The rationale to treat an UIA is to prevent the rupture and its consequent aneurysmal subarachnoid hemorrhage(SAH) that can lead to significant morbidity and mortality.
Our understanding of the natural history and treatment outcomes of cerebral aneurysms has significantly increased over the last few decades,
but choosing the optimal management for each patient requires the careful consideration of numerous medical,
clinical and anatomic factors.
CLINICAL PRESENTATION
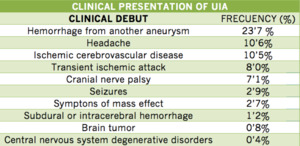
Fig. 8
References: J Neurol Neurosurg Psychiatry 2006;77:572–578
DIAGNOSTIC IMAGING TESTS
The radiographic studies available to delineate the size and morphologic features of an intracranial aneurysm are CT angiography (CTA),
magnetic resonance angiography (MRA),
and angiography by direct intra-arterial catheterization (digital subtraction angiography),
which is considered the gold standard.
CTA uses thin-section contrast-enhanced CT with the aid of software generated images to show cerebral vessels in three-dimensional views.
These reconstructed images can be generated in a few minutes,
and they allow evaluation of the vasculature in close relation to the brain and the bones of the skull base,
therefore facilitating surgical planning.
Sensitivities of CTA range from 77% to 97% and specificities range from 87% to 100%.
Sensitivity for aneurysms smaller than 3 mm drops to a range of 40% to 91%.
The role of CTA is very limited in patients with impaired renal function,
since a large bolus of contrast material is often administered.
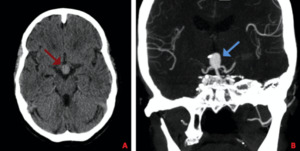
Fig. 9: Non-enhanced CT reveals the presence of an aneurysm at the top of the basilar artery (A), which is subsequently confirmed with a CTA (B)
MRA is highly sensitive and specific for the detection of intracranial aneurysms.
MRA has a sensitivity of 70% to 99%,
and a specificity of 100% for aneurysms 3mm or greater in diameter; however,
the sensitivity diminishes for very small aneurysms (under 3 mm in diameter) to as low as 40%. False-negative and false-positive aneurysms detected on MRA were mainly located at the skull base and middle cerebral artery. MRA is more difficult to use in critically ill patients because it takes considerably more time to perform.
For those patients in whom contrast administration is con- traindicated (including patients with renal failure in whom MRI-related gadolinium contrast is contraindicated),
MRA is the screening method of choice because gadolinium contrast administration is not needed.There are data to support that MRIs may be useful in follow-up studies post treatment.
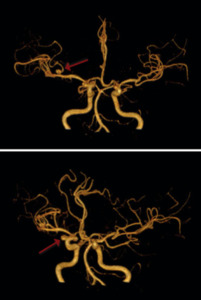
Fig. 10: Reconstruction of 3D-TOF MRA shows the presence of two aneurysms, located in the right ICA and in the right MCA.
Digital subtraction angiography (DSA) remains the gold standard imaging modality for intracranial aneurysms.
Using techniques like 3D rotation,
small aneurysms can be identified.Another advantage of DSA is morphological assessment of the aneurysms and relationship of small branches and perforators near the aneurysms.
DSA may also help distinguish between true infundibula and aneurysms.Its risks,
even in the hands of experienced operators,
include neurologic complications occurring in 1.0% to 2.5% of cases,
with permanent impairment in 0.1% to 0.5%.
Other associated risks include femoral-artery injury (0.05% to 0.55%),
groin hematoma (6.9% to 10.7%),
and adverse renal effects induced by contrast material (1% to 2%).
Overall,
brain MRA or CTA are the methods of choice for screening of unruptured aneurysms because of their noninvasive nature; however,
a cerebral angiogram is sometimes used to better clarify the details of an aneurysm.
MANAGEMENT
Optimum management of an unruptured intracranial aneurysm should involve the comparison of the risk of aneurysmal rupture without any intervention with the risks of surgical clipping or endovascular treatment.
The factors that should be considered include:
- Aneurysmal factors: location,
size,
morphology,
whether a thrombus exists within the aneurysm,
and the presence of a daughter sac or multiple lobes,
- Patient factors: age,
medical history,
history of subarachnoid hemorrhage,
and family history of subarachnoid hemorrhage.
There are three management options for unruptured cerebral aneurysms: conservative management,
surgical clipping or endovascular treatment.
Currently,
there is a lack of prospective randomized controlled trials to guide therapy,
particularly in comparing intervention with conservative management.
Many published articles are retrospective in nature,
and they lack objective short- and long-term assessment of outcomes.
Currently our best information regarding management of unruptured aneurysm is based on observed rates of complications in aneurysm treatment compared to the natural history of unruptured aneurysm.
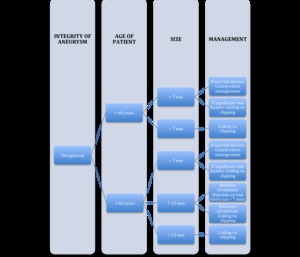
Fig. 11
References: Norman Ajiboye, Nohra Chalouhi, Robert M. Starke, Mario Zanaty, and Rodney Bell. 2015. Unruptured Cerebral Aneurysms: Evaluation and Management. The Scientific World Journal
Volume. Article ID 954954.
ENDOVASCULAR TREATMENT
The different endovascular techniques include (1) packing the aneurysm with coils,
with or without adjunct techniques such as balloon inflation or stent placement at the aneurysm neck for more difficult cases; (2) use of flow diverting stents; and (3) use of flow disrupters devices.
- The most common form of endovascular management is the deployment of the detachable coils into the aneurysm via microcatheter.
These coils cause local thrombosis and isolation of the aneurysm from the parent artery.
Patients that are ideal candidates for the use of coils are aneurysms with a narrow neck (<4 mm) and low dome-to-neck ratio (<2).
Adjunct techniques such as balloon inflation or stent placement at the aneurysm neck are increasingly used in some of the more difficult cases such as wide neck (>4 mm) or high dome-to-neck ratio (>2).
Adjunctive techniques for coil embolization prevent coils from protruding through the aneurysm neck into the parent artery,
therefore reducing the risk of thromboembolic complications.
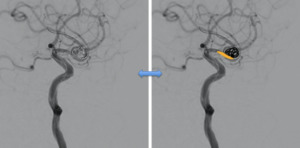
Fig. 12: Treatment of the sacular aneurysm located in the ICA was perfomed with balloon assisted coiling technique
- Flow diverting stents such as pipeline embolization device (Covidien,
Irvine,
CA) are indicated for large unruptured saccular or fusiform intracranial aneurysms (>10 mm) of the anterior circulation from the petrous segment to the superior hypophyseal segment.
Pipeline embolization devices (Covidien,
Irvine,
CA) consist of tightly braided mesh that allow flow into vessel branches,
but cases stagnation of blood in the sac which results in the occlusion of the aneurysm.
Patients need to be pretreated with dual antiplatelet therapy (aspirin and clopidogrel).
The recommendation is for the patient to be on lifelong daily aspirin (81–325mg) and clopidogrel therapy is continued for a duration of 3 to 6 months after procedure.
The results of a multicenter retrospective study of the pipeline embolization device were recently published showing a 30-day morbidity and mortality of 6.3% and a long-term neurologic morbidity and mortality of 8.4%.
The morbidity and mortality were the highest in the posterior circulation group at 16.4% and the lowest in the internal carotid artery aneurysm (with size <10 mm) group at 4.8%.
The pipeline embolization study included a total of 793 patients with 906 aneurysms; when patients with ruptured aneurysm (9% of cases) were excluded,
the overall morbidity and mortality for unruptured aneurysms was 5.7%.
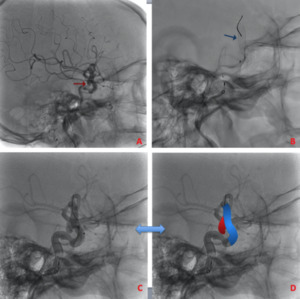
Fig. 13: A wide-neck sacular aneurysm depending on the ICA (A) was embolized with a Pipeline flow diverter device (B,C and D).
- Intrasaccular flow disruption is an innovative endovascular treatment that aims to create thrombosis of the aneurysmal sac by disrupting the blood flow in the aneurysm by covering its neck with an intrasaccular device (WEB-DL; Sequent Medical,
Aliso Viejo,
California).
According to series presented in the literature,
this approach applies mainly to complex wide-neck bifurcation aneurysms (MCA,
basilar artery,
AcomA,
ICA).
Because no antiplatelet medication is needed for aneurysm treatment with the WEB,
a rapid thrombosis of at least of the distal compartment and aneurysm dome is usually observed.
Retreatment of previously embolized,
recanalized aneurysms is also feasible with the WEB (7.1%).
WEB treatment failures were due to navigation difficulties (3 patients) or inadequate visualization of the device.
The microcatheters used for theWEB deployment are relatively larger compared with those used for simple coiling; this difference makes it more difficult to navigate in tortuous anatomy,
which mainly applies to AcomA aneurysms.
After deployment,
the WEB position is sometimes difficult to verify completely,
due to poor visualization of the device,
especially when it is superimposed on bony structures of the skull base.
Improvement of its radiopacity will potentially improve the feasibility of the treatment.
In fact,
the failure rate is not higher compared with that observed with standard coiling if one takes into account the complexity of aneurysms treated with the WEB and the learning curve in each center.
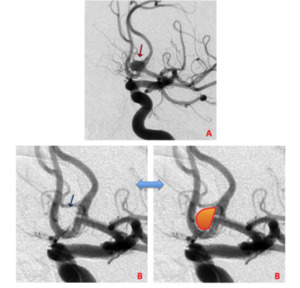
Fig. 14: Anterior communicating artery unruptured aneurysm with a wide neck, before (A) and immediately after (B) WEB deployment.








