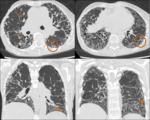
UIP pattern
On HRCT images UIP pattern is defined by these specific features:
- Honeycombing. On CT honeycombing is defined as subpleurally located,
clustered,
thick-walled,
cystic spaces of similar diameters between 3–10 mm (occasionally up to 25 mm).
Usually honeycombing consist of several stacked layers of cysts,
however,
for diagnosis a single subpleural layer of 2-3 contiguous cysts is adequate. Honeycombing is one of the key findings of this pattern as it is an indicator of end-stage pulmonary fibrosis. Traction bronchiectasis,
large cysts,
paraseptal or centrilobular emphysema may complicate the evaluation of honeycombing presence.
Review of sequential multiplanar images as well as post-processing reconstruction algorithms (e.g.
minimum intensity projection) is particularly important in such cases.
- Reticular pattern. A result of thickening of the interlobular septa,
characterized by a network of fine lines.
In UIP,
reticulation is irregularly spaced with a mixture of thick and thin lines.
- Traction bronchiectasis. This feature represents irregular bronchial and bronchiolar dilatation caused by retractile fibrosis in the surrounding lung parenchyma.
Affected airways typically demonstrate an irregular varicose appearance.
This appearance,
along with the background of lung fibrosis represented by reticulation and ground glass opacity,
helps to distinguish traction bronchiectasis from bronchiectasis that is unrelated to fibrosis.
- Ground glass opacity (GGO). Pure ground glass opacity is not usually a feature of UIP.
However,
many patients with fibrotic lung disease have ground glass opacity due to intralobular fibrotic thickening admixed with reticular abnormality and/or traction bronchiectasis.
Usually minimal GGO is seen in areas of reticulation,
but never extensive. The presence of abundant GGO particularly in non-fibrotic areas of lung,
should suggest acute exacerbation.
- Other findings. Mild mediastinal lymph node enlargement is evident on CT in approximately 70% of cases of UIP.
Occasionally,
fine linear or small nodular foci of calcification are observed within areas of fibrosis as a result of ossification.
Some patients with otherwise typical UIP may also have some features of idiopathic pleuropulmonary fibroelastosis,
with bilateral irregular pleuroparenchymal thickening in the upper and mid lungs.
As mentioned,
IPF is defined by UIP pattern (Fig. 2). When IPF is suspected,
the radiologist must indicate whether a UIP pattern is present and evaluate the level of diagnostic confidence. In 2018 the Fleischner Society published the latest IPF diagnostic criteria,
which allow radiologist to classify the CT pattern into one of four diagnostic categories (Fig. 3).
Idiopathic pulmonary fibrosis
Idiopathic pulmonary fibrosis (IPF) is a chronic progressive fibrotic interstitial lung disease of unknown origin that accounts for 50-60% of all idiopathic interstitial pneumonias.
The diagnosis of IPF is a complex procedure that requires integration of clinical,
radiological and histological data (Fig. 4).
The diagnosis can only be made if a radiological/histological typical UIP/probable UIP pattern is found without a known cause or radiological/histological features characteristic for other disease.
IPF has a poor prognosis with the median survival lenght of 2,5 - 3,5 years from the time of diagnosis.
Rheumatoid arthritis
Rheumatoid arthritis (RA) is an autoimmune systemic disorder that primarily affects joints,
but can manifest in many other organs,
most commonly lungs.
Risk group,
clinical,
radiological findings of rheumatoid arthritis are provided in the table (Fig. 5).
Prognosis of RA-ILD is poor: 1-year survival is 68%,
3-year is 46% and lung involvement is the leading cause of mortality.
Patients with UIP pattern predominantly are men,
older age,
seem to be less responsive to conventional treatment and are associated with shortened survival compared with patients without UIP pattern.
Systemic sclerosis
Systemic sclerosis is a chronic autoimmune connective tissue disease that cause microangiopathy,
excessive fibrosis of the skin and various internal organs leading to organ dysfunction.
There are two different types of SSC: diffuse and limited cutaneous sclerosis,
although,
disease can manifest in lungs without skin involvement.
Risk factors,
clinical symptoms laboratoric and radiological findings of SCC are provided in the table (Fig. 6).
Median survival of patients with SSC-ILD is 5-8 years and pulmonary involvement the major cause of mortality (60%).
Hypersensitivity pneumonitis
Hypersensitivity pneumonitis (HP) is a diffuse parenchymal lung disease caused by aberrant immune response to inhalation of a variety of organic dust antigens in susceptible individuals.
HP is divided into three types: acute,
subacute and chronic,
but they may overlap both radiologically and clinically.
Risk group,
clinical symptoms and radiological findings of HP are provided in the table (Fig. 7).
For the diagnosis of CHP,
sensitisation,
proven or suspected exposure associated with exposure-related symptoms,
compatible chest radiography/HRCT abnormalities must be proven.
Differential diagnosis
There are several reasons that makes IPF differentiation from conditions mimicking IPF (“secondary UIP”) challenging:
- For some patients fibrosing lung disease could be the first sign of an immune condition.
- UIP pattern seen in IPF may be often radiologically indistinguishable from the UIP pattern seen in collagen tissue diseases (CTD),
chronic hypersensitive pneumonitis (HP) and others.
Occasionally,
specific coexisting chest CT features in patients with a UIP pattern can help to raise the possibility of an underlying CTD or HP.
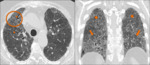
Rheumatoid arthritis (Fig. 8):
- Pleural,
pericardial effusion;
- Coexisting small airway disease (bronchiolitis): fuzzy centrilobular nodules,
ground-glass opacities,
or bronchial and bronchiolar wall thickening with heterogeneous lung attenuation,
air trapping on expiratory CT scanning;
- Rheumatoid nodules;
- Porotic bones;
- Nontypical UIP pattern (e.g.
without honeycombing).
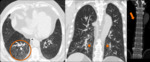
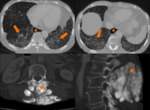
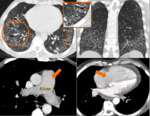
Systemic sclerosis (Fig. 9 ,
Fig. 10,
Fig. 11):
- Pulmonary hypertension;
- Dilated esophagus;
- Calcifications in lungs,
muscles and other soft tissues.
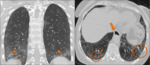
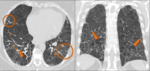
Chronic hypersensitive pneumonitis (Fig. 12,
Fig. 13):
- Ground-glass opacities,
common;
- Mid and upper lung zones commonly affected in a bronchovascular distribution with resulting micronodules;
- Non-basilar distribution common;
- Mosaic attenuation;
- Irregular bronchovascular bundles;
- Subpleural honeycomb cysts,
not always basilar.
In differential diagnosis incorporation of clinical context,
laboratory tests together with additional radiological findings is extremely important.
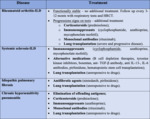
Treatment options
Treatment of UIP depends on the etiology of interstitial lung disease.
Our described conditions with secondary UIP (RA-ILD,
SSC-ILD,
CHP) are usually treated with combination of corticosteroids,
immunosuppressants,
monoclonal antibodies and alternative medications (Fig. 14).
Meanwhile,
treatment of idiopathic UIP (IPF) with the same drugs did not result in good outcomes and for several years antifibrotic agents have been introduced to treatment of IPF.
These drugs have proven to be efficacious and tolerated of patients with severe IPF and had no increased risk after lung transplantation.
Although,
in some cases antifibrotic agents can be included in treatment of secondary UIP,
the best results can be achieved when the primary cause of interstitial lung disease is diagnosed and treated with specific drugs.
Specific,
etiology-based treatment of UIP results in better outcomes,
quality of life,
higher survival rate,
cost effectiveness and therefore the correct diagnosis of UIP cause is vital.