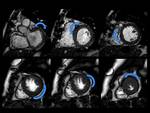
»CMRI TECHNIQUES APPLIED TO HCM
CMRI tecniques let us a new perspective about the frequency,
management,
and prognosis of HCM.
CINE SSFP MR IMAGING SEQUENCES
(Cine Steady-state free precession) are used to evaluate:
-Cardiac function measurement
- Biventricular volume quantification and function
- Elevation of left ventricle ejection fraction(LVEF)
- Sistolic disfunction often develops with end-stage HCM.
- Assessment of global and segmental systolic thickening
-Morphological aspects:
- Quantify the Myocardial thickness and mass accurately which are related to the diagnosis and prognosis of HCM:
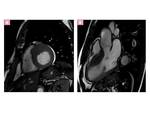
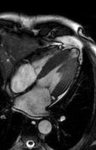
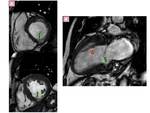
- Diagnostic criterion for HCM is a maximal LV wall thickness ≥ 15 mm in the end-diastolic phase Fig. 1
- Maximum wall thickness ≥30 mm is a risk factor for SCD
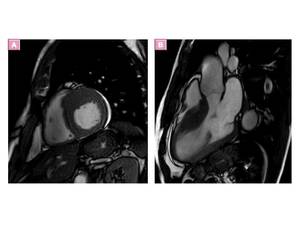
Fig. 1: asymmetric septal wall hypertrophy of anterior and anteroseptal myocardium at the basal level.
- Phenotypes:
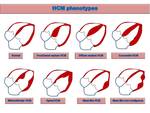
- CMRI no limited view is able to define the broad range of phenotypes of HCM Fig. 2 ,
with asymmetric involvement of the interventricular septum being the most common pattern (60-70% of the cases)
- Apical hypertrophic cardiomyopathy (APH) can be missed by echocardiography,
is easily identified with SSFP
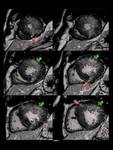
- Evaluation of aneurysms and crypts, anomalíes of Papillary muscle Fig. 22, Fig. 21
- Left atrial size ( and function)
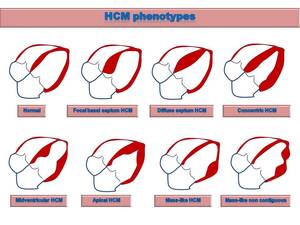
Fig. 2: Drawings of HCM phenotypes.
-Pathophysiological components:
- Contractility anomalies
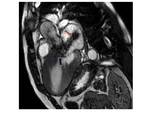
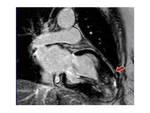
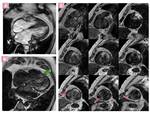
- Left ventricle outflow tract obstruction (LVOT) with turbulence jet across the LVOT in patients with asymmetrical septal hypertrophy (ASH) Fig. 3
- Outflow tract area <2,7cm in systole (100% sensitivity and specificity to differentiate obstructive from nonobstructive HCM)
- Mitral SAM and mitral regurgitation associated to ASH Fig. 3
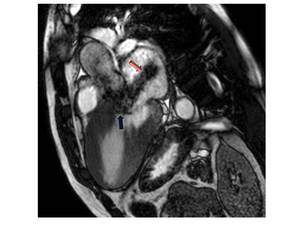
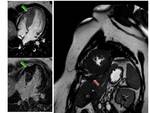
Fig. 3: Three-chamber SSFP image, show the interventricular septal hypertrophy and SAM of the anterior mitral valve leaflet accompanied by a signal-void jet flow into the LVOT(blue arrow). There is also mid-systolic jet of mitral regurgitation (pink arrow) directed to posterior wall of a moderately enlarged left atrium.
FLOW QUANTIFICATION SEQUENCES. (Velocity-encoded cine imaging)
- Direct measurement of outflow tract gradients.
- Quantification of concomitant mitral regurgitation
- Diastolic (dys)-function (mitral inflow/pulmonary vein flow)
LATE GADOLINIUM ENHANCEMENT (LGE)
Is the most valuable MR imaging sequence for HCM, identifies myocardial fibrosis that contributes to risk stratification. It is significantly related to ventricular tachyarrhythmia and SCD associated with HCM
- LGE of ≥15% of an LV mass was associated with a 2-fold increase in SCD event risk (Maron et al)
LGE patterns reflects different types of HCM fibrosis:
- Replacement fibrosis:
-
-
Fibrosis due to increased extracellular matrix commonly seen at the junction of the right ventricle with the LV,
likely due to a different,
nonischemic mechanism.
- LGE pattern of patchy foci at insertion points of RV indicates a bening course Fig. 5
-
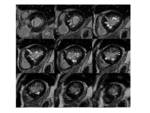
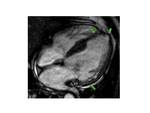
Fine interstitial Fibrosis that is diffusely found in HCM midwall. In our experience probably reflected by the light diffuse and confluent pattern of midwall LGE. Fig. 5 and Fig. 6
- Diferential diagnosis from Phenocopies with different pattern of fibrosis.
- Evaluation of effects of therapy (scar tissue)
- Identificate thrombus formation in left atrial appendage (atrial fibrillation)
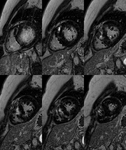
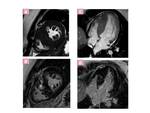
Fig. 4: Asymmetric septal HCM with apical hypertrophy of right ventricle. Short-axis IR images show midwall patchy nodular and confluent areas of intense delayed enhancement in the thickened myocardium.
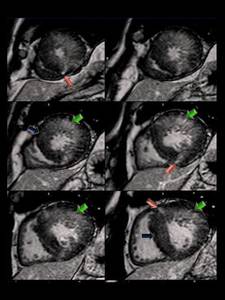
Fig. 5: Concentric HCM. Short-axis LGE images show: intense punctate foci o LGE at RV insertion points (pink arrows); patchy confluent and intense midwall LGE areas (green arrows) and tenuous diffuse midwall LGE areas(blue arrows) in the thickened myocardium ,reflecting conspicuity of LGE due to different types of fibrosis.
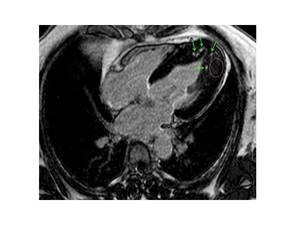
Fig. 6: Apical HCM.-LGE image shows punctate nodular foci of intense LGE (green arrows) and tenuous diffuse LGE (pink elipse) in the thickened apical myocardium.
PERFUSION IMAGING
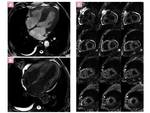
- Evaluation of Microvascular function with infarct-like pattern usually in areas of more hypertrophied and fibrotic myocardium.
Fig. 7
- Microvascular abnormalities precede and predispose to development of myocardial fibrosis; thus,
these perfusion abnormalities may represent an early risk marker (Noureldin et al).
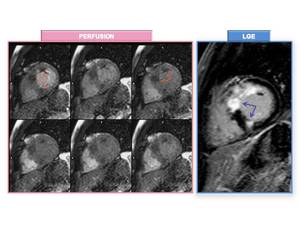
Fig. 7: "PSEUDO INFARCT" PATTERN. Perfusion MR images at rest (left) demonstrate perfusion defects (orange arrows). LGE image shows transmural enhancement(purple arrows) coinciding with perfusion defects and areas of increased myocardial hypertrophy .
T2-WEIGHTED IMAGING/T2 MAPPING
shows myocardial edema or inflammation associated with HCM,
which may be related to chest pain,
syncope,
or increase in troponin.
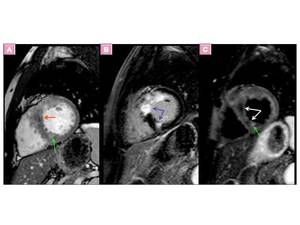
Fig. 8: The same patient of Fig. 8 with ASH. Short axis T2-weighted imaging in combination with double inversion recovery black blood preparation (C) shows myocardial edema or inflammation probably related to ischemia as hyperintensity of te myocardium (green arrow) and hypointense more extensive areas revealing fibrosis (white arrows). Fibrosis and edema/ischemia show identical LGE (B). short axis SSFP MRI (A) shows similar signal pattern than T2 weighted STIR image (C)
TAGGING MRI
The hypertrophied myocardium with LGE tends to show decreased wall motion on tagging.
T1 MAPPING MRI
T1 mapping MR imaging techniques are able to identify myocardial injuries associated with HCM without gadolinium-based contrast agents (Native T1 values).
- Native T1 values and ECV are elevated in HCM and lower post-contrast T1 value (Ying Liu et al).
- Native T1 value and ECV are able to detect more diffuse fibrosis not seen on LGE imaging.
- ECV fraction differs between HCM and Phenocopies ( Swoboda et al).
- ECV is increased in mutation carriers without LV hypertrophy (Ho et al )
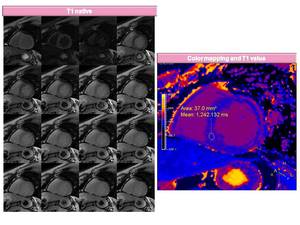
Fig. 9: T1 native and colour T1 Mapping image. Purple áreas reflect myocardial fibrosis. T1 value 1242 ms in a patient with HCM.
RESEARCH TECHNIQUES:
Diffusion imaging
MR spectroscopy
»CARDIAC MRI FINDINGS OF HCM
PHENOTYPES OF HCM:
Diagnostic criteria for HCM is a maximal LV wall thickness ≥ 15 mm in the end-diastolic phase.
The morphologic expression of HCM is widely variable and heterogeneous because HCM may affect any portion of the left ventricle.
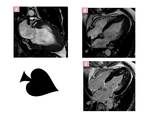
- In most HCM patients the hypertrophied myocardium follows a longitudinal pattern along a spiral trajectory in a counter clockwise direction from the base to the apex of the LV. Fig. 10
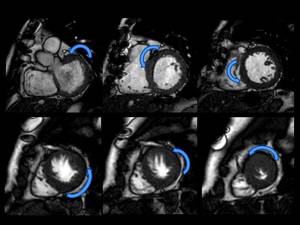
Fig. 10: Spiral pattern of Hypertrophy
ASYMMETRIC (SEPTAL) HCM (ASH)
Is the most common phenotype of HCM (60–70% of HCM cases) with the anteroseptal myocardium most commonly involved.
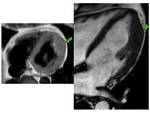
- Reverse-type ASH Fig. 11 hypertrophy of the basal and midventricular septum protruding the left ventricular cavity.
Aassociates genetic mutations and LGE and frequently related to serious ventricular arrhythmia
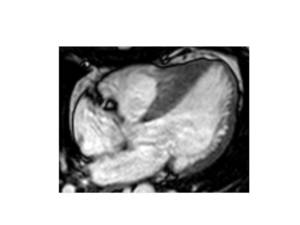
Fig. 11: Four-chamber SSFP image of Reverse curve type HCM.
- Localized hypertrophy of the high septum, the inferior septum or diffuse septum hypertrophy Fig. 12 ,
mass-like and noncontiguous form are other types of ASH.
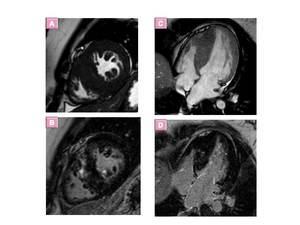
Fig. 12: Asymmetric diffuse septal HCM. images show diffuse hypertrophy and delayed enhancement of the thickened septal myocardium.
APICAL HCM (APH)
Predominantly involves the apical myocardium, giant negative T waves on ECG.
- Cardiac MR imaging identifies this phenotype ussually missed at echocardiography.
- Apical wall thickness > 15 mm or a ratio of apical to basal LV wall thicknesses of 1.3–1.5
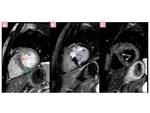
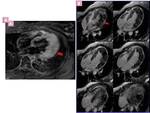
- “spadelike” configuration of the LV cavity at end diastole Fig. 13
- Rarely associated with sudden cardiac death. .
Extensive LGE is related to ventricular tachyarrhythmia even in cases of APH Fig. 13, Fig. 14.
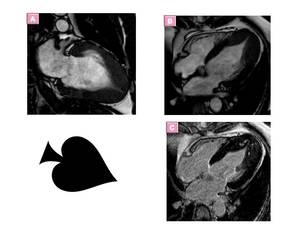
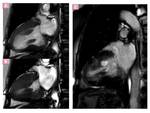
Fig. 13: Apical HCM in a patient with electrocardiographic abnormalities that included QRS voltages associated with LV hypertrophy and giant negative T waves in leads V5 and V6. (A) Two-chamber and (B)Four-chamber SSFP images shows apical hypertrophy and obliteration of the LV apical cavity at end diastole, with a typical spadelike configuration.(C) Four-chamber LGE image with typical mesocardial enhancement.
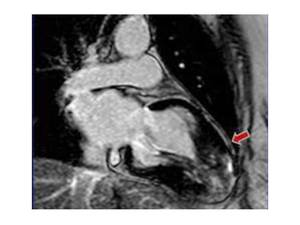
Fig. 14: Patient with Apical HCM and arrhythmic complications. Extensive LGE of the thickened apical myocardium.
MASS-LIKE HCM
Manifests as a focal segmental hypertrophy of the myocardum, MR imaging allows to differentiate it from neoplastic masses.
- Masslike HCM usually shows the homogeneous signal intensity characteristics and perfusion of adjacent normal myocardium,
and typical patterns of LGE Fig. 15 whereas tumors show different pattern from myocardium and heterogeneous LGE Fig. 16.
- Myocardial tagging: absence of active contraction in tumors.
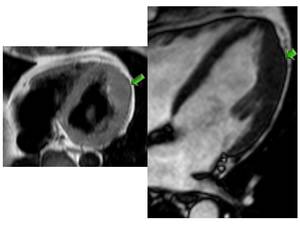
Fig. 15: Masslike HCM axial black blood (A) and four chamber SSFP (B) MR images show a masslike bulging contour at apical lateral wall (arrows).LGE images (not shown) revealed typical enhancement.
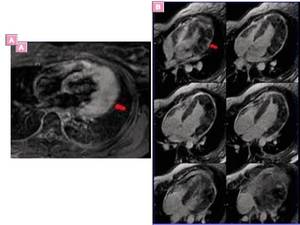
Fig. 16: Cardiac recurrence of B-cell lymphoma.-
Axial black-blood triple IR image (A) and four chamber multislice LGE images show diffuse thickening of the lateral wall of the left ventricle with late heterogeneous enhancement with midwall foci of LGE, but also subepicardial and transmural LGE enhancement, Cine SSFP and tagging images (not shown) revealed contractility anomalies of infiltrating process.
SYMMETRIC HCM(Concentric HCM)
Is the second most common phenotype and is characterized by diffuse concentric thickening of the LV with an associated decrease in the size of the LV cavity and no evidence of a secondary cause.
- 42% of the cases of HCM.
- Cardiac MR imaging plays an important role in differentiating other causes of symmetric myocardial hypertrophy (see below in section phenocopies )
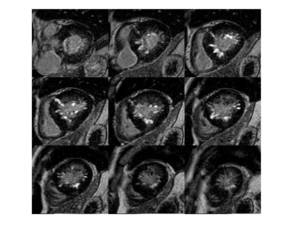
Fig. 17: Symmetric or Concentric HCM. LGE images show extensive patchy confluent and intense midwall next to subendocardium LGE areas and tenuous diffuse midwall LGE areas in the thickened myocardium, reflecting double pattern of LGE.
MIDVENTRICULAR HCM
Rare variant of asymmetric HCM.
Hypertrophy occurring predominantly in the middle third of the LV wall and by systolic apposition of the mid-ventricular wall causing Midventricular obstruction (MVO).
May be associated with an apical aneurysm.
Associates with ventricular arrhythmia,
myocardial necrosis,
systemic embolism.
MR imaging shows:
- “Dumbbell” configuration of the LV cavity with marked muscular midcavity systolic constriction .
- jet flow and midventricular hypertrophy in MVO.
- apical aneurysm, apical thrombus significantly related to adverse cardiac events.
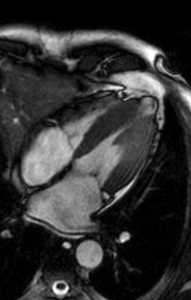
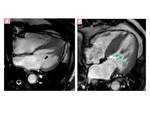
Fig. 18: Midventricular HCM.- four-chamber SSFP cine MR image shows midventricular hypertrophy and apical aneurysm.
RIGHT VENTRICLE (RV) INVOLVEMENT
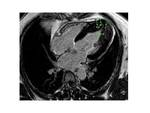
Predominantly affects the mid to apical portion Fig. 19 .
CMRI is the most valuable imaging technique to delianete RV hypertrophy because echocardiography is limited for RV evaluation
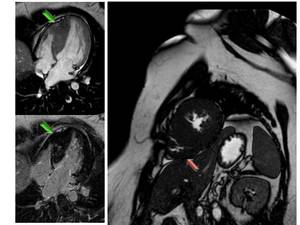
Fig. 19: Asymmetric diffuse septal HCM with right ventricle involvement. SSFP(Up left) and inversion-recovery LGE (down left and right) MR images, show diffuse hypertrophy of the whole interventricular septum and associated apical RV hypertrophy (green arrows).
PAPILLARY MUSCLES ANOMALIES,
MUSCLE BUNDLES AND CRYPTS
Papillary muscle anomalies,
apical –basal muscle bundles and crypts Fig. 22 are commonly present in HCM patients and in G+/P- family members,
may represent an additional morphologic marker for HCM diagnosis in genotype-positive status.
Papillary muscles (PM) abnormality include:
- Hypertrophy ,
increase in the number Fig. 21 ,
abnormal attachment of the muscles.
- LGE may be observed in hypertrophied papillary muscles Fig. 20 .
- Isolated PM hypertrophy is a variant of HC (Correia et al)
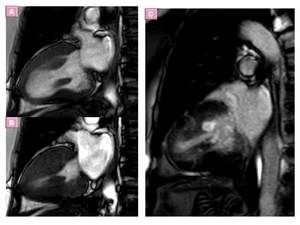
Fig. 20: Midventricular HCM and hyperthopy of papillary muscles.- two chamber SSFP cine MR images (a) Diastole and (B) systole show midventricular hypertrophy and papillary muscles hypertrophy. LGE image (C) shows intense enhancement of papillary muscle.
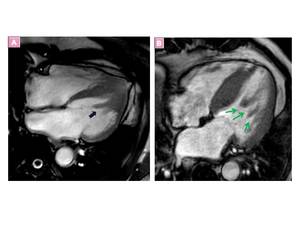
Fig. 21: A, Bifid papillary muscle and B, An increase in papillary muscles is associated with hypertrophic cardiomyopathy(arrows)
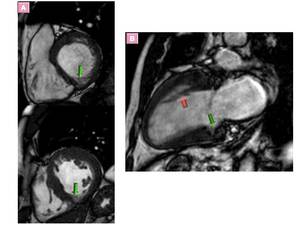
Fig. 22: Myocardial crypts are found at the basal and middle inferior region in end-diastole (arrow) (A). Another patient with HCM presented inferior basal myocardial crypt (green arrow) and basal to apical myocardial band (pink arrow)
»CLINICAL MANIFESTATIONS,
COMPLICATIONS, AND NATURAL HISTORY
Phenotypic expression of HCM generally occurs in adolescents.
There is an increasing population of asymptomatic genotype-positive but phenotype-negative family members,
in whom long-term monitoring with CMRI is very relevant.
It is important to make a distinction between phenotype-negative HCM and “NONHYPERTROPHIC” HCM,
displaying:
- Electrocardiographic abnormalities
- Subtle functional morphologic and functional abnormalities at echocardiography or cardiac MR
- Without LV hypertrophy.
In most patients,
a lifelong process of progressive cardiac remodeling with increasing myocardial fibrosis takes place.
Cardiac remodeling with overt NYHA class III - IV heart failure occurs in a 2%–5% of patients (obstructive phenotype or restrictive phenotype)
DIASTOLIC DYSFUNCTION AND PULMONARY HYPERTENSION
Impaired LV diastolic function is a major pathophysiologic feature in all patients with HCM.and can be detected in genotype-positive individuals before significant hypertrophy or clinical symptoms appear.
- Cardiac MR imaging is increasingly being used to assess diastolic function by means of velocity encoding and spatial modulation of magnetization(SPAMM) sequences .-
- Independent of hypertension or the presence of coronary artery disease Left ventricular hypertrophy is the most common structural abnormality in patients with with heart failure with preserved ejection fraction (Webb et al).
- CMR allows accurate assessment of the right ventricle function and volumes, pulmonary artery flow and diameters.
SYSTOLIC DYSFUNCTION,
END-STAGE HCM
HCM usually shows normal or supernormal systolic function,
LVEF up to > 75%.
15% of patients LVEF 50%–65% indicating adverse remodelling and 5‒10% of patients show LVEF< 50%,
which is called “end-stage” or “burn-out” HCM.
Indicates a poor prognosis in the 5 years at its initial diagnosis because of heart failure and thromboembolic complications but also from a substantial incidence of SCD.
Cardiac MR imaging is able to measure the ejection fraction accurately and demenstrate de differnt patterns of systolic dysfunction:
- Adverse remodelling:Fig. 23
- LVEF 50%–65%
- Myocardial hypertrophy is evident but progressive thinning may occur.
- Loss of prior LV obstruction
- LGE pattern Midwall/ transmural
- Moderate/severe left atrium dilatation
- Microvascular dysfunction: moderate to severe
- Apical aneurysm
- End-stage HCM: Fig. 24
-
- LVEF< 50%
- Midwall/ transmural “infarct”-like LGE
- “Dilated – hypokinetic” form:
-
- May mimic dilated cardiomyopathy
- absent LV obstruction
- LV wall thinning
- Severe microvascular dysfunction
- May mimic dilated cardiomyopathy
- “Restrictive – hypokinetic” form:
-
- May mimic restrictive cardiomyopathy
- Small LV
- Myocardial hypertrophy is evident but progressive thinning may occur.
- Severe biatrial dilatation
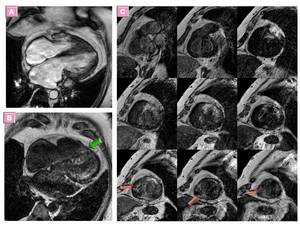
Fig. 23: Adverse remodelling.- 54-year-old woman with LVEF:60% who subsequently underwent cardiac transplantation.
Four chamber SSFP CMR image shows hypertrophy from mid to apical septum of LV and RV apex. Dilated left and right atrium (patent foramen ovale). Four camber (B) and multislice short axis (C) LGE images reveal extensive midwall but also transmural LGE indicating extensive fibrosis and resembling amiloidosis but nulling signal pattern type 2 or 3 (Pandey et all) was not present. histopathological analysis of the cardiac explant showed extensive replacement fibrosis and consistent findings of hypertrophic cardiomyopathy
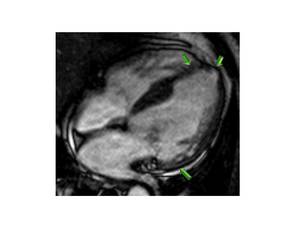
Fig. 24: Burned-out phase of HCM.-Four chamber SSFP CMR image shows hypertrophy (basal septum), coexisting with thinning of LV myocardium in a patient witn systolic dysfunction, LVEF<50%.
»CLINICAL RELEVANCE OF CMRI TO HCM
DIFERENTIAL DIAGNOSIS - PHENOCOPIES
HCM phenocopies is refered to several genetic conditions that are not caused by cardiac sarcomere mutations, associated with severe LVH.
include a variety of disorders such as glycogen storage disorders,
lysosomal storage disorders (Anderson-Fabry Disease Fig. 28 ),
mitochondrial cytopathies,
cardiac amyloidosis Fig. 27 and disorders of fatty acid metabolism. LVH is also observed in,
hypertensive cardiomyopathy,
aortic valvular disease and Athlete’s heart (SCD can occur in young athletes with HCM).
Cardiac MR imaging is valuable for differentiating between HCM and other myocardial diseases showing LVH Fig. 25 because of its accurate identification of:
- The patterns of myocardial hypertrophy and LGE.
- basal crypt,
accessory apical-basal muscle bundle and apical pouching which can suggeste the diagnosis of HCM
- T1 native and postcontrast ECV values elevated in HCM Fig. 26
Endomyocardial fibrosis Fig. 29,
LV noncompaction and cardiac tumors Fig. 16 are other rare HCM phenocopies.
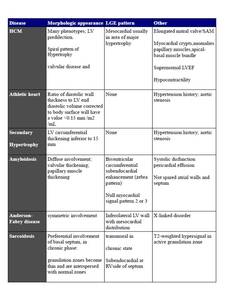
Fig. 25: Table1.-Differentiating HCM MR imaging Features from Those of Phenocopies

Fig. 26: Table 2.-T1 mapping of HCM and phenocopies
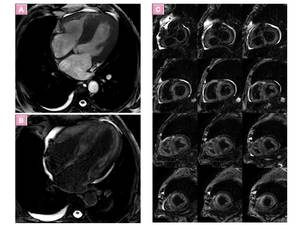
Fig. 27: Cardiac amyloidosis. Four chamber SSFP and LGE and short axis multislice LGE MR image 4-5 minutes after contrast administration (optimal delay time for amyloidosis)show concentric LV hypertrophy.
Endocardial and transmural diffuse enhancement is seen in both
ventricles and atriums and dark signal of blood pool is also evident.

Fig. 28: Fabry's disease–related hypertrophy. Concentric myocardial hypertrophy, more evident at lateral wall of LV (right). Typical pattern of delayed enhancement: thick striae involving inferolateral wall of basal segment of left ventricle in mesocardial distribution. Sparing of subendocardial layer (arrows).
Read More: https://www.ajronline.org/doi/full/10.2214/ajr.08.1201
Fig. 29: Endomyocardial disease in a 65-year-old woman who received chemo and radiotherapy treatment for breast cancer. MR images show thickening and deformation of LV apex with cavity obliteration. Four chamber LGE left show subendocardial enhancement due to subendocardial fibrotic tissue (arrow) and resolution of apical thrombus seen in previous CMRI study (blue arrows)
RISK STRATIFICATION
| Major risk markers and risk modifiers associated with increased risk of sudden cardiac death (SCD) in hypertrophic cardiomyopathy |
| Major risk factors |
| Family history of sudden cardiac death |
| Nonsustained ventricular tachycardia |
| Syncope |
| Maximal left ventricular wall thickness |
| LV apical aneurysm |
End-stage HCM
(LVEF <50 percent) |
| Risk Modifiers |
| Marked LV outflow tract obstruction |
| Late gadolinium enhancement by CMR |
| Age |
| Abnormal blood pressure response to exercise |
Cardiac MR imaging is valuable because of its ability to depict accuretly:
- LVEF<50%
- Myocardial thickness and myocardial mass. Massive hypertrophy and maximum wall thickness (wall thicness>30mm Fig. 30
- Identify myocardial fibrosis noninvasively and cuantify LGE enhanacement porcentage.
Extensive LGE can indicate risks for the ventricular tachyarrhythmia or systolic impairment.
-
- LGE of ≥15% of LV mas Fig.
31 is associated with a 2-fold increase in SCD event risk.
the extent of LGE is strongly associated with the risk of SCD,
thus,
the extent of LGE may be a more potent marker than its presence alone (Kramer et al)
- Maron et al:"LGE represents a novel powerful clinical tool that dictates HCM clinical course along 2 divergent adverse pathways: risk for sudden death and for advanced end-stage heart failure.
For these and other reasons,
it would seem justified to regard CMR as firmly integrated in the contemporary assessment of virtually all HCM patients".
- Evaluate risk phenotypes:
-
- Midventricular obstruction,
that is is a “high risk” type and apical aneurysm,
possibly leading to ventricular tachyarrhythmia or systemic thrombosis.
- advanced fibrotic remodeling of the LV and end-stage HCM,
LVEF<50%.
- LV outflow tract obstruction: basal gradient ≥ 30 mm Hg or of ≥50 mm Hg with exercise
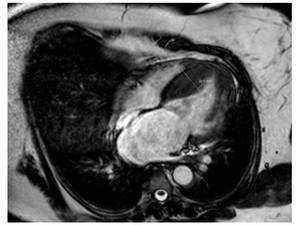
Fig. 30: Diffuse septal hypertrophy with a risk factor of SCD, septal thickness>30mm