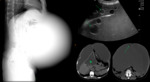
We retrospectively selected and analysed relevant radiological findings of X-ray,
Ultrasound,
CT,
and MRI images from our Institutions’ databases.
Hepatobiliary typical and atypical spectrum of manifestations of Entamoeba hystolitica,
Schistosomiasis,
Echinococcus,
Fasciola,
and Ascaris lumbricoides will be highlighted.
Parasites are endemic in some regions,
but are spreading worldwide due to travel.
Involvement of the hepatobiliary system is infrequent.
The diagnosis is challenging because most patients are asymptomatic in the initial phases.
Suspicion is raised by considering epidemiologic data.
Imaging examinations are often the key to the diagnosis,
but differential diagnosis includes neoplasia,
trauma,
and autoimmune diseases.
Laboratory confirmation is frequently required.
This presentation reviews the radiologic appearances of the most common hepatic and biliary system parasitic diseases.
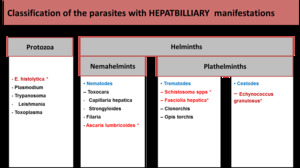
Fig. 24: Classification of the parasites with HEPATOBILLIARY manifestations
Entamoeba hystolitica
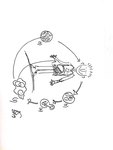
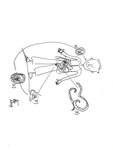
Transmission route and life-cycle Fig. 1 :
- Individuals are infected upon eating or drinking contaminated food
- Swallowed cysts (1) excyst in the small bowel (2),
where they damage the epithelial cells,
invade the mucosa and cause amoebaean colitis
- Trophozoites (3) can travel to the liver and form abscesses.
- Diaphragmatic rupture can occur secondarily.
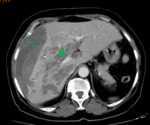
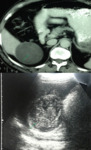
Imaging findings (liver):
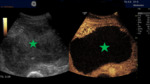
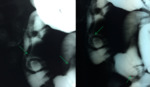
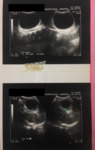
- Ultrasound: well-defined non-pure liquid lesions Fig. 2
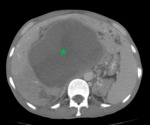
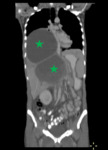
- CT: Well-defined hypodense lesions,
without contrast enhancement.
A true hepatic amoebic abscess is nonsuppurative,
without a discernible wall.
Fig. 3 Fig. 4
- MRI: T1: low signal.
T2: high signal.
Perilesional oedema.
Clues to the diagnosis:
- Epidemiology (+++)
- Imaging : non-specific hepatic abscess –Diaphragmatic disruption is suggestive of amoebic aetiology.
- Serology
Ascaris lumbricoides
Transmission route and life-cycle Fig. 5 :
- Individuals are infected through ingestion of eggs.
- Humans are permanent hosts.
Adult A.
lumbricoides(1) live in the bowel.
Transmission occurs through the eggs (2),
in an oral-fecal route,
due to poor sanitation.
- Sometimes the worms migrate,
through the ampulla of Vater, to the biliary tree.
- Bile duct obstruction,
cholangitis and pancreatitis can be the initial presentation of the parasitosis.
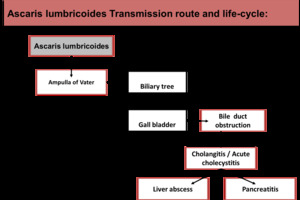
Fig. 25: Ascaris lumbricoides Transmission route and life-cycle
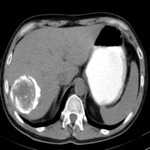
Imaging findings:
- X-ray: linear filling defects can be found inside the small bowel or colon in contrasted studies,
as well as in ERCP Fig. 6 .
- Ultrasound: linear structures can be found inside bile ducts or the gallbladder.
- CT: may show complications,
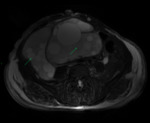
such as bile duct dilatation or pancreatitis Fig. 7 .
- MRI: may also show worms as filling defects in the bile ducts.
Clues to the diagnosis:
- Epidemiology (non-specific,
worldwide distribution).
Consider the possibility of poor sanitation.
Infection is often asymptomatic.
- Imaging can be straightforward,
when the existence of parasites is clearly demonstrated,
or challenging in cases of biliary involvement.
- The worms or eggs can be found by directly examining the faeces.
- Serology – may be useful,
but due to the high prevalence of infection is many times unspecific.
Fasciola hepatica
Transmission route and life-cycle Fig. 8 :
- The parasite is present worldwide.
- Individuals are infected through ingestion of metacercaria (1) present in water or raw aquatic plants (watercress)(2).
- Parasitic flukes (3) can migrate to the peritoneum and perforate Glisson’s capsule.
- Parasitic flukes then invade the liver,
causing hepatic fascioliasis.
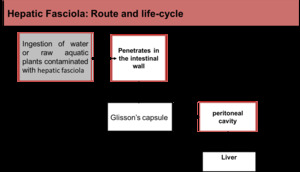
Fig. 26: Fasciola hepatica: Transmission route and life-cycle
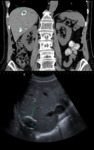
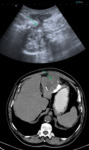
Imaging findings:
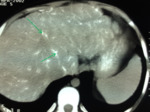
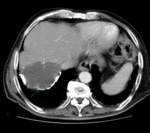
•Ultrasound and CT: hepatic abscesses can be found in subcapsular locations.
Subcapsular fluid collections and hemoperitoneum can be found in the invasive phase Fig. 9 .
Clues to the diagnosis:
- Epidemiology (often non-specific).
- Imaging can be challenging.
Subcapsular fluid/hematic collections can be a clue to the diagnosis,
especially in the absence of trauma or other feasible explanations Fig. 10 .
- Lab tests (eosinophilia) and Serology – +++
Shistosoma mansoni
Transmission route and life-cycle Fig. 11 :
- The parasites are present in Africa,
Asia,
India,
and Latin America.
- Individuals are infected due to penetration of the skin by swimming cercaria (1) in contaminated waters.
- Within the body,
metacercaria became Shistosomulae,
and migrate to the bowel veins,
where they mature and produce eggs (2).
- Through the faeces,
eggs are transferred to the fresh water (3).
- There,
miracidia (4) are released,
and penetrate snails (intermediate hosts) (5).
- In snails,
the parasite matures to sporocysts.
These release cercaria,
and the cycle re-starts.
- Liver infection occurs when eggs are trapped in the hepatic sinusoidal circulation.
An inflammatory reaction ensues,
resulting in hepatomegaly,
fibrosis,
and chronic liver disease.
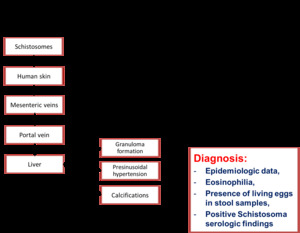
Fig. 27: Schistosoma: Transmission route and life-cycle
Imaging findings:
- Late manifestations of the disease.
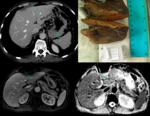
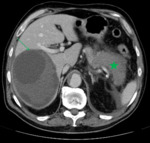
- Ultrasound and CT: Hepatosplenomegaly,
with characteristic hyperechoic thick linear bands along the portal tracts in ultrasound; calcified peri-portal spaces in CT (“tortoise shell” or “fish scale” appearance.
–Liver cirrhosis and portal hypertension are often present Fig. 12 Fig. 13 .
Clues to the diagnosis:
- Epidemiology (+++).
- Imaging: In late phases the diagnosis is straightforward.
Differential diagnosis is performed with autoimmune hepatitis.
- Lab tests: Serology (+++)
Echinococcus granulosum
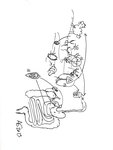
Transmission route and life-cycle Fig. 14 :
- Worldwide endemic parasitosis.
Life-cycle includes sheep and carnivores (e.g.
dogs).
Humans are accidental intermediate hosts (1).
- Infestation occurs due to ingestion of eggs of Echinococcus granulosum(2),
released from adult worms living in dogs (3).
- Life-cycle includes a larval stage of the tapeworm,
present in sheep offal (4).
- The eggs hatch,
and the larvae migrate from the intestine to other parts of the body,
where cysts are formed.
The liver is the most frequently affected organ.
Imaging findings:
The infection can be asymptomatic
- In the liver,
symptoms (pain,
upper right quadrant discomfort,
jaundice),
are due to compression of adjacent structures. Rupturing of the cyst can cause anaphylaxis.
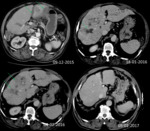
- Imaging findings include a spectrum of manifestations,
related to cyst progression,
host reaction to the cyst,
and possible complications Fig. 22 Fig. 23 .
The WHO-IWGE classification was based on the first classification (by Gharbi).
It is based on US features,
but can also be applied to CT findings.
An active (CE 1 and 2),
transitional (CE 3),
or inactive (CE 4 and 5) stage of the infection can be inferred based on imaging findings.
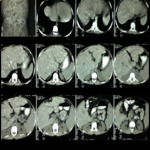
CE1 (Gharbi I) – simple cyst Fig. 15
CE2 (Gharbi III) – multivesicular cyst Fig. 16 Fig. 18
CE3A (Gharbi II) – cyst with detached membranes (“water-lily” sign) Fig. 15
CE3B (Gharbi III) – daughter cysts in solid matrix
CE4 (Gharbi IV) – heterogeneous cysts (no daughter cysts present) Fig. 17 Fig. 19
CE5 (Gharbi V) – CE4 with calcified wall Fig. 20 Fig. 21
Clues to the diagnosis:
- Epidemiology (+++).
- Imaging: In late phases the diagnosis (US,
CT) is straightforward.
- Lab tests: Serology ( +++)