufficient border of normal mucosa.
This technique does not allow wide excision of the tumour or excision of the mesorectum with all lymph nodes.
The rectum is then washed out and the defect is closed with sutures via the TEM scope.
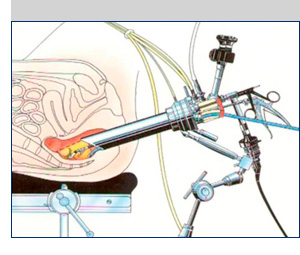
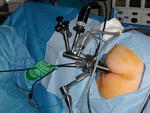
Fig. 9: Insertion of the TEMS scope through the anus, allowing passage of instruments for dissection and removal of the polyp or T1 cancer via the anus.
References: Oxford Colorectal Surgery 2011-2017
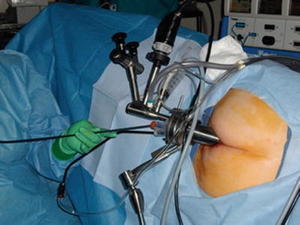
Fig. 8: Insertion of the TEMS scope through the anus, allowing passage of instruments for dissection and removal of the polyp or T1 cancer via the anus.
References: Hedrick, T. L., & Bleier, J. (2018). Transanal Approaches: Transanal Endoscopic Surgery. In Techniques in Minimally Invasive Rectal Surgery (pp. 17-37). Springer, Cham.
Complications (12):
- Conversion to radical surgery in up to 4.3% (12)
- Haemorrhage (27%)
- Urinary tract infection (21%)
- Suture line dehiscence (14%)
- Faecal incontinence – often temporary (1%)
Benefits in comparison to radical resection:
- Shorter operation time
- Shorter hospital stay: Same day or next day discharge (13)
- Avoid pelvic floor disruption,
perineal wound complications and stoma formation
- Improved post-operative quality of life (14)
- Comparable survival,
disease-free survival and distant metastases (14)
Drawbacks in comparison to radical resection:
- Limited data comparing oncologic outcomes of TEMS and radical resection
- Unable to excise local nodes
- Tumours that are less than 5 cm from the anal verge may not be visualized adequately with TEMS (15)
What the surgeon wants to know
Is this patient suitable for a TEMS/TEO procedure?
Careful patient selection is essential prior to local excision of early rectal cancer,
and should involve a multidisciplinary team meeting discussion between the surgeon,
radiologist and histopathologist.
To avoid the need for a radical salvage resection the following selection criteria are recommended (15):
Suggested criteria for local excision by TEMS:
|
Physical examination
|
Tumour < 3 cm
Tumour <30% bowel circumference
Tumour within 15 cm of dentate line
Tumour freely mobile
|
|
Imaging (ERUS and MRI)
|
Tumour limited to submucosa (T1)
No lymph node involvement (N0)
|
|
Histology
|
Well to moderately differentiated
No lymphovascular invasion
No perineural invasion
No mucinous or signet cell component
|
Following a TEMS procedure,
has the resection been adequate?
The definitive histopathology must be reviewed following TEMS.
Ideally TEMS would be the endpoint of operative management,
however it may turn out to be part of the diagnostic work-up.
If there are inadequate resection margins or if the histology is high grade,
then a radical salvage resection should be offered to the patient (16).
How should patients be followed up after TEMS?
In the context of favourable histology and clear resection margins,
an appropriate follow-up strategy is required to ensure early detection of local recurrence or distant metastasis.
MRI is a feasible follow-up strategy (17). However,
nodal recurrence remains challenging and postoperative changes can also make interpretation of MRI difficult,
leading to underestimation of recurrence (17).
Therefore surveillance varies centre to centre,
and may involve both colonoscopy and MRI.