1. IDIOPATHIC CHRONIC CHOLANGIOPATIES
1) Primary Sclerosing Cholangitis (PSC)
Primary sclerosing cholangitis is an idiopathic,
chronic,
fibrosing inflammatory disease of bile ducts that leads to bile duct obliteration,
cholestasis and can progress to cirrhosis,
end-stage liver disease.
Patients have an increased risk of hepatobiliary and colorectal cancer and median survival after diagnosis is 20 years [4,
5].
Approximately 70% of patients with PSC also have inflammatory bowel disease (IBD),
mainly ulcerative colitis,
being the diagnosis of IBD typically made first.
Up to 80% of patients with PSC have signs of IBD in endoscopy or biopsy whereas only 5 to 10% of patients diagnosed of ulcerative colitis will develop a PSC in their lifespan.
Most patients are asymptomatic at diagnosis and the disease is suspected when chronic cholestasis is detected in a blood test.
PSC symptoms are fatigue and pruritus,
and its presence in the moment of diagnosis is associated with worse survival rates.
Typical histopathologic feature of PSC is periductal fibrosis with minimal inflammatory cells.
This finding appears in less than 20% of biopsies,
so biopsy is not recommended [1].
Diagnosis of PSC is made when typical cholangiographic features are present after excluding secondary causes of cholangitis.
Endoscopic retrograde cholangiopancreatography (ERCP) was the classic diagnostic test,
but being ERCP an invasive technique with a rate of complications,
now MRCP is the first choice for diagnosing this condition.
MR cholangiopancreatographic findings include multifocal short-segment strictures in the intra and extrahepatic bile ducts,
alternating with normal or mildly dilated ducts (‘beaded appearance’); and peripheral pruning of the intrahepatic ducts [2].
PSC involves both intrahepatic and extrahepatic ducts in 75% of patients [1].
Areas of circumferential narrowing (webs) and focal saccular dilatations (diverticula) can be seen [4].
Cystic duct and gallbladder abnormalities can be part of the spectrum of the disease [7].
(Figures 2,
3 and 4).

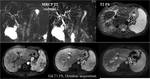
Fig. 2: Figure 2. Advanced PSC: MRCP and MRI findings. This 36 years old man has signs of PSC on MRCP with multiple stenosis on large intrahepatic ducts and in the extrahepatic bile duct and moderate dilatations, giving a beaded appearence to the biliary tree. Pancreatic duct is normal.
The liver has a rounded shape with sligthly increased signal intensity on T2 on posterior segments of right liver lobe and lateral segments of the left lobe. On Gd-enhanced images these liver segments show transient increased enhancement, with no focal liver lesions. Bile ducts show thickened walls with diffuse increase of enhancement. Splenomegaly and hylar lymph nodes are also seen.

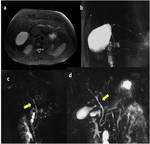
Fig. 3: Figure 3. PSC subtle MRCP changes. When PSC is suspected, subtle MRCP features should be analysed. Normal bile ducts have smooth contours and they should taper to the periphery of the liver. On images a and b axial T2WI and MRCP of a 48 y.o. man with UC and suspected PSC showing stenosis and subtle dilatations on intrahepatic bile ducts of the left liver lobe. On images c and d, a normal MRCP study of a 35 y.o. woman. There are areas of discontinuity on common and left bile ducts secondary to vascular crossing, but the biliary ducts are smooth and decreasing in size to the periphery of the liver.

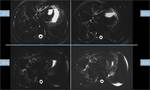
Fig. 4: Figure 4. PSC progression on MRCP images. These are axial thick MRCP slices of a patient with PSC showing progression of the disease on the intrahepatic bile ducts. Axial images are better suited to show the segmentary distribution of the disease on the early stages and the progressive deformation of the bile ducts and the liver.
Associated liver parenchymal changes are frequent,
and they can be detected on MRI.
The liver tends to have a rounded morphology due to hypertrophy of the caudate lobe and to atrophy of the left lateral and right posterior segments.
High signal wedged areas on T2 and DWI due to edema,
related to cholestasis,
inflammation and fibrosis (Figure 5).
Lymphadenopathies and signs of portal hypertension can also be found.
Contrast-enhanced T1WI show wall thickening and late mural enhancement of biliary ducts,
suggesting an underlying inflammatory process.
On arterial phase images,
enhancement of the liver parenchyma is often heterogeneous reflecting the segmental liver inflammatory changes.

Fig. 5: Figure 5. PSC. Parenchymal liver changes. This PSC patient has bile ducts stenosis and large pseudosaccular dilatations predominantly affecting the posterior segments of the right liver lobe and the lateral segments of the left lobe. These liver segments show higher signal intensity on T2 and DWI (better seen whit fat suppression) reflecting edema and inflammation and also increased arterial enhancement on DCE images. On T1 opposed phase these areas show fatty sparing, probably secondary to haemodynamic changes.
PSC patients bear a higher risk of cancer.
Cholangiocarcinoma is described in up to 10% of PSC patients.
Some MRI findings as a rapid progression of strictures and marked ductal dilatation proximal to a stricture suggest cholangiocarcinoma,
although the diagnosis in PSC is difficult due to the presence of multiple stenoses.
The tumor usually locates in the common hepatic duct or at the bifurcation,
and it shows mural thickening with fibrotic tumor enhancement pattern,
with delayed accumulation of contrast [4].
The risk of colorectal cancer in patients with ulcerative colitis increases by a factor of 4 when PSC is diagnosed (Figure 6 and 7).
Hepatocellular carcinoma is related to cirrhosis,
the end-stage of PSC,
but it occurs quite infrequently [8].
Therefore,
ACG Practise Guideline 2015 [9] recommends screening for cholangiocarcinoma with serial US or MR,
CA 19.9 and cholecystectomy if >8 mm polyps,
and also periodic colonoscopy,
but the level of evidence is low.

Fig. 6: Figure 6. Ulcerative colitis and PSC. This patient with known ulcerative cholitis presented with an stenosing neoplasm in the descending colon (A). Total proctocolectomy and ileo-anal anastomosis was performed. Six months later, liver metastases were diagnosed (C) and treatment included liver surgery (right posterior hepatic bisegmentectomy and metastasectomy on segment IVA). Two years later the patient is disease-free and developed cholestasis. On MRCP (D and E) stenosis on large intrahepatic and extrahepatic ducts, consistent with PSC were found. Reviewing the liver images on the first CT examination, subtle biliary abnormalities could be detected (B).

Fig. 7: Figure 7. 47 yo women with Ulcerative Colitis and cholestasis. A. Thick slice MRCP. B. Thin slice 3D MRCP. Note saccular dilatations in right intrahepatic biliary ducts and segmental stenosis of the biliary tree in left hepatic lobe. C. Axial T2 weighted image. D. Axial T2 fat saturated weighted image. Liver parenchymal wedge-shaped areas of increased signal intensity related to inflammatory changes, best shown in D due to the fast suppresion.
Small-duct primary sclerosing cholangitis is a variant of classical PSC that only affects intrahepatic ducts,
described in 5% of cases,
with a better long-term prognosis than PSC [10].
This entity manifests with cholestasis without cholangiographic abnormalities,
hence there is an indication for liver biopsy,
showing characteristic histological appearance as concentric periductal fibrosis (“onion skin”).
2) Primary Biliary Cholangitis (PBC)
Primary Biliary Cholangitis (PBC) is a chronic non-suppurative autoimmune cholestatic liver disease,
predominantly affecting middle-aged women (90%) [11].
It is a slowly progressive intrahepatic illness that can evolve into hepatic fibrosis,
cirrhosis,
hepatic failure and hepatocellular carcinoma in some cases.
Chronic cholestasis,
circulating anti-mitochondrial antibodies (AMA) and nonsuppurative destructive cholangitis and interlobular bile duct destruction is the characteristical triad of this disease.
Diagnosis of PBC is given by two of the following three criteria: AMA titer > 1:40; alkaline phosphatase (AP) > 1.5 times the upper limit of normal for > 24 weeks; and compatible liver histology [13].
Histopathologically,
PBC consists in a periportal inflammation that progresses to necrosis and destruction of small and middle-sized bile ducts.
Four stages of PSC have been described: cholangitis or portal hepatitis (stage 1); periportal fibrosis or periportal hepatitis (stage 2); septal fibrosis,
bridging necrosis or both (stage 3); and biliary cirrhosis (stage 4) [12].
In the above-mentioned scenario,
imaging is not essential for the diagnosis of PBC,
and no abnormalities are frequently found in cholangiography.
However,
MRCP can be useful in distinguishing PBC and other cholestatic diseases,
mainly PSC,
stone disease and other secondary cholangitis.
Some imaging features can be found in PBC as periportal halo sign (40%,
and more prevalent in advanced stages) [14].
Periportal halo sign is the only specific imaging finding and it consists of numerous rounded lesions showing low signal intensity in T1WI and T2WI,
involving all hepatic segments,
5 mm to 1 cm in size,
and centered on a portal venous branch,
without mass effect.
Periportal hyperintensity (cuffing) on T2 is a common but not specific sign present in early stages and it indicates active inflammation (Figure 8).

Fig. 8: Figure 8. PBC. (a, b) Axial T2 fat saturated images,(c, d) axial T1WI fat-sat without and with contrast material and (e) axial DWI showing slightly enlarged liver, hipointense periportal halo sign, lace like liver cirrhosis and some perihepatic fluid . (f) Axial DWI showing enlarged lymph nodes in gastrohepatic ligament.
Other common imaging findings in PBC are enlarged lymph nodes in porta hepatis,
gastrohepatic ligament or upper retroperitoneal space.
In more advanced stages signs of micronodular cirrhosis,
splenomegaly,
ascites,
portosystemic collaterals and portal vein thrombosis [14].
Active inflammation in portal triads may produce arterial-portal shunting,
which is seen in MRI as early contrast enhancement of portal veins.
MRCP is usually normal (excluding other cholangitis) but in cirrhotic stage,
large ducts can be displaced or irregular and small ducts are usually absent.
Ursodeoxycholic acid (UDCA) is the standard treatment for PBC,
as it can improve cholestasis and symptoms,
delay disease progression improving long-term clinical outcomes,
and is well-tolerated.
3) Ig-G4 Cholangiopathy
Immunoglobulin G4 (IgG4) - related disease is a systemic immune-mediated condition characterized by increased serum levels of IgG4.
Histologically there is infiltration by IgG4-positive plasma cells [2].
After the pancreas,
bile ducts are the second most common organ involved in this disease.
About 80% of autoimmune pancreatitis patients suffer stenotic complications of the distal common bile duct.
Occasionally,
IgG4-cholangiopathy is associated with other IgG4-related diseases such as symmetrical dacryoadenitis/sialadenitis and retroperitoneal fibrosis [15] (Figure 9).

Fig. 9: Figure 9. IgG4 pancreatitis with retroperitoneal fibrosis and secondary dilatation of the bile duct. From left to right: CECT arterial phase, CECT portal phase, T2WI.
On MRI the typical features include circumferential wall thickening and delayed contrast enhancement in long segments of the biliary tract,
with prestenotic dilatation.
The location most commonly involved is the intrapancreatic segment of the CBD followed by the hilar bile ducts [1] being less common the involvement of intrahepatic ducts (Figure 10).
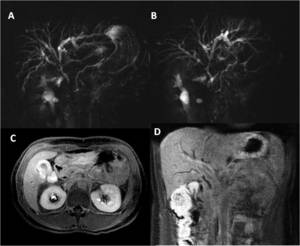
Fig. 10: Figure 10. Ig-G4 related cholangiopathy. (A) Coronal and oblique-coronal (B) thick MRCP showing multiple stenosis of central intrahepatic and extrahepatic bile ducts with proximal dilated ducts. (C) Axial and (D) Coronal T1FS + Gd iv in the delayed phase, showing thickening of gallbladder wall and choledochal dilatation and increased contrast enhancement.
Although there are some imaging features that suggest Ig-G4 cholangiopathy,
it is sometimes difficult to distinguish from PSC or from bile duct malignancy.
It is important to assess the distribution (distal) and type of strictures (smooth wall thickening) and look for pancreatic or retroperitoneal abnormalities as they strongly favor a diagnosis of Ig-G4 cholangiopathy [16,
17] (Figure 11).
It is important to consider this entity as it usually responds to steroid therapy [15].

Fig. 11: Figure 11. IgG4 cholangiopathy with pancreatic disease. (a) STIR , (b) T1FS C+ (arterial), (c) DWI, (d) T1FS C+ (arterial), (e) T1FS C+ (portal), (f) CECT. Note the thickened gallbladder and CBD walls with increased contrast enhancement.
HISORt criteria [18],
including a combination of Histology,
Imaging findings,
Serology,
Other organ involvement and Response to treatment,
first described in the Mayo Clinic,
have been widely used for diagnosing IgG4-related diseases over the last years.
2. SECONDARY CHRONIC CHOLANGIOPATHIES
1) Ischaemic cholangitis
Perfusion of the biliary system depends only on the arterial supply and different etiologies can compromise either the main hepatic arteries or the peribiliary plexus causing ischaemic cholangiopathy.
Most cases are iatrogenic,
including complications of liver transplantation,
hepatic embolization o chemotherapy or any surgery involving the biliary tract [1].
Patients with hereditary hemorrhagic telangiectasia or PAN vasculitis can rarely present this type of cholangitis [4].
Clinical and radiologic features are related to the extent of the ischaemic damage and the stage of the disease.
During the acute phase,
the clinical symptoms can include fever,
abdominal pain,
jaundice and biliary sepsis.
At this time radiology can detect biliary casts or intraductal filling defects and bilomas in the area close to the injured bile ducts [1].
At the chronic stage,
patients have cholestasis and radiological findings are multiple strictures (non-anastomotic,
usually affecting the proximal bile ducts) and dilatations of bile ducts [4] (Figure 12).

Fig. 12: Figure 12. Ischaemic cholangiopathy. (a) Coronal thick-slab T2WI, (b) Axial T2WI, (c) CECT, (d) 3D CECT. 38 years old man with chronic cholestasis after OLT. On MRCP and T2WI there are enlarged intrahepatic bile ducts with stenosis of left and right hepatic ducts, not related to the surgical biliary anastomosis. On the CT image and 3D vascular reconstruction a complete thrombosis of the main hepatic artery is demonstrated.
Courtesy of Dra. García Pérez. Hospital Gregorio Marañón.
Treatment of the underlying condition may halt the progression of the disease.
2) Recurrent Pyogenic Cholangitis
Recurrent pyogenic cholangitis,
formerly called oriental cholangiohepatitis due to its Asian prevalence,
is a progressive biliary disease characterized by recurrent episodes of infectious cholangitis and intrahepatic pigmented stones.
The initial biliary damage is probably secondary to parasitic infestation.
Inflammatory strictures and stones produce recurrent biliary obstruction and cholangitis [1].
Complications include liver abscess,
sepsis,
portal vein thrombosis,
pancreatitis and cirrhosis.
Diagnosis is made by a conjunction of epidemiologic,
clinical and radiological findings.
Diagnostic accuracy of MRCP is superior to that of endoscopic retrograde cholangiopancreatography in this entity [19].
MRCP usually shows intra or extrahepatic bile duct stones (Figure 13), multiple intrahepatic biliary strictures,
dilatation of central bile ducts and rapid sharpening of the proximal extrahepatic bile ducts (“arrowhead appearance”).
Intrahepatic pigmented stones are usually detected as hyperintense on unenhanced fat-suppressed T1WI (Figure 14).
Intrahepatic signs show predilection to the lateral segment of the left hepatic lobe and posterior segments of the right hepatic lobe leading to atrophy of these segments with hypertrophy of the caudate lobe and left medial segment,
creating a round-appearance of the liver [1,
2].

Fig. 13: Figure 13. Recurrent pyogenic cholangitis (oriental cholangiohepatitits). (a) Coronal thick-slab T2WI, (b) Coronal T2WI, T1 fat - sat, (d) Axial T2WI FS, (e) Non-CECT showing intraductal stones, (f) CECT. 39 yo Asian man with recurrent pyogenic cholangitis. Large intrahepatic biliary stones are shown. These pigmented stones are hypointense on T2WI but hyperintense on T1WI and they are intraductally located in dilated central intrahepatic bile ducts.

Fig. 14: Figure 14. Recurrent pyogenic cholangitis. (A) Axial fat suppressed T1WI, (B) axial fat suppressed T2WI, (C) coronal thick-slice MRCP. Intrahepatic bile duct stones are hyperintense in T1 sequences, better seen with fat suppression (A). Note liver parenchymal changes (B) and dilatations of the intrahepatic biliary tree (C) with stenoses in the central ducts.
Treatment usually entails antibiotics and interventional (or surgical) procedures are usually performed to remove bile duct stones in order to avoid bile stasis and prevent recurrence of suppurative cholangitis and stone formation.
3) HIV-related cholangiopathy
HIV-related/AIDS cholangiopathy is a disease found in patients with advanced immunosuppression (CD4 count <100/mm3) and it is far less common now in countries where antiretroviral therapy is standard [20].
Different pathogens have been associated with this condition,
as CMV or Cryptosporidium,
among other [20].
Diagnosis of this condition is made by a conjunction of clinical,
laboratory and radiological features (especially MRCP).
MR cholangiopancreatographic findings include extra and intrahepatic abnormalities: papillary stenosis is present in up to 75% of the patients (causing upper abdominal pain),
often associated to multifocal intrahepatic biliary strictures (similar to those in PSC).
A less common finding is an isolated intermediate to long segment extrahepatic bile duct stricture [6].
Treatment involves restoration of the immune system that may stop or reverse the disease progression.
If the obstruction is persistent,
interventional procedures to relieve the symptoms are usually performed [20].
4) Portal biliopathy
Portal biliopathy is an entity characterized by cholangiographic alterations in patients with portal cavernous transformation.
Portal cavernoma are serpiginous and dilated venous collaterals that appear in the hepatic hilum after a portal vein thrombosis or liver cirrhosis [22].
Mechanical protrusion of the veins on the bile ducts and secondary ischemic injury can lead to significant biliary strictures.
Radiological features include portal thrombosis,
portal venous collaterals (cavernoma) and dilatation of biliary ducts.
These findings can be detected on ultrasound or CT and MRCP offers a better delineation of the biliary tract and vascular anatomy and could help to diagnose the cause of the portal thrombosis [22] (Figure 15).

Fig. 15: Figure 15. Portal biliopathy. (a) Thick-slab T2WI, (b) T2WI FS, (c) T1FS +C (arterial phase), (d) T1FS +C (portal phase, portal thrombosis and portal cavernoma). Portal cavernoma (venous collaterals) are compressing the common, left and right hepatic ducts at the hepatic hilum and causing dilatation of the intrahepatic ducts in both liver lobes. This patient has a spherocytosis and splenectomy with known portal vein thrombosis and developed chronic cholestasis.
5) Mirizzi syndrome
Mirizzi syndrome is caused by stenosis and obstruction of the common hepatic duct caused by extrinsic compression from a gallstone impacted in the gallbladder infundibulum or the cystic duct; sometimes a cholecysto-biliary fistula can occur.
MRI findings include cholelithiasis,
stone in the cystic area,
compression of the CHD with proximal dilation of the biliary tract with normal-caliber distal CHD and CBD [2] (Figure 16).

Fig. 16: Figure 16. Mirizzi syndrome. (a) Coronal thick-slab T2WI, (b) coronal T2WI, (c) T2FS. Please note the large gallstone impacted on the gallbladder infundibulum compressing the common hepatic duct and producing upstream biliary dilatation.
6) Eosinophilic cholangitis
Eosinophilic cholangitis is a very rare disease characterized by eosinophilic infiltration of the biliary tract associating peripheral eosinophilia in 50% of cases,
being the most helpful laboratory finding.
MRI findings include wall thickening of the CBD,
cystic duct and gallbladder with diffuse central bile duct strictures [1].
Patients suffering from eosinophilic cholangitis usually respond to steroid therapy.
7) Chronic cholangitis secondary to prior surgery.
One of the most frequent causes of chronic or recurrent cholangitis is a prior hepatobiliary surgery.
In operated patients,
cholangitis can be secondary to bile duct stenosis or enterobiliary reflux.
It is very important to know the particular surgical procedure as specific complications are related to some types of surgery.
High-quality images of the biliary anastomosis should be obtained,
as most stenosis will occur there.
Defining the exact location and length of the stenosis will be critical to deciding the therapeutic approach.
MRCP with different sequences and imaging planes is usually the best imaging technique to diagnose abnormalities in the operated bile ducts and associated findings as lithiasis (Figure 17).
In some patients,
MRCP can be hampered by the presence of pneumobilia or metallic artifacts and CT or even direct contrast cholangiography can be used to better image bile ducts and the surgical anastomosis.

Fig. 17: Figure 17. Postoperative chronic cholangitis. A patient with prior hepatojejunostomy years ago and chronic cholestasis. Stenosis on the surgical anastomosis is demonstrated on coronal T2 weighted image (A) and MRCP (B) with dilatation of the intrahepatic biliary ducts, and elevated T2 signal patchy areas in liver parenchyma on T2WI (C), reflecting inflammatory changes.