Type:
Educational Exhibit
Keywords:
Infection, Acute, Abscess, Diagnostic procedure, Comparative studies, Abscess delineation, Ultrasound, Digital radiography, CT, Liver, Kidney, Biliary Tract / Gallbladder
Authors:
G. Carbo1, L. Valls Masot1, G. Sanchez1, V. Garriga2, V. Cheranovskiy3, A. Gimeno4, C. V. Nadal1, A. Maroto1, S. Pedraza1; 1Girona/ES, 2Santa Maria de Palautordera/ES, 3Sabadell, Barcelona/ES, 4Barcelona, Barcelona/ES
DOI:
10.26044/ecr2019/C-2334
Findings and procedure details
In general,
diabetic patients have an increased risk for gas-forming abdominal infections.
Abdomino-pelvic emphysematous pathologies share similar imaging characteristics,
as they manifest as acute necrotizing gas-forming infections due to involved bacteria,
most often Escerichia coli although it is also associated with Klebsiella pneumoniae,
Enterobacter aerogenes,
Pseudomonas sp,
Proteus Mirabilis and fungi.
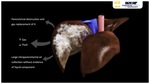
- EMPHYSEMATOUS HEPATITIS
The term emphysematous hepatitis (EH) was first described by Blachar et al (1) and the postmortem studies of 2 patients by López Zárraga et al (2).
Imaging characteristics and a fulminant clincal onset were key to diagnose these cases.
EH is a very rare entity described only 4 times in the literature over the last 50 years.
In contrast with other affected organs,
such as emphysematous pyelonephritis or cholecystitis,
emphysematous changes in the liver are extremely rare.
Most of the patients suffering from this disease were diabetic (> 90%) and all of them died within 8-72 hours of admission to the hospital,
hens this affection is ominous if left untreated (3).
The pathogenesis of EH remains unclear.
However,
an hyperglycemic state together with the presence of a thrombotic event leading to an ischemic environment subserves the proliferation of this type of gas-forming bacteria,
that in turn alters the bowel wall,
permitting intraluminal gas to enter the portal venous system reaching the liver parenchyma.(3)
There is no definite treatment guideline for EH.
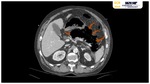
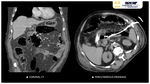
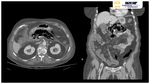
CT imaging characteristics of EH Fig. 1 Fig. 2 Fig. 3 closely resemble those seen in emphysematous pyelonephritis: parenchymal destruction and gas replacement of it.
The gas collection adopts a globular morphology characteristically in the absence of fluid component/pus.
The fundamental difference between pyogenic liver infections and EH is the fluid/gas ratio.
Hepatic abscesses are usually spherical/oval pus collections,
septate or not,
which may be encapsulated and exert mass effect to adjacent structures.
Few pyogenic liver abscesses contain gas and,
if present,
it will form a gas-fluid level or present small air bubbles.
Emphysematous hepatitis characteristically presents a large intraparenchymal air collection without evidence of liquid component. Fig. 3
Hepatic infarction should be taken into account in the differential diagnosis as it can also demonstrate hepatic gas collections.
The keypoint is the compromise of arterial and portal flow in an infarct in the abscence of concomitant infection.
Typically,
infarction cases appear in liver transplantation or trauma patients.
- PYLEPHLEBITIS AND GAS-FORMING HEPATIC ABSCESSES
Pylephlebitis, or ascending septic portal thrombophlebitis,
is a thrombotic occlusion of the portal vein secondary to intra-abdominal infections whose venous drainage depends on the portal system (diverticulitis,
appendicitis,
hypercoagulative status,
hepatobiliary diseases,...) Fig. 4 Fig. 5.
Hepatic abscesses are localized collections of necroinflammatory tissue caused by bacterial,
parasitic or fungal agents.
Few pyogenic liver abscesses contain gas (∼20 %)(4) and,
if present,
it will form a gas-fluid level or present small air bubbles.
As mentioned above,
the fundamental difference between pyogenic liver infections and emphysematous hepatitis (EH) is the fluid/gas ratio.
EH characteristically presents a large intraparenchymal air collection without evidence of liquid component Fig. 3.
Hepatic abscesses are usually spherical/oval pus collections,
septate or not,
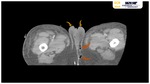
which may be encapsulated and exert mass effect to adjacent structures Fig. 4.
Simple X-ray is not sensitive for evaluating liver abscesses.
An indirect sign is depiction of gas within the liver silhouette representing the abscess,
biliary tree gas (pneumobilia) or gas beneath the diaphragm.
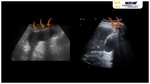
The sonographic features of liver abscesses is typically ill-definied collections with a variable appearance,
ranging from predominantly hypoechoic (with some internal echoes) to hyperechoic.
Gas bubbles may also be seen as "dirty" shadowing or "ring-down" artifacts (5) inside the collection.
Contrast-enhanced US shows wall enhancement during arterial phase and progressive washout during portal or late phases.
The liquefied necrotic area does not enhance.
The use of contrast allows to characterize the lesion,
to measure the necrotic area,
and to depict internal septations for management purposes.
In small abscesses (< 3 cm) and in highly septated abscesses,
drainage is not recommended.
** Be careful: in patients with monomicrobial K.
pneumoniae abscesses,
the lesion may appear solid and mimic an hepatic tumor (5).
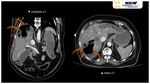
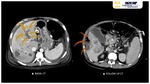
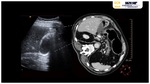
On contrast-enhanced CT they often appear as peripherally enhancing and centrally hypoattenuating collections (6).
Occasionally they contain gas (~20%)(4) visualized as bubbles or air-fluid levels(7).
Hepatic abscesses are characteristicaly low attenuation lesions (fluid-filled) surrounded by a high attenuation inner rim (abscess capsule/membrane) and a low attenuation outer ring (parenchymal oedema) Fig. 4 Fig. 5.
** Be careful: capsular enhancement may be absent in immunocompromised patients (8).
On MRI hepatic abscesses have similar characteristics as commented on CT imaging.
Abscesses content tends to restrict on diffusion studies (high DWI/ low ADC).
-
EMPHYSEMATOUS CHOLECYSTITIS
Emphysematous cholecystitis (EC) is a rare form of acute cholecystitis where gallbladder wall necrosis causes gas formation in the lumen or wall. It is a surgical emergency,
due to the high mortality from gallbladder gangrene and perforation.
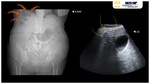
On simple X-ray imaging Fig. 6,
the condition is diagnosed when there is radiographic demonstration of air in the gallbladder wall +/- biliary ducts,
in the absence of an abnormal communication with the gastrointestinal tract (9).
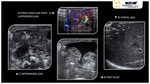
US Fig. 9 Fig. 10 may demonstrate highly echogenic reflectors with low-level posterior shadowing and reverberation artifact ("dirty" shadowing and "ring-down" artifact) (10).
A less common but more specific finding is small,
non-shadowing echogenic foci rising up from the dependent portions of the gallbladder lumen,
similar to effervescing bubbles in a glass of champagne (champagne sign).
CT Fig. 7 Fig. 8 Fig. 10 is considered the most sensitive and specific imaging modality for identifying gas within the gallbladder lumen or wall (11). The presence of pneumoperitoneum indicates perforation.
Because there is usually cystic duct obstruction,
gas is present in the bile ducts in only 20% of cases.
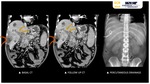
- EMPHYSEMATOUS PANCREATITIS
Emphysematous pancreatitis (EP) is an unusual complication of acute pancreatitis caused by necrotising infection of the pancreas and is a potentially life-threatening condition.
It is associated with gas-forming bacteria and characterized by the presence of gas within or around the pancreas.
EP fits into the necrotising pancreatitis category and corroborates to the highest score in the CT severity index (CTSI).
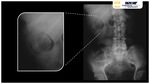
Conventional abdominal radiography may demonstrate mottled gas overlying the epigastric region (not an specific finding).
Computed tomography is the imaging modality of choice because of its sensitivity and specificity in detecting gas bubbles.
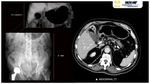
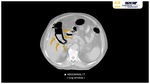
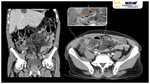
Imaging findings are: focal or diffuse parenchymal enlargement with lack of enhancement due to liquefactive necrosis associated to the presence of gas (infected necrosis). Fig. 11 Fig. 12 Fig. 13
Also,
CT findings associated with pancreatic/peripancreatic inflammation must be seen: changes in peripancreatic tissues density because of oedema associated to surrounding retroperitoneal fat stranding; indistinct pancreatic margins owing to inflammation; intraabdominal free fluid; circumscribed fluid collection and abscess formation (it can also be infected and filled with air-bubbles).
The main role of ultrasound is:
- to identify gallstones as a possible cause
- to diagnose vascular complications (e.g.: portal thrombosis)
- to identify areas of infected (air-filled) necrosis which appear as multiple heterogeneous high echogenic foci,
often with posterior dirty acoustic shadowing (5).
- EMPHYSEMATOUS GASTRITIS
The stomach is one of the less common viscera affected by gas-forming infections (12).
There are no predilections with regard to age,
sex or diabetic status (12-13).
Predisposing conditions include caustic ingestion and alcohol abuse,
and infrequently due to recent gastroduodenal surgery,
trauma or gastric infarction.
Characteristic findings may be seen at conventional radiography within the first few days of illness and may persist up to 4 weeks.
Innumerable bubbles are seen to outline the stomach in a mottled distribution. Fig. 14
CT is considered the modality of choice for detection of intramural gas and evaluation for the presence of pneumoperitoneum or portal venous gas. CT may also demonstrate irregular mucosal fold thickening and may be used to monitor response to treatment or disease progression.
An important differential diagnosis to consider is benign gastric emphysema.
Gas collections form within the gastric wall without associated infection by gas-forming organisms(12).
Gas may enter the wall from the lumen,
peritoneal surface,
or esophageal or duodenal connection and is usually associated with violent coughing,
vomiting,
or severe obstructive pulmonary disease.
Gastric fold inflammation and thickening are not present,
and the patient is usually asymptomatic with spontaneous resolution expected.
The characteristic radiographic features of benign gastric emphysema include thin,
linear streaks of air running parallel to the border of the stomach that do not change with patient position(12).
CT will help confirm this linear distribution of mural air and,
when performed with enteric contrast material,
will demonstrate a normal gastric mucosal thickness of 3 mm or less(14).
- EMPHYSEMATOUS PYELONEPHRITIS / PYELITIS
Emphysematous pyelitis (EP) is isolated gas limited to the renal excretory system,
secondary to acute bacterial infection.
It has women predilection and is often associated with underlying diabetes or obstructing stone disease.
It carries a mortality rate of up to 20% (15),
which is significantly lower than that of emphysematous pyelonephritis (EPN) (mortality rate ∼ 50 %).
In the latest,
gas production from an infection occurs in the renal parenchyma rather than just in the collecting system.
Underlying poorly controlled diabetes is present in up to 90% of patients who develop EPN (14).
It is more common in females.
Other etiologies include urinary collecting system obstruction,
urothelial neoplasm or sloughed papilla.
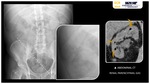
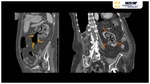
On simple X-ray mottled gas within renal fossa can be seen Fig. 15,
diffusely mottled kidney with radially oriented gas corresponding to the renal pyramids or crescentic gas collection within Gerota's fascia. Linear gas shadows along paraspinal region may also be seen,
representing retroperitoneal air.
Ultrasound may show an enlarged kidney with coarse echoes within the renal parenchyma or collecting system.
Dirty echogenic foci with reverberation/ring-down artifacts representing air ('dirty shadowing') may also be seen.
Bowel gas over kidneys may lead to false positive diagnosis.
CT is the best diagnostic tool for emphysematous pyelitis and emphysematous pyelonephritis.
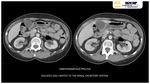
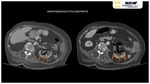
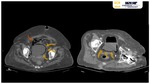
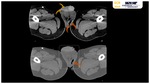
It may confirm the presence and extent of parenchymal gas in the EPN Fig. 17 or in the excretory system in the EP Fig. 16 Fig. 18.
It will often allow identification of the source of obstruction when present.
The use of intravenous contrast material will often reveal asymmetric renal enhancement or delayed excretion,
and,
during the nephrographic phase,
will help identifying areas of focal tissue necrosis or abscess formation.
A CT classification scheme proposed by Wan et al.
(16) divides emphysematous pyelonephritis into two types encompassing different prognostic significance.
Type I EPN is characterized by parenchymal destruction (greater than one-third) with streaky or mottled gas collections but no fluid collections.
Mortality: 70 %.
Type II EPN is characterized by bubbly or loculated gas within the parenchyma or collecting system with associated renal or perirenal fluid collections that are thought to represent a favorable immune response.
Mortality: 20 %.
In mild cases,
intravenous antibiotics are successfully administered,
and percutaneous catheter drainage of perirenal or retroperitoneal collections can be required. Severe cases may warrant nephrectomy.
- EMPHYSEMATOUS CYSTITIS
Emphysematous cystitis (EC) refers to gas-forming infection of the bladder wall.
Risk factors include diabetes (common predisposing factor,
present in aprox.
50 % of cases)(17); female; immunocompromised state; neurogenic bladder and transplant receptors.
Possible noninfectious sources of pelvic air should be considered including recent bladder instrumentation,
vesicocolic or vesicovaginal fistulas,
trauma,
and pneumatosis cystoides intestinalis(14).
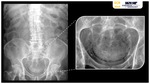
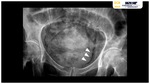
Conventional radiography characteristically shows curvilinear or mottled areas of increased radiolucency in hypogastric region,
separate from more posterior rectal gas Fig. 19 Fig. 20.
Intraluminal gas will be seen as an air-fluid level that changes with patient position,
and,
when adjacent to the nondependent mucosal surface,
may have a cobblestone or “beaded necklace” appearance.
This is thought to reflect the irregular thickening produced by submucosal blebs as seen at direct cystoscopy.
Ultrasound (US) can demonstrate focal echogenic air within the bladder wall with dirty shadowing artifact.
Intraluminal air may be seen in extensive cases. US will also commonly demonstrate diffuse bladder wall thickening and increased focal echogenicity foci due to mural air replacement.
CT is a highly sensitive ex-amination that allows early detection of intraluminal or intramural gas(18) Fig. 21.
It is also useful in evaluating other causes of intraluminal gas such as enteric fistula formation from adjacent bowel carcinoma or inflammatory disease.
- GANGRENOUS or EMPHYSEMATOUS APPENDICITIS
Acute appendicitis is typically a disease of children and young adults with a peak incidence in the 2-3rd decades of life.
Gangrenous appendicitis is usually the sequel of lumen obstruction, often by a faecolith (19).
The continuum of disease starts with obstruction,
progresses to suppurative inflammation and gangrene,
and ends with appendiceal necrosis and perforation (20).
In many publications,
these patients were placed in the category of “complicated appendicitis” and treated identically to those with frank perforations (20).
Ultrasound findings supporting a gangrenous or emphysematous appendicitis include typical US findings of acute appendicitis: aperistaltic,
non-compressible,
dilated appendix (> 6 mm) associated with focal or diffuse dirty echogenic foci within the appendiceal wall with reverberation/ring-down artifacts representing air ('dirty shadowing').
Other US findings are echogenic prominent pericaecal and periappendiceal fat; free fluid or periappendiceal fluid collection; periappendiceal or pericaecal adenopathy; appendicolith.
CT is highly sensitive (94-98 %) and specific (up to 97 %) for the diagnosis of acute appendicitis and allows for alternative causes of abdominal pain when appendicitis is ruled out (21). The presence of pneumoperitoneum indicates perforation. CT may also demonstrate irregular mucosal fold thickening and will help to confirm the distribution of appendiceal mural air Fig. 22. The use of intravenous contrast material will often reveal a lack of mural enhancement due to necrosis.
-
NECROTIZING ENTEROCOLITIS
Necrotizing enterocolitis (NEC) is the most common gastrointestinal condition in premature neonates.
It is characterised by inflammation,
ischaemia,
and permeability of the neonatal bowel wall to bacteria. It is potentially life-threatening with significant associated morbidity (22).
Risk factors include: prematurity (50-80 %)(23); congenital heart disease; perinatal asphyxia; decreased umbilical flow in utero.
NEC is usually idiopathic and multi-factorial. Inflammation starts from the mucosal surface and progresses to haemorrhagic and coagulative necrosis.
There is ensuing loss of mucosal integrity,
transmural necrosis and perforation.
Although NEC can affect any part of the large or small bowel,
the most common location is the terminal ileum.
The right colon (including caecum) is occasionally involved.
Supine abdominal x-rays are the mainstay of diagnosis,
potentially showing dilated bowel loops,
loss of the normal polygonal gas shape,
bowel wall oedema with thumbprinting,
pneumatosis intestinalis,
portal venous gas and pneumoperitoneum.
Ultrasound may include the following findings Fig. 23:
- Bowel wall thickening
- Alteration of the vascular state with two different wall enhancement patterns:
› Hypervascular (viable wall but engorged in early stage)
› Hypovascular (infarcted wall in a later stage)
- Intramural gas manifesting as hyperechoic foci within the bowel wall
- Free fluid,
especially with echogenic debris,
suggests perforation
- FOURNIER GANGRENE
Fournier gangrene is a necrotizing fasciitis of the perineum typically seen in diabetic men aged 50-70 years but rarely occurs in women Fig. 25.
Other than age,
predisposing factors include diabetes,
alcoholism,
immunosuppression or debility.
Diagnosis is usually clinical.
The role of imaging is not only establishing the diagnosis but to determine the extent of disease and determine the underlying cause.
Radiography characteristically reveals marked swelling of the scrotal tissues and areas of hyperlucency representing interstitial air.
Subcutaneous emphysema may extend laterally to the inguinal canal,
superiorly to involve the abdominal wall or inferiorly towards the thighs.
Ultrasound may show thickening of scrotal wall due to oedema,
echogenic gas foci of dirty shadowing in the scrotum with or without reactive extratesticular fluid. US is also helpful in excluding other causes of pneumoscrotum seen at radiography such as an incarcerated inguinoscrotal hernia (24).
Although CT Fig. 24 Fig. 25 can help confirm Fournier gangrene,
it is best used to evaluate the extent of disease,
detect a potential underlying cause,
and follow up therapeutic response.
Findings of fat stranding and asymmetric fascial thickening correspond well to diseased tissue at surgical debridement,
and deep pelvic or abdominal fluid collections are reliably detected with CT (25).