I- The story of LI-RADS
Liver Imaging and Reporting Data System (LI-RADS) aims to standardize the terminology,
technique,
interpretation,
reporting,
and data collection of liver imaging in patients at risk for hepatocellular carcinoma (HCC).
It was developed by a multi-disciplinary group of professionals in the field of liver cancer imaging including diagnostic and interventional radiologists,
hepatologists,
hepatobiliary surgeons,
and hepatopathologists.
[6]
LI-RADS was first released by the American College of Radiology (ACR) in 2011.
This was followed by three major updates in 2013,
2014 and 2017.
[7,8,9,10]
LI-RADS version 2018 (LI-RADS v2018) is the fourth edition of this data system.
[11] This update was required due to the recent adoption of LI-RADS into the clinical practice guidelines of the American Association for the Study of Liver Diseases (AASLD).
[12]
LI-RADS uses the term observation to refer to an area with an imaging appearance that is distinctive from the surrounding liver parenchyma.
[13]
The CT/MRI LI-RADS diagnostic algorithm describes a four-step approach to the assessment of liver observations which stand out relative to composite background liver tissue,
at multiphasic CT or MRI.
It is intended for use only for untreated observations without a histologic diagnosis in patients who are considered at high risk for HCC.
[11,13] (Figures 13-16)
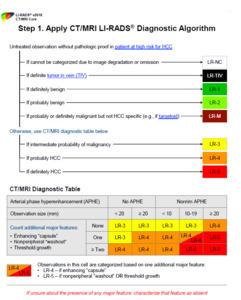
Fig. 13: Step 1 of the LI-RADS algorithm is the designation of a preliminary LIRADS category
References: Reproduced from LI-RADS v2018 with permission from the American college of Radiology
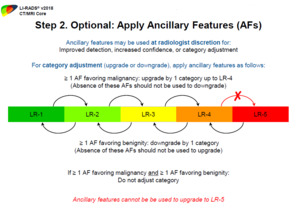
Fig. 14: Step 2 of the algorithm is the optional application of ancillary features for improved detection, increased confidence, or category adjustment excluding upgrading from LR-4 to LR-5, which is not allowed
References: Reproduced from LI-RADS v2018 with permission from the American college of Radiology
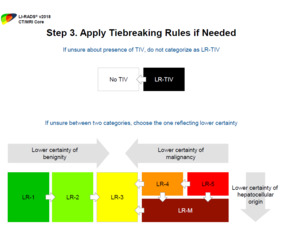
Fig. 15: Step 3 is the application of tiebreaking rules in situations of diagnostic uncertainty; if a radiologist is unsure between two categories, the category with the lower certainty should be assigned
References: Reproduced from LI-RADS v2018 with permission from the American college of Radiology
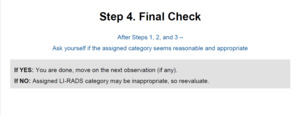
Fig. 16: Step 4 is the final check to verify that the assigned category is reasonable and appropriate
References: Reproduced from LI-RADS v2018 with permission from the American college of Radiology
LI-RADS 2011 version 1.0 included criteria and lexicon for CT and MRI with extracellular agents (MRI-ECA).
Version 2013 added new algorithmic display and illustrative atlas for CT and MRI-ECA.
In version 2014,
new material on hepatobiliary agents (HBA) was added,
designation A and B was removed from categories LR-4 and LR-5 and key modifications were introduced to achieve congruency with the Organ Procurement and Transplantation Network (OPTN) [14] and AASLD.
[12]
In 2017,
ultrasound LI-RADS and CEUS LI-RADS were released,
treatment response algorithm was added to the CT/MRI LI-RADS and a manual was created.
The major changes in prior LI-RADS versions are summarized in figure 17 & 18.
[7,8,9,10,
14]
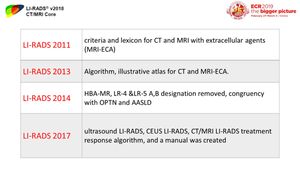
Fig. 17: Major changes in previous LI-RADS versions
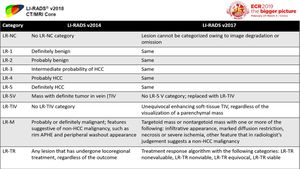
Fig. 18: Comparison between LI-RADS 2014 & LI-RADS 2017
References: Adopted from reference [14] Elsayes, K.M, et al. (2017) 2017 Version of LI-RADS for CT and MR Imaging: An Update. Radiographics. 37(7): p. 1994-2017.
Version 2018 changes from LI-RADS v2017 are:
1) Definition of threshold growth
The definition of threshold growth was revised and simplified.
Threshold growth is now defined as size increase of a mass by ≥ 50% in ≤ 6 months.
[11] (Figures 19 & 20)
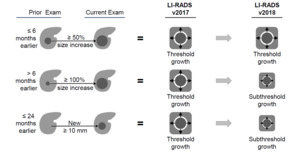
Fig. 19: Change in threshold growth definition in LI-RADS v2018.
Two other definitions of threshold growth used in the prior LI-RADS versions (i.e., a new observations ≥ 10 mm in ≤ 24 months and size increase of a mass by ≥ 100% in > 6 months) are considered subthreshold growth in v2018
References: Reproduced from LI-RADS v2018 with permission from the American college of Radiology
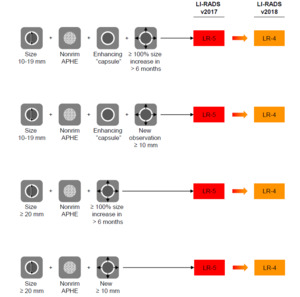
Fig. 20: The impact of change in threshold growth definition on categorization of certain subset of observations.
References: Reproduced from LI-RADS v2018 with permission from the American college of Radiology
2) Revised and simplified criteria for LR-5
LR-5 criteria were revised to match those advocated by AASLD.
The -g and -us designations were eliminated for simplicity.
[6,
11] (Figure 21)

Fig. 21: Changes in LR-5 criteria between LI-RADS 2017 and LI-RADS 2018
The LR-5g category was previously applied to observations 10–19 mm in size with nonrim arterial phase hyper-enhancement (APHE) on CT/MRI in addition to ≥ 50% increase in size in < 6 months, but without ‘‘washout’’ or ‘‘capsule’’. This category was originally introduced to facilitate translation to OPTN class 5 criteria—specifically OPTN 5A-g. In LI-RADS v2018, 10-19 mm observations with APHE and threshold growth are now simply categorized LR-5, as the threshold growth definition is identical to that of OPTN.
The LR-5us category was previously applied to observations 10–19 mm in size with nonrim APHE, ‘‘washout’’ and visibility at antecedent screening ultrasound, in absence of either threshold growth or a ‘‘capsule’’. In LI-RADS v2018, the requirement for antecedent visibility on ultrasound has been removed, and a 10–19 mm observation with nonrim APHE and nonperipheral ‘‘washout’’ is categorized LR-5.
The impact of these changes is shown in (Figure 22).
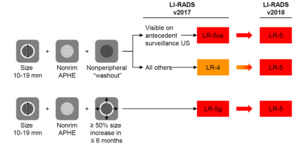
Fig. 22: The impact of change in LR-5 criteria in LI-RADS v2018
References: Reproduced from LI-RADS v2018 with permission from the American college of Radiology
New LR-5 criteria are shown in (Figure 23)

Fig. 23: New LR-5 criteria in LI-RADS v2018
References: Reproduced from LI-RADS v2018 with permission from the American college of Radiology
LI-RADS population:
LI-RADS is exclusively applied in a population of patients who are at high risk for developing HCC.
The aim of defining this population is to ensures high specificity of LI-RADS categories for the diagnosis of HCC.
(Figure 24)

Fig. 24: LI-RADS population (patients at risk of HCC)
References: Reproduced from LI-RADS v2018 with permission from the American college of Radiology
LI-RADS categories:
LI-RADS defines eight diagnostic categories based on imaging appearance that reflect the probability of HCC or malignancy with or without tumor in vein (Figures,
25-33).

Fig. 25: LI-RADS diagnostic categories
References: Reproduced from LI-RADS v2018 with permission from the American college of Radiology

Fig. 26: LR-NC category
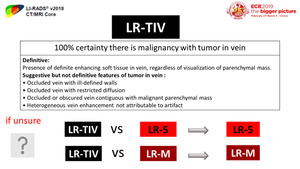
Fig. 27: LR-TIV category
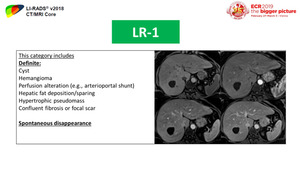
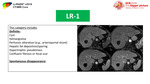
Fig. 28: LR-1 category
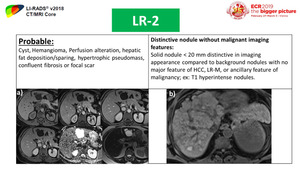
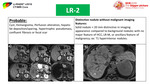
Fig. 29: LR-2 category
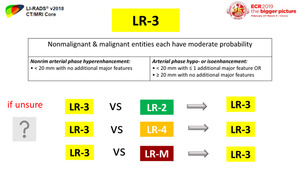
Fig. 30: LR-3 category
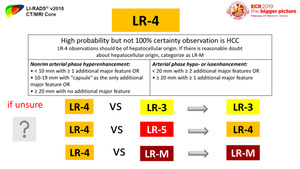
Fig. 31: LR-4 category

Fig. 32: LR-5 category
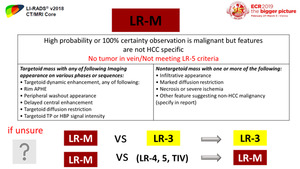
Fig. 33: LR-M category
LI-RADS imaging features
Major features for HCC [2,11]
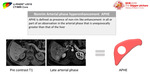
1) Non-rim arterial phase hyperenhancement (APHE) (Figure 34 & 35)
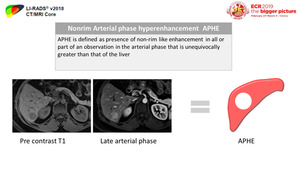
Fig. 34: Definition of APHE

Fig. 35: Difference between non-rim APHE and rim APHE
References: Reproduced from LI-RADS v2018 with permission from the American college of Radiology
2) Non-peripheral washout appearance ‘‘washout’’ (Figure 36-38)
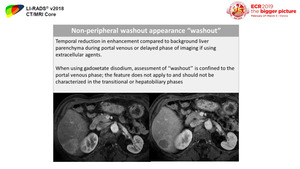
Fig. 36: Definition of non-rim washout appearance "washout"
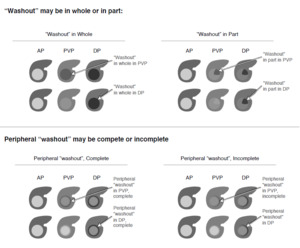
Fig. 37: Difference between non-peripheral and peripheral washout
References: Reproduced from LI-RADS v2018 manual with permission from the American College of Radiology

Fig. 38: Patterns of APHE & "washout"
References: Reproduced from LI-RADS v2018 with permission from the American college of Radiology
3) Enhancing ‘‘capsule’’ (Figure 39-40)
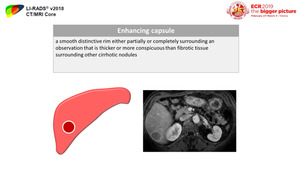
Fig. 39: Definition of enhancing capsule appearance

Fig. 40: Capsule appearance with ECA, Gadobenate and Gadoxetate
References: Reproduced from LI-RADS v2018 manual with permission from the American College of Radiology
4) Observation size (Figure 41)
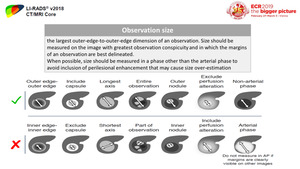
Fig. 41: Definition of observation size and accurate measurements
References: Reproduced from LI-RADS v2018 manual with permission from the American College of Radiology
5) Threshold growth (Figure 42)
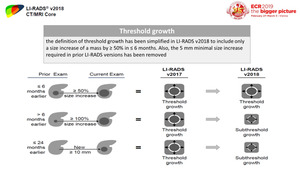
Fig. 42: Definition of threshold growth in LI-RADSv2018
References: Reproduced from LI-RADS v2018 manual with permission from the American College of Radiology
LR-M features
Definitions of LR-M features remain unchanged in LIRADS v2018.
(Figures 43-45)

Fig. 43: LR-M features

Fig. 44: Imaging appearance of targetoid mass
References: Reproduced from LI-RADS v2018 manual with permission from the American College of Radiology

Fig. 45: Differential diagnosis and algorithm for LR-M category
References: Reproduced from LI-RADS v2018 manual with permission from the American College of Radiology
Ancillary features (AFs)
AFs are divided into three subsets: those that favor malignancy in general (Figure 46); those that favor HCC in particular (Figure 47); and those that favor benignity (Figure 48)

Fig. 46: Ancillary features favoring malignancy in general not specific for HCC
References: Reproduced from LI-RADS v2018 manual with permission from the American College of Radiology
AFs allow for improved detection and increased confidence in diagnosis.
The use of AFs is optional at radiologist’s decision.
AFs may be used to adjust a LI-RADS category up or down by 1 category except for upgrading to LR-5 which is not allowed.
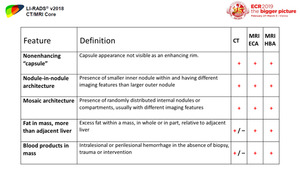
Fig. 47: Ancillary feature favoring HCC in particular
References: Reproduced from LI-RADS v2018 manual with permission from the American College of Radiology

Fig. 48: Ancillary features favoring benignity
References: Reproduced from LI-RADS v2018 manual with permission from the American College of Radiology
II- Interpreting approach
Do not consider LI-RADS diagnostic algorithm as a separate step to perform after finishing your MRI reporting.
Instead,
follow LI-RADS diagnostic algorithm simultaneously with your interpreting approach and checklist.
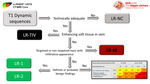
The first part of interpretation is to evaluate the T1 pre and post contrast dynamic sequences
1- evaluate if the technique is adequate.
If there is significant image degradation or omitted sequences that will prevent categorization,
assign LR-NC category,
if not,
proceed to the next step.
2- Look for the patency of the vessels,
if there is enhancing soft tissue within a vein,
assign LR-TIV.
3- Scan the entire liver carefully in a systemic way to ensure examining all
segments.
Search for distinct observations.
4- Detect if there is any targetoid masses or non-targetoid masses with infiltrative appearance or sever necrosis that will qualify to be categorized LR-M.
5- Check for benign observations like cysts,
hemangiomas or benign nodules,
if present,
assign LR-1 or LR-2 according to your judgment.
Indeed,
if there are no distinct observations,
assign LR-1 category.
If the previous possibilities are not present and you end up with observation/s which do not qualify to any of the previous categories,
then it is the time for LI-RADS diagnostic table.
6- Check for HCC major features.
It is worth noting that initial step of LI-RADS diagnostic algorithm and all major features of HCC are evaluable on the dynamic T1 sequences solely.
We have not done yet,
neither with the LI-RADS algorithm nor with the MRI interpretation.
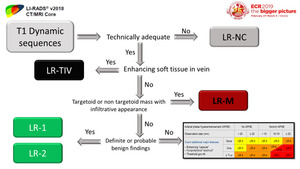
Fig. 49: Interpreting approach, part 1
The second part is to evaluate the unenhanced sequences
confirm the initial categorization performed in the first step,
declare any uncertainty of your categorization and apply the ancillary features.
Remember that although ancillary features are optional,
their application is in the favor of radiologist’s confidence about his/her diagnosis.
Evaluation of the ancillary features simultaneously with your routine checking of the MRI sequences will save your time and help you building up your interpreting checklist.
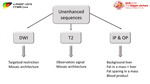
Evaluate DWI images:
Generally,
DWI is considered an excellent conspicuity sequence,
so check first for any initially missed abnormalities before evaluating the previously defined observations.
If initially LR-1 or LR-2 categories were selected,
confirm the benignity of the observation or otherwise,
upgrade to LR-3 if you are not sure.
If you previously assigned LR-5 category,
pay attention to presence of targetoid diffusion restriction,
if so,
question your previous selection and search for the presence of LR-M features.
If you previously assigned LR-M category,
check for marked diffusion restriction to confirm your selection.
Pay attention to the mosaic appearance on DWI,
if this is present,
you should rethink your previous LR-M assignment and reconsider possibility of LR-5.
T2 weighted images
Evaluate observation/s signal intensity preferably on SFE and moderately weighted T2 images.
If the examination contains T2 fat sat images,
be careful that observations may appear falsely of high signal if there is marked background fatty infiltration.
Check for the mosaic architecture.
Again,
if present and you selected LR-M initially,
rethink your choice.
Non-enhanced T1 OP and IP
Check for the background liver whether normal or steatotic.
In fatty livers,
check for fat sparing within an observation.
In non fatty livers,
check for focal fat within the observation.
Check for blood products within the observation.
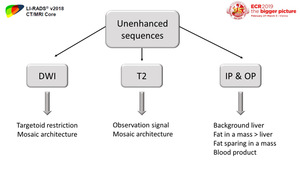
Fig. 50: Interpreting approach part 2
Note: If the MR study was performed with either gadoxetate or dimeglumine,
check for the presence of hepatobiliary phase hypointensity.
If this feature is present in absence of other features favoring benignity,
it may be used to upgrade LR-2 observation to LR-3 or LR-3 to LR-4,
but is less likely to alter your previous selection of LR-5 or LR-M.






![Fig. 18: Comparison between LI-RADS 2014 & LI-RADS 2017 References: Adopted from reference [14] Elsayes, K.M, et al. (2017) 2017 Version of LI-RADS for CT and MR Imaging: An Update. Radiographics. 37(7): p. 1994-2017.](https://epos.myesr.org/posterimage/esr/ecr2019/147112/media/798280?maxheight=150&maxwidth=150)