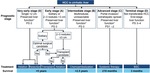
Radiologist should be aware of normal post-LT appearance [Fig.2] and of clinically non-significant abnormal findings such as pleural effusion,
mild ascites,
small perihepatic fluid collections,
subtle perihepatic hematoma and periportal edema [Fig.3].
These findings must be evaluated during follow-up but should not cause concern, as they resolve themselves within few weeks.
Most frequent complications are usually classified in vascular (arterial/venous) and biliary.
Hepatic artery (HA) complications include thrombosis,
stenosis and pseudoaneurysm.
HA trombosis occurs in 8% of transplant,
classified in early (within 15 days of LT) and delayed thrombosis.
Early thrombosis may by related to increased cold ischemic time of the donor liver,
ABO blood type incompatibility and acute rejection.
Delayed thrombosis may occur years after LT and it is generally associated with chronic rejection and sepsis.
The hepatic artery is the sole arterial supply to the transplanted liver so the clinical presentation ranges from fulminant hepatic failure to delayed biliary leak.
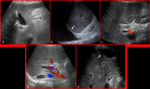
Doppler examination shows complete absence of both proper hepatic and intrahepatic arterial flow [Fig.4].
After delayed hepatic artery thrombosis,
arterial collateral vessels may develop and intrahepatic flow may be identified.
Intrahepatic arterial waveform will be abnormal,
displaying a tardus-parvus pattern.
Treatment consists of urgent revascularization of the graft with thrombectomy nevertheless up to 60% of patients require retransplantation.
HA stenosis occurs in up to 11% of transplant recipients,
most frequently at the anastomotic site.
Causes include clamp injury,
intimal trauma due to perfusion catheters at the time of surgery,
or disrupted vasa vasorum leading to ischemia.
Clinically,
arterial stenosis may lead to biliary ischemia,
with hepatic dysfunction and eventual hepatic failure.
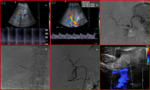
Doppler examination shows accelerated velocity greater than 2–3 m/sec at the site of narrowing with associated turbulence distal to the stenosis.
Intrahepatic arterial waveforms display a tardus-parvus pattern with prolonged acceleration time and decreased resistive index.
Treatment includes balloon angioplasty or stenting [Fig.5].
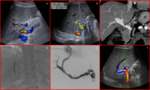
HA pseudoaneurysms are rare,
most often mycotic,
occurring mostly at the vascular anastomotic site.
Occasionally pseudoaneurysms may be intrahepatic or peripheral due to focal parenchymal infection or following percutaneous interventions.
The clinical presentation is often late,
with hepatic failure or acute shock if the pseudoaneurysm ruptures.
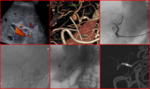
US reveals a periportal or intrahepatic anechoic structure usually in juxtaposition to the course of the hepatic artery.
Doppler examination demonstrates a disorganized arterial flow pattern.
Intrahepatic arterial tardus-parvus waveforms may be present.
Treatment include stenting or surgery [Fig.6].
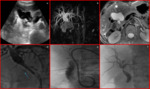
Venous complications may be divided in portal vein and inferior vena cava complications; both including stenosis and thrombosis.
Portal vein complications occur in 1%–2% of transplant recipients.
Causes include misalignment or excessive vessel length,
hypercoagulable states,
or previous portal vein surgery.
Clinical presentation includes portal hypertension,
hepatic failure,
edema and massive ascites.
IVC complications are rare,
diagnosed in less than 1% of transplant cases,
more common in case of retransplantation and in the pediatric population.
Acute IVC stenosis can be secondary to anastomotic size discrepancy or suprahepatic caval kinking from organ rotation while delayed IVC stenosis may be secondary to fibrosis,
chronic thrombus or neointimal hyperplasia.
US findings of venous thrombosis are vessel narrowing or echogenic luminal thrombus with no duplex flow [Fig.7].
Acute thrombus may be anechoic and evident only at color flow and spectral duplex analysis .
Color-US reveals vein stenosis as focal aliasing with stenotic to prestenotic velocity ratio >3.
A significant suprahepatic caval stenosis may result in reversed flow or absence of phasicity in the hepatic veins.
Treatment options are balloon angioplasty,
thrombolysis,
stenting placement performed either percutaneously (for portal complications)[Fig.8] or transjugular (for both portal and IVC treatment) [Fig.9,10] and open surgical correction.
Biliary complications occur in 80% within the first 6 months after transplantation.
We recognize leaks,
strictures,
stones or sludge.
Biliary leakage is located at the end-to-end biliary anastomosis or at the site of T-tube entry into the duct.
Non-anastomotic leaks are associated with hepatic artery thrombosis in 90% of cases.
The bile extravasates into the peritoneal cavity or forms a perihepatic fluid collection with variable clinical presentation (from mild abdominal symptoms to septic shock).
Treatment includes biliary stent placement and/or collection drainage [Fig.11].
Biliary anastomotic stenosis is secondary to scar tissue causing retraction and narrowing of the common bile duct at the suture site. It often requires surgical or radiologic intervention.
US shows dilated intrahepatic and proximal common bile duct dilatation while the common bile duct distal to the anastomosis is normal or near-normal size.
Biliary non-anastomotic stenosis is generally secondary to ischemia caused by hepatic artery compromise.
Clinical findings consist of deranged liver function or progressive obstructive jaundice.
US may show focal segmental intrahepatic or hilar ductal dilatation without an obvious obstructing mass or stone: when facing this scenario the hepatic artery should be carefully examined [Fig.12].
Ischemic type biliary lesions (ITBL) represent partial or complete sloughing of the biliary epithelium due to necrosis of the biliary tree in the absence of hepatic artery thrombosis,
resulting in intra/extrahepatic bile duct strictures or dilatations.
US reveals dilatation of the intrahepatic biliary tree with accompanying echogenic intraluminal debris.
Treatment includes balloon dilation with or without percutaneous transhepatic biliary drainage,
or direct ERCP; in our department clinical improvement was found using reabsorbable stent placement,
though further studies are needed to estimate the long term efficacy of these devices [Fig.13].