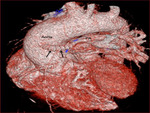
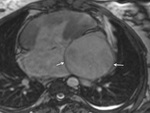
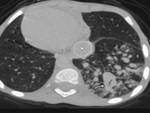
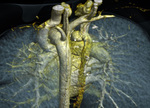
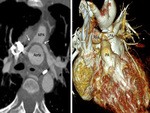
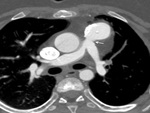
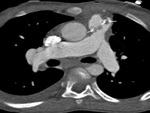
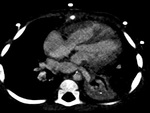
We retrospectively analyzed the scans done in post-operative cyanotic CHD patients from 2014 to 2018 in the department of Cardiovascular Radiology and endovascular intervention,
All India Institute of Medical Sciences,
New Delhi,
India.
CT Protocol: Patients were scanned with a 256/384 slice CT scanner.
CT angiogram was performed using an 80 kV technique either using a retrospective ECG gated scan or an ultra-high pitch prospective gated scan depending upon the complexity of the CHD.
MRI protocol: MR imaging protocol included cine vertical long axis (VLA),
short axis (SA),
horizontal long axis (4-chamber) and routine axial images for functional and anatomical assessment.
Additional sequences included Contrast enhanced MR Angiography (CE MRA),
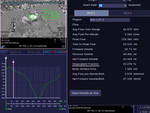
late gadolinium enhancement (LGE) images and phase contrast sequences to quantify shunt/regurgitant fractions.
PALLIATIVE PROCEDURES: The palliative procedures in cyanotic CHD are either aimed at increasing the pulmonary blood flow or to enlarge the pulmonary arteries (PA) in patients with pulmonary stenosis/atresia (for example systemic artery/vein to PA shunts) or to protect the pulmonary bed from the changes of pulmonary arterial hypertension (PAH) in cases with higher pulmonary flow (for example,
PA bands)(1).
A) Systemic artery to PA shunts: These are done in young children up to 4-5 months of age,
when the PA pressures are higher,
which requires an active flow of blood from a higher pressure circuit into the PA.
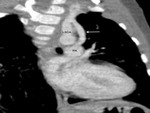
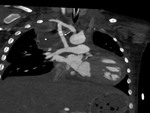
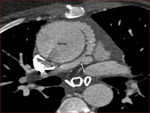
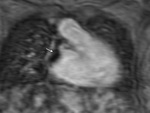
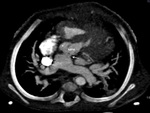
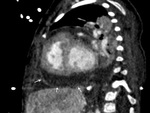
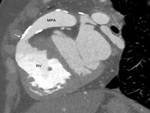
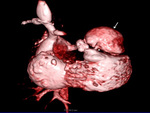
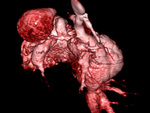
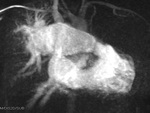
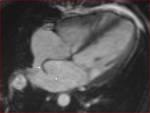
- Modified Blalock-Taussig (BT) shunt- These are side-to-side shunts,
made of PTFE,
between the subclavian artery and the ipsilateral PA (Fig.
1).
The common complications are:
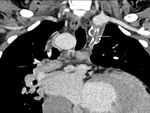
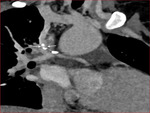
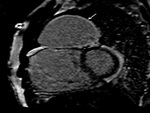
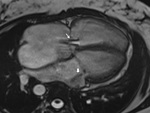
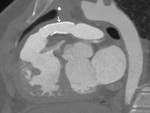
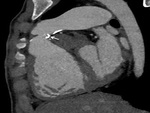
i) Thrombosis/ occlusion of the shunt (Fig.
2 and 3) with decreased
lung vascularity (Fig.
4)
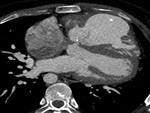
ii) Leakage from the shunt
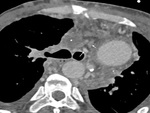
iii) Infection of the shunt with abscesses/collection around the shunt
(Fig.
5)
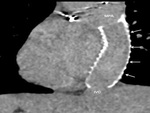
iv) Stenosis of the shunt (Fig.
6) or at its anastomotic sites (Fig.
7)
v) Aneurysms/ pseudo aneurysms at the anastomotic sites
vi) PAH
vii) Volume overload of the ventricles with ventricular dysfunction.
2.
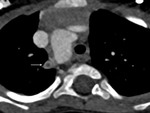
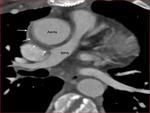
Central shunt- These are side to side PTFE shunts between the ascending
aorta and the main PA (Fig.
8).
Similar complications as
described above for BT shunts can be seen in a central shunt,
e.g.
Stenosis at the anastomotic sites (Fig.
9),
thrombosis/occlusion
(Fig.
10),
PAH,
aneurysms,
infection etc.
B) Systemic vein to PA shunts- These shunts aim to bring the systemic venous blood directly to the PA for oxygenation and are done in older children >5months of age with conditions with single ventricle physiology like tricuspid atresia,
Ebstein’s anomaly etc.
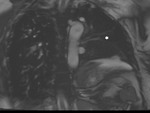
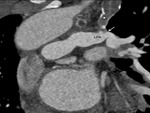
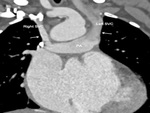
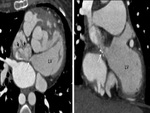
- Bidirectional Glenn shunt- Herein,
the superior vena cava (SVC) is connected end to side with the PA (Fig.
11).
This is usually a staged procedure followed by a completion Fontan procedure at a later date.
The goal of imaging is to identify the sizes of PA's and associated complications,
which are:
i) Thrombosis/ occlusion of the shunt (Fig.
12,13)
ii) Stenosis at the venous anastomosis
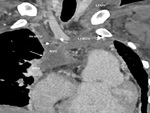
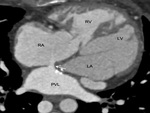
2) Extracardiac Fontan shunt- This procedure involves redirection of entire
systemic venous return to the PA,
which is usually done
by creating a lateral intra-atrial tunnel or an extra cardiac conduit (Fig.
14).
This is usually performed at the age of 2-5 years when PA size is
good enough to permit high pulmonary arterial flow,
in
children who are not the candidates for biventricular repair.
A small
fenestration may be created between the tunnel and the right atrium to
decompress the pulmonary circulation (Fig.
15).
The usual complications are:
i) Thrombosis/occlusion of the shunt (Fig.
16,17),
thrombo-embolism
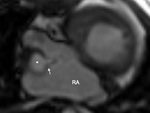
ii) Enlargement of the atria (Fig.
15),
iii) Enlargement of the Fontan conduit (Fig.
18)
iv) Pulmonary arteriovenous malformations (AVM) (Fig.
19)
v) Venovenous collaterals- between systemic veins and pulmonary
veins, due to elevated central venous pressures.
They may cause
persistent cyanosis.
vi) Stenosis at the site of cavopulmonary connections
vii) Ventricular dysfunction
viii) Coronary artery injuries during surgery leading to myocardial
ischemia
ix) Atrioventricular regurgitation (Fig.
18)
3) Kawashima procedure: The Kawashima procedure is performed in
patients with interruption of the inferior vena cava and its azygous
continuation into SVC.
The procedure involves creation of Glenn shunt
followed by anastomosis of the azygous vein to the PA
(equivalent to Fontan),
which redirects the entire systemic blood flow to
the PA (Fig.
20).
A completion Fontan is not required.
Complications are:
i) Pulmonary AVMs
ii) Stenosis/thrombosis of hepatic vein conduits connecting to innominate
vein.
C) Pulmonary banding: It is done by tying a prosthetic material around the PA to protect the pulmonary arterial bed from changes of PAH in patients with increased pulmonary flow,
e.g.
DORV without pulmonary stenosis or to prepare the left ventricle to pump against high pressure prior to an arterial switch operation in TGA (2). The major complications are:
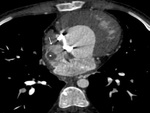
i) Dilatation of the PA proximal to the site of banding (Fig.
21)
ii) Pulmonary regurgitation.
iii) Others: PA pseudo aneurysms,
stenosis,
migration of
the band into distal branch PA etc.
CORRECTIVE PROCEDURES: The complications arising out of corrective procedures in the common cyanotic CHD are hereby described.
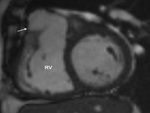
A) Tetralogy of Fallot: The three common repair procedures for TOF are a) annuloplasty (infundibulectomy) or b) transannular patch repair or c) right ventricle (RV)-PA conduit along with VSD repair.
The incidence of late complications in corrected TOF is approximately 10%.
I) Immediate post- operative complications:
1) Infection (Fig.
22)
2) Coronary injury causing myocardial ischemia.
II) Cardiac complications-
1) Residual right ventricular outflow tract (RVOT) stenosis- This is the
commonest late finding after infundibulectomy (Fig.
23).
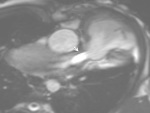
2) RVOT patch dyskinesia/aneurysm/pseudo aneurysm (Fig.
24,25,26).
3) Tricuspid regurgitation (TR) (Fig.
27).
4) Recurrent/residual VSD (Fig.
28).
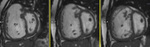
5) LV dysfunction- It is a late outcome in post TOF repair patients and is
attributed to abnormal ventricular-ventricular interactions in patients
with RV dysfunction (Fig.
29).
6) LMCA compression by dilated PA- It is rare complication but can occur
due to operative vascular injury.
III) Pulmonary arterial complications-
1) Pulmonary regurgitation (PR)- Obligatory PR develops most commonly
after TA patch annuloplasty and is the most common late complication
of post operative TOF (Fig.
30).
2) Residual main PA stenosis (Fig.
31).
3) Pulmonary arterial dilatation (Fig.
32)
4) Branch PA stenosis (Fig.
33) or aneurysm (Fig.
34)
IV) Complications related to RV to PA conduit repair-
1) Conduit degeneration (Fig.
35)
2) Conduit stenosis- Stenoses can be identified in the conduit (Fig.
36) or
at the site of anastomoses with the PA(3).
B) Transposition of Great Arteries (TGA):
I) Post atrial switch operation: An atrial baffle is created using either
homologous tissue (Senning operation) or prosthetic material (Mustard
operation) to redirect caval blood to the left atrium and further into the
left ventricle in patients with D-TGA. These procedures are now rarely
done because of the complications associated with them.
Major
complications that can be seen are:
1) Baffle leakage (Fig.
37)
2) Baffle stenosis- Stenosis of the systemic venous limb (Fig.
38) is
more common than the stenosis of pulmonary venous limb (Fig.
39).
3) Venacaval obstruction
4) RV enlargement
5) RV failure,
tricuspid regurgitation.
II) Post Jatene’s arterial switch operation (ASO): This is procedure of choice
in neonates with TGA with an intact ventricular septum or with a small
VSD.
While switching the great vessels,
Lecompte’s maneuver is usually
performed which involves bringing the PA anterior to the
neo-aorta.
Major complications seen are:
1) Stenosis at the anastomotic site of the PA to the new
pulmonic root- the most common complication after ASO (Fig.
40).
2) Central or peripheral stenosis of the PA's: This occurs
especially as a complication of Lecompte’s maneuver while PA crosses
the aorta (Fig 41).
3) Supravalvular aortic stenosis,
left ventricular dysfunction
4) Neo-aortic root dilatation and insufficiency
5) Coronary artery ostial kinking and stenosis: As a sequel to coronary
artery button transfer(4).
III) Rastelli procedure: It can be performed in patients with D-TGA with a
large VSD and left ventricular outflow tract obstruction where arterial
switch is not feasible.
Common complications are:
1) Stenosis / occlusion of the extracardiac conduit or at its anastomotic
site (Fig.
42),
branch PA stenosis.
2) Conduit degeneration (Fig.
42)
3) Infection of the conduit (Fig.
43)
4) Ventricular dysfunction
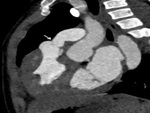
C) Post Total Anomalous Pulmonary Venous Connection (TAPVC) Repair: Pulmonary veins are rerouted to the left atrium by anastomosis of the common channel with the LA.
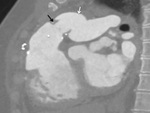
The most important complication is the obstruction to pulmonary venous flow (Fig.
44).