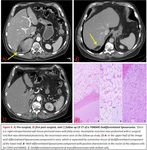
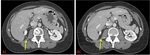
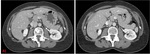
The retroperitoneum is a complex space that contains solid organs and hollow viscera,
lymphatics and lymph nodes,
major vascular structures,
and stromal tissues.
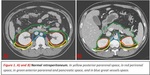
It is located between the posterior parietal peritoneum and transversalis fascia.
It is limited cranially by the diaphragm and caudally by the pelvis.
It is divided into four compartments: perirenal,
anterior pararenal,
posterior pararenal and the great vessels space.
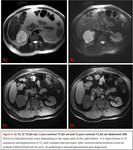
Fig. 1
Different imaging techniques can be used to evaluate RPLPS:
- Conventional radiography (CR)
- Ultrasound (US) and Doppler US
- Contrast-enhanced computed tomography (CT)
- Magnetic resonance imaging (MRI)
- 18F-Fluorodeoxyglucose positron emission tomography combined with CT (PET-CT)
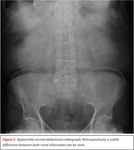
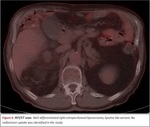
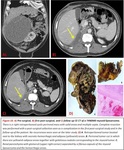
CR of the abdomen is usually the first imaging technique performed as an initial approach to the patient,
usually in the emergency context.
The information provided by this technique is very scarce.
However,
mass effect with a displacement of bowels,
intestinal altered aeration or the presence of calcification in the tumoral mass can be seen.
Fig. 2
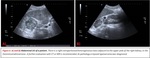
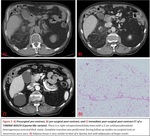
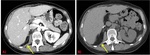
US is useful as a fast first technique for abdominal abnormalities. It cannot properly assess the extent or depth of the tumor,
especially in people with a thick constitution or with a lot of abdominal meteorism.
Sometimes RPLPS are detected as incidental findings in US performed for other reasons.
It presents usually as a large hyperechoic mass that can demonstrate posterior acoustic shadowing.
Due to their big size and location,
they are frequently difficult to evaluate.
Doppler may offer additional information detecting the presence of intratumoral vessels and assessing external vascular permeability or compression.
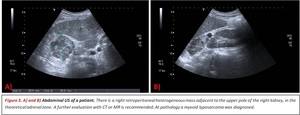
Fig. 3
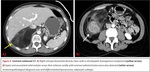
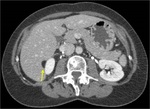
The initial imaging modality of choice for the study of RPLPS is abdominopelvic CT (on which this presentation will focus).
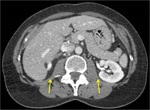
RPLPS tend to be large masses with the presence of great proportions of macroscopic fat.
The existence of fat within a retroperitoneal mass will be very useful to establish an adequate differential diagnosis.
They can be seen as a soft tissue mass with low attenuation values with measurements ranging within -10 to -100 Hounsfield Units (HU).
If the proportion of fat within a voxel is small,
then the UH may increase and can be difficult to identify.
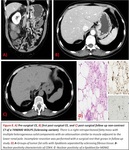
Solid components within the tumor can also be found depending on RPLPS subtype.
Calcification within the mass is infrequently present and have proved to be a sign of poor prognosis,
often indicating dedifferentiation.
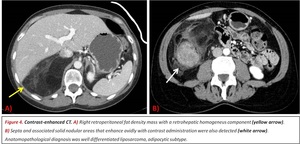
Fig. 4
MR is more sensitive for detecting fat than CT as it has better soft-tissue contrast resolution but lower spatial resolution.
It is usually less accessible and more time consuming than CT so it is not routinely used for the follow up of patients with RPLPS.
There are two imaging sequences that can detect the presence of fat within a lesion: fat saturation for detecting macroscopic fat and chemical shift phenomenon in phase and opposed phase sequences for distinguishing microscopic fat.
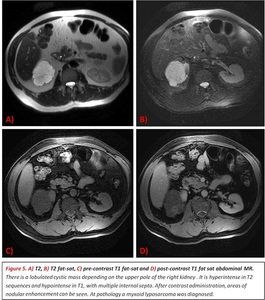
Fig. 5
18F-Fluorodeoxyglucose PET-CT is not an adequate technique for distinguishing RPLPS subtypes,
treatment monitoring or recurrence detection as approximately half of the LPS have a SUVmax < 2.5 g/mL.
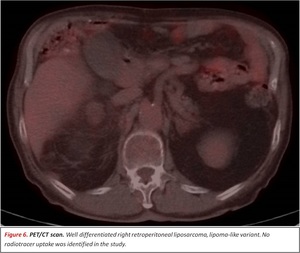
Fig. 6
Liposarcoma subtypes and imaging characteristics
Well-differentiated liposarcoma (WDLPS)
It is generally described as a predominantly hypoattenuating solid mass because of its high-fat content with minimal soft-tissue attenuation (typically < 1 cm).
In our review,
the majority of RPLPS were WDLPS most of them of the adipocytic subtype.
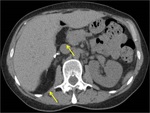
Many WDLPS were found in our study with solid parts associated with the fat component,
even when no dedifferentiated component was diagnosed at pathology. Fig. 4
They commonly contain thick septa (> 2 mm) within the fatty component that enhance homogeneously after contrast administration.
When no soft-tissue component is found,
the appearance may be indistinguishable from lipoma.
Only fat RP lesions must be considered a WDLPS rather than a lipoma until proven otherwise with pathology.
Radiologically,
there is currently no reliable method to differentiate the specific variants,
but there are some clues that can lead us to select one over the other:
- Adipocityc (lipoma-like): homogeneous low attenuation soft tissue mass on CT.
Lesion with similar intensity to subcutaneous fat in all MR sequences.
Fibrous septa may be thick,
irregular or nodular,
greater than those seen in lipomas.
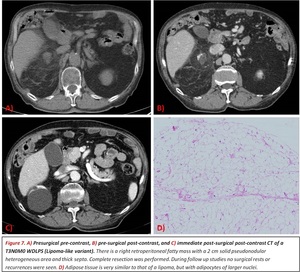
Fig. 7
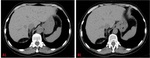
- Sclerosing: areas of CT attenuation and MR signal intensity similar to muscle can be found within the tumor.
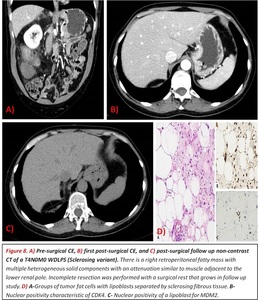
Fig. 8
- Inflammatory: rare extensive lymphoplasmacytic infiltration.
Appears as a fibro-fatty mass with a homogeneous hyperintense signal on T2-weighted MR images.
- Spindle cell: this subtype is very uncommon and has no specific imaging characteristics.
Dedifferentiated liposarcoma
Dedifferentiation always occurs within WDLPS throughout their evolution.
It can appear very similar to WDLPS.
Dedifferentiation is suggested when focal nodular soft-tissue or nonlipomatous components appear within a previous homogeneous fat-containing mass. The appearance of nodules greater than 1 cm is described in the literature as a sign of dedifferentiation.
In our work,
this finding had poor value.
Solid components,
even with great solid nodules,
appeared also in WDLPS (figure 1).
However,
a close relationship between the maximum diameter of the solid component and the dedifferentiation of the RPLPS was found.
All of our cases with dedifferentiated liposarcoma had a solid nonlipomatous component > than 5 cm (range from 5 to 27 cm)
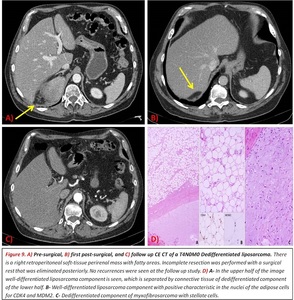
Fig. 9
Myxoid liposarcoma
They generally have components with lower attenuation than adjacent muscle but higher than simple fluid (> 20 HU).
They present low signal intensity on T1- and high signal intensity on T2-weighted images,
so they can resemble cystic lesions on non-contrast images because of the large amounts of extracellular myxoid material.
They enhance heterogeneously after contrast administration.
They also have thick septa and patchy or nodular soft-tissue components associated.
They are really aggressive with an unusual pattern of metastatic spread,
commonly to paraspinal regions,
bone and opposite retroperitoneum.
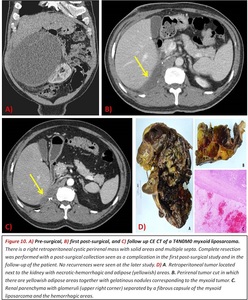
Fig. 10
Pleomorphic liposarcoma
It is the least common subtype,
a high-grade and very aggressive tumor with a high incidence of metastasis and recurrence.
They usually appear as a well-circumscribed mass containing little or no fat,
with a similar attenuation to that of muscle on CT.
They often show internal hemorrhage and necrosis.
It is very difficult to differentiate from other retroperitoneal sarcomas because they have no characteristic findings on MR.
Differential diagnosis of fat-containing retroperitoneal lesion
Other fat-containing masses that can be found in the RPS are listed above1:
Adrenal
- Common diagnoses: Adenoma,
myelolipoma.
- Rare diagnoses: Carcinoma,
pheochromocytoma,
neurogenic tumors,
angiomyolipoma (AML),
lipoma and liposarcoma,
metastasis.
Renal
- Common diagnoses: AML.
- Rare diagnoses: Renal cell carcinoma (CCR),
lipoma and liposarcoma,
replacement lipomatosis.
Pancreatic
- Common diagnoses: Lipoma,
focal pancreatic steatosis.
- Rare diagnoses: Pancreatic pseudolipohypertrophy,
pancreatic teratoma.
Primary retroperitoneal
- Common diagnoses: Lipoma and liposarcoma.
- Rare diagnoses: Myelolipoma,
primary and secondary germ cell tumors (GCTs),
neurogenic tumors,
lipomatosis,
fat necrosis,
extramedullary hematopoiesis.
Facing these studies: tips for diagnosis and follow up evaluation.
Complete surgical resection is the only potential curative management option for RPLPS.
Multidisciplinary teams performing multivisceral resections are usually required.
Complete resections are difficult to achieve and local recurrence occurs in a large proportion of patients,
in some studies close to 100%.
Therefore long period follow up is mandatory for these patients with many studies performed searching for surgical rests of recurrences.
Metastases are very uncommon,
none in our review.
It is very important that the initial extension of the tumor is accurately assessed in the diagnostic CT.
RPLPS can affect many compartments and findings can be very subtle.
Surgical management will be determined by CT initial findings.
After treatment,
first postsurgical CT must be correctly evaluated as it will be used as a control for the following studies.
Surgical rests can be easily overlooked as they can be confused with normal RP fat or postsurgical changes.
Detecting surgical rests and recurrences in follow up studies can be very challenging.
There are some tips that can help identifying them:
- Take your time: abdominopelvic CT for evaluating postsurgical findings of a RPLPS is not an easy study; a careful search and the need to compare with many previous studies is often very time-consuming.
- Always review the patient´s clinical history: the subtype of tumor and surgery performed is important information that will help us comprehend which findings to expect.
- Revise first diagnostic study: evaluate the first CT where the tumor was diagnosed.
Where did it extend,
how it looked like and behaved.
Recurrences will usually appear in the same areas.
- Always compare with the first postsurgical studies: recurrent RPLPS can grow very slowly and changes between two immediate studies may be very subtle.
Comparing with earlier studies will make the modifications more evident and the detection of recurrence easier.
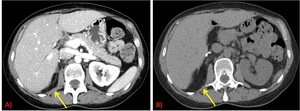
Fig. 11
- Check the contralateral side: if there are asymmetries between RPS structures and components that were not seen on prior studies look carefully,
it can be the clue for recurrence detection.
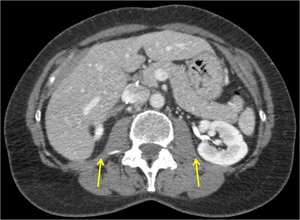
Fig. 12
- Analyze the composition of the new findings: Whether fat,
fibrosis or soft-tissue component is present is important.
Tumor characteristics can change with the appearance of solid components.
Sometimes the detection of a tiny solid component in a previous fatty area can be the key to the diagnosis of a recurrence.
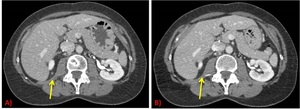
Fig. 13
- Look carefully at the contours of the neighboring organs: surgical rests and recurrences can present as a loco-regional mass effect,
deforming the edges or contours of adjacent organs and structures.
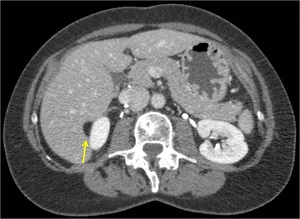
Fig. 14
- Search in adjacent compartments: usually,
RPLPS extend through many compartments and locations don´t limit the search for recurrences to just one compartment.
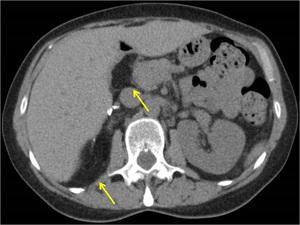
Fig. 15
- Take measures and pictures of any suspicious findings: therefore in the next follow up study an adequate comparison can be performed and possible growth or stability of the finding can be stablished.
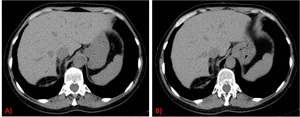
Fig. 16
- If you are not sure of a finding write it in your report: subsequent radiologist evaluating the next study will search more actively in that area.
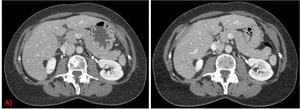
Fig. 17