USEFULNESS OF CEUS BEFORE PERCUTANEOUS TREATMENT:
1) Characterization of focal lesions in ultrasound screening for hepatocellular carcinoma: CEUS is included in LIRADS®,
the internationally recognized guidelines for the standardization of diagnostic imaging for hepatocellular carcinoma.
Observations are classified into 8 categories ( Fig. 2 ):
-
CEUS LR-NC: cannot be categorized due to image degradation or omission
-
CEUS LR-1: definitely benign
-
-
Cyst: anechoic lesion with increased posterior through transmission showing no contrast enhancement in any phase
-
Hemangioma: variable echogenicity lesion with peripheral discontinuous globular enhancement in arterial phase followed by progressive centripetal contrast filling and iso- or hyper-enhancement in portal venous and late phases.
The filling may be complete or partial depending on lesion size.
-
Hepatic fat deposition/sparing: nonmasslike,
nonspherical,
hyper/hypoechoic area of parenchyma in a characteristic location for fat (around the gallbladder fossa and anterior to the right portal vein in segment IV).
-
CEUS LR-2: probably benign
-
-
Distinct isoenhancing solid nodule <10 mm
-
Nonmasslike isoenhancing observation not located in a region characteristic for fat deposition/sparing.
-
CEUS LR-3 nodules with interval size stability ≥ 2 years.
-
Nodules meeting these criteria are probably regenerative or low-grade dysplastic nodules.
-
CEUS LR-M: probably or definitely malignant lesion but not hepatocellular carcinoma specific.
Any of the following:
-
-
CEUS LR-TIV: definite tumor in vein (TIV)
-
-
CEUS LR-3,
LR-4,
and LR-5.
Described in Fig. 2 .
2) Monitoring changes in the enhancement pattern for LR3 or LR4 lesions to determine whether they progress to higher categories.
3) Study of nodules characterized as LI-RADS LR3,
LR4,
or LR-M at CT or MRI: The enhancement pattern of the lesion may differ between CT or MRI and CEUS,
because CEUS is purely intravascular.
This is why lesions deemed suspicious for malignancy at MRI can be studied with CEUS to improve characterization ( Fig. 3 ).
4) Nodules not visible on CT or MRI: A nodule detected at US might not be visible on CT or MRI.
In these cases,
the usual procedure is regular follow-up,
but LIRADS® suggests that CEUS can be particularly helpful in characterizing these lesions.
5) Complement to CT/MRI:
-
When the vascular phases acquired are not optimal: This limitation can lead to significant errors in characterizing lesions,
especially in hepatocellular carcinomas whose noninvasive diagnosis is based on enhancement criteria.
Ultrasound contrast agents have the great advantage that they can evaluate the different vascular phases in real time,
thus enabling more accurate characterization of lesion enhancement.
-
Pseudolesions: Vascular alterations such as spontaneous arterioportal fistulas are common in cirrhosis.
On CT and MRI,
when these shunts are small and pseudonodular,
they can be mistaken for tumors.
However,
on CEUS these pseudolesions are not seen,
so unnecessary biopsies can be avoided.
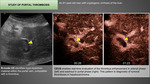
6) Study of portal thrombosis: The detection of tumor-related portal thrombosis in a cirrhotic patient is highly significant because its poor prognosis changes the therapeutic strategy completely.
The differential diagnosis between benign and malignant portal thrombosis is based on the detection of arterial neovascularization within the thrombus,
which indicates tumor-related thrombosis.
CEUS is sensitive and specific and can even be done in the same examination in which the thrombosis is detected (Fig. 4 ).
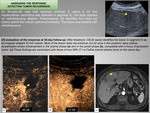
7) Locating a nodule to biopsy: When a lesion is indeterminate at CT or MRI,
one common approach is percutaneous biopsy for histologic study.
However,
US-guided percutaneous biopsy can be difficult and less likely to be successful when the lesion is not identified or is not clearly delimited on US.
CEUS enables accurate location and definition of relations with other vascular structures,
making the biopsy safer and more effective.
Moreover,
in large lesions,
it makes it possible to better identify the best areas to be biopsied.
These are the areas with greatest enhancement,
which have greater cellularity,
whereas the areas within the lesion that have less or no enhancement correspond to necrotic areas that should not be biopsied ( Fig. 5 ).
USEFULNESS OF CEUS DURING PERCUTANEOUS TREATMENT:
Percutaneous ablation aims to eliminate lesions without excising them; ablation can be done with chemical (ethanol,
acetic acid) or thermal (mainly radiofrequency or microwaves) agents.
All ablation techniques require imaging to identify the lesion and guide treatment.
US is the imaging technique of choice for guiding ablation,
and US guidance requires that the lesion be visible in B mode.
However,
occasionally,
a lesion identified for treatment by CT or MRI is ill-defined or impossible to detect in B mode.
This occurs most often in very heterogeneous livers or those that have more steatosis and/or fibrosis,
making it more difficult to perform US-guided ablation safely and effectively.
In these cases,
CEUS is immediately useful in identifying the lesion thanks to the differences in enhancement between the lesion and the surrounding parenchyma ( Fig. 6 ).
Local recurrence after treatment is a challenge because in B mode it is difficult to differentiate between a focus of recurrence and nonviable tissue,
although it is easy to identify the site of prior ablation.
On CEUS,
tumor foci enhance like the prior lesion,
enabling the radiologist to determine the exact location and guide the needle accurately,
increasing the effectiveness of the treatment.
Another advantage of CEUS is that it enables the position of the needle with respect to the lesion to be checked after it is introduced and to reposition it if necessary.
Furthermore,
contrast material can be repeatedly administered in the same session,
thanks to its excellent tolerability and safety ( Fig. 7 and video in Fig. 8 ).
USEFULNESS OF CEUS AFTER PERCUTANEOUS TREATMENT:
1) Evaluating the response: CT and MRI are considered the gold standards for evaluating the response to the treatment of liver tumors.
Their accuracy and wide field of view make it possible to evaluate the primary tumor as well as local and distal spread.
Nevertheless,
various studies have demonstrated that the diagnostic accuracy of CEUS is similar to those of CT and MRI in the follow-up of percutaneously treated tumors.
Thus,
CEUS should be considered another tool in the follow-up algorithm for treated tumors.
Completely ablated lesions have no enhancement inside or around them ( Fig. 9 ),
whereas persistent tumor foci are characterized by focal enhancement with characteristics similar to those of the treated tumor ( Fig. 10 and video in Fig. 11 ).
Various studies attest to the usefulness of CEUS immediately after ablation and within 24 hours after ablation; one of the advantages of this approach is that it makes it possible to identify regions that might not have been treated,
so they can be treated in the same session.
However,
it is important to remember that it is common to see increased enhancement of the region treated and the needle tract (about 10 mm thick) in the days following treatment due to hyperemia caused by an inflammatory reaction to ablation ( Fig. 12 ).
This finding is source of false-positives and false-negatives,
so a follow-up 30 days after treatment is recommended.
CEUS is especially useful in cases where CT and MRI cannot be done or are inconclusive.
Because US contrast agents are purely vascular,
they are very sensitive for the detection of abnormal residual vessels in treated lesions.
2) Evaluating complications: CEUS can detect complications immediately after the procedure; the main ones are:
-
Vascular thrombosis
-
Active peritoneal bleeding
-
Hepatic infarction
-
Hematomas and abscesses ( Fig. 13 )
-
Biliary tract lesions
-
Evaluating the extent of ablation: one of the factors that condition local recurrence is adequate ablation margins (10 mm for hepatocellular carcinoma and 5 mm for metastases).