Cases of acute pancreatitis of the author’s clinical practice were collected.
The CT examinations were reviewed to look for the different mattering radiological features of AP according to AC.
Acute pancreatitis is considered to begin in 1st day of pain,
and not the day on which the patient looks for medical evaluation.
The revised Atlanta Classification stratifies acute pancreatitis into two morphologic types based on imaging findings: interstitial edematous pancreatitis (IEP) and necrotizing pancreatitis.
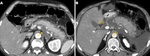
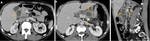
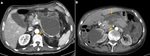
Interstitial edematous pancreatitis is more common and represents nonnecrotizing inflammation of the pancreas.
On CECT,
diffuse or localized enlargement of the pancreas with normal homogenous enhancement of the parenchyma will be present (Fig. 1).
Although interstitial edema may cause the gland to enhance less avid and in a more heterogeneous way than the normal pancreas (Fig. 1).
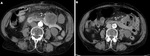
The retroperitoneal and peripancreatic tissues can appear normal or show mild inflammatory changes: mild stranding peripancreatic densities and/or small amounts of nonenhancing areas of fluid density (Fig. 1 and Fig. 2).
Necrotizing pancreatitis accounts for a minority of cases and it can present in three configurations:
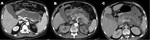
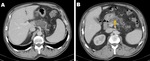
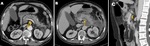
- Pancreatic necrosis alone (< 5%): Presents as portions of nonenhancing pancreatic parenchyma without associated peripancreatic necrosis (Fig. 3).
Initially,
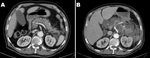
parenchymal necrosis appears as a more homogeneous nonenhancing area of variable attenuation and,
later in the course of the disease,
as a more heterogeneous area.
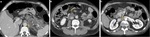
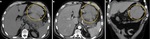
The extent of parenchymal necrosis is usually quantified as less than 30%,
between 30% and 50%,
and greater than 50%.
In order to correctly evaluate the amount of pancreatic necrosis,
CT should be preferably performed 3 to 7 days after onset of symptoms (Fig. 4).
- Peripancreatic necrosis alone (20%): Pancreas enhances normally but there is necrosis of the peripancreatic tissues,
presenting as heterogeneous areas of variable density containing fluid and nonliquefied components (Fig. 3).
- Combined pancreatic and peripancreatic necrosis (75%): The radiologic appearance is a combination of the findings described for the other two subtypes (Fig. 3).
Both morphologic pancreatitis subtypes can develop collections as local complications.
The revised Atlanta classification distinguishes between collections composed purely of fluid (occur in EIP) and collections composed of fluid and necrotic debris (occur in necrotizing pancreatitis).
Thus,
the collections are named according to the pancreatitis subtype,
but also according to time since disease beginning (Fig. 5).
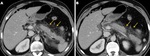
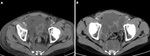
Acute peripancreatic fluid collection (APFC) are peripancreatic collections composed only of fluid without nonliquefied components arising in patients with IEP during the first 4 weeks.
At CECT they have homogeneous fluid density,
have no complete definable wall,
and conform to the anatomic boundaries of the retroperitoneum usually immediately next to the pancreas (Fig. 1).
Most APFCs remain sterile and are spontaneously reabsorbed,
so intervention at this stage is to be avoided,
because it could introduce infection.
Only the rare infected APFC necessitates drainage.
When an APFC persist beyond 4 weeks it becomes more organized and develops a capsule,
being termed pseudocyst.
At CECT,
it is depicted as well circumscribed,
usually round or oval,
homogeneous peripancreatic collection with fluid attenuation (some dependent debris may be present),
surrounded by a well-defined enhancing wall (Fig. 6).
Many pseudocysts resolve spontaneously,
although some can have associated complications and symptoms.
Pseudocysts may have a connection to the pancreatic ductal system.
The identification of a ductal communication can be determined by CECT,
however it is best seen at MR cholangiopancreatography.
These ductal connections can seal off,
often leading to cyst resolution.
Acute necrotic collection (ANC) correspond to nonencapsulated collections of inflammatory fluid and necrotic pancreatic/peripancreatic tissue arising in patients with necrotizing pancreatitis during the first 4 weeks.
On CECT,
they can be depicted as homogeneous fluid density or heterogeneous collections,
because of the presence of nonliquefied debris,
in the pancreatic and/or peripancreatic area.
If no intravenous contrast material is given,
the presence of fat attenuation within the collection is helpful for identifying necrosis (Fig. 7).
During the first week of pancreatitis,
imaging differentiation between APFC and ANC can be difficult,
since both could appear as homogeneous hypodense collections adjacent to the pancreas (Fig. 8).Usually,
imaging in the second week will allow the distinction to be made,
since ANC usually become more complex on imaging (Fig. 8).
ANC are usually asymptomatic and undergo progressive liquefaction and decrease in size.
It is important to note that:
- Any fluid collection in the presence of imaging findings of pancreatic parenchymal necrosis must be designated as ANC even those of homogeneous fluid attenuation (Fig. 9);
- Any fluid collections in the pancreatic parenchyma should be diagnosed as necrosis and not as APFC (Fig. 10).
When an ANC persists for more than 4 weeks it matures and develops a thickened nonepithelialized wall,
becoming an encapsulated collection named walled-off necrosis (WON).
Thus WON contains fluid and necrotic pancreatic and/or peripancreatic tissue,
just like ANC,
which is demonstrated at CECT as nonliquefied debris within the fluid,
in an heterogeneous appearance similar to that of ANC (Fig. 7).
Nearly half WON can present with symptoms or complications,
so radiologic follow-up for growth and complications is recommended,
even for initially asymptomatic cases.
Walled-off necrosis can have a similar appearance to pseudocyst on CECT,
making imaging differentiation difficult.
However,
accurate distinction is important since treatment management of these collections is notably different.
Symptomatic pseudocysts require only simple drainage,
whereas walled-off necrosis frequently requires removal of necrotic material.
The presence of imaging documented pancreatic necrosis from earlier in the disease course may be helpful,
and magnetic resonance imaging,
transabdominal ultrasonography,
or endoscopic ultrasound can be valuable for documenting the presence of necrotic material within the collection.
Each of these collections can be sterile or infected,
being necrotic collections far more likely to become infected.
Infected collections have poor outcomes and require a different treatment plan than do sterile collections,
thus accurate distinction is important even though the diagnosis of infection can be difficult on imaging.
Infection can be suggested on CECT images by the presence of multiple small gas bubbles scattered throughout the collection (Fig. 11),
which is not always present.
Furthermore,
collections can contain gas if a communication between the collection and the gastrointestinal tract exists,
or it can be present after drainage procedures.
In the absence of gas in the collection,
infection can be diagnosed based on image-guided fine-needle aspiration and culture.
Vascular complications can also occur with acute pancreatitis,
causing substantial morbidity and mortality.
Venous thrombosis is the most common vascular complication of pancreatitis,
it results from the inflammatory reaction associated with the disease.
Splenic vein is the vessel more frequently affected,
although the portal and superior mesenteric veins can also be involved.
Thrombosed vessels appear as nonenhancing tubular structures on CECT (Fig. 12).
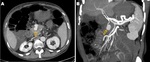
Vessel erosion by pancreatic enzymes can lead to spontaneous arterial haemorrhage with the formation of haematoma (Fig. 13),
or the formation of pseudoaneurysm.
Pseudoaneurysm usually occur in the splenic,
gastroduodenal,
or pancreaticoduodenal arteries,
and it takes time to be formed,
so they are not present early in the disease.
Pseudoaneurysms tend to gradually enlarge and may rupture,
being associated with high mortality rates.
Pseudoaneurysms are depicted as rounded structure arising from and showing enhancement similar to the donor artery at CECT (Fig. 14).