Keywords:
Liver, Abdomen, Ultrasound, Elastography, Comparative studies, Biopsy, Cirrhosis
Authors:
W. L. Leong1, L. L. Lai1, N. R. Nik Mustapha2, A. Vijayananthan1, S. Mahadeva1, K. Rahmat1, W. K. Chan1; 1Kuala Lumpur/MY, 2Alor Setar/MY
DOI:
10.26044/ecr2019/C-2416
Methods and materials
Patient population: Consecutive adult patients with NAFLD scheduled for a liver biopsy in University of Malaya Medical Centre were included for this study.
We excluded patients with significant alcohol intake,
medication usage that can cause fatty liver,
viral hepatitis B and C infection and other causes of chronic liver diseases.
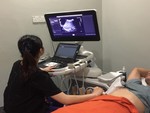
pSWE: pSWE was performed using Phillips EPIQ 7 ultrasound system with a C5-1 convex probe.
Patient is placed in supine position with right arm in maximal abduction and probe placed on right lobe of liver through the right intercostal spaces.
Scanning and measurement was done during breath-hold to minimize breathing artefacts.
The mean value of three shearwave measurement was taken.
[4] pSWE was performed four times by two different operators independently for the same patient.
Figure 1 shows an image of an operator performing pSWE.
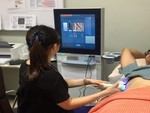
TE: TE performed using FibroScan; Echosens,
Paris,
France.
TE was performed with patient in supine position and right arm in full abduction,
with adequate pressure of probe on skin surface.
An examination was considered successful if ≥ 10 valid measurements obtained,
and reliable if IQR/med ≤30,
or IQR/med>30 and LSM<7.1kPA.
[6] Figure 2 shows an image of an operator performing TE.
Liver biopsy and histopathological examination: All patients underwent ultrasound guided liver which was performed by one of two operators using 18G Temno ® II semi-automatic biopsy needle (Cardinal Health,
Dublin,
Ohio,
USA).
Liver biopsy specimens were processed using standard laboratory procedures,
stained with hematoxylin and eosin stain & masson trichrome stain,
finally examined by an experienced histopathologist who was blinded to clinical data.
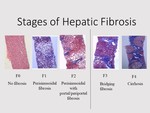
Histopathological examination was reported according to the NASH Clinical Research Network scoring system.
[7] Figure 3 demonstrates the histopathological slides in each fibrosis stage according to NASH Clinical Research Network Scoring System.
Statistical analysis: Data was analysed using standard statistical software program (SPSS 21.0,
SPSS Inc.,
Chicago,
IL,
USA).
Diagnostic accuracy was measured using areas under receiver operating curves (AUROC) and interpreted as follows: 0.90-1.00 = excellent,
0.80-0.90 = good,
0.70-0.80 = fair,
<0.70 = poor.
R Statistical Software (R 3.4.3,
R Foundation for Statistical Computing,
Vienna,
Austria)was used to determine the 95% confidence interval for AUROCs and pairwise comparison of AUROC.