Among the "historical" signs of IMD,
the best known is the HS (figure 1),
defined as a solid parenchymal nodule with a peripheral halo of ground-glass; it corresponds histopathologically to a pulmonary infarction surrounded by a halo of alveolar hemorrhage.
In angioinvasive mycoses,
fungal hyphae invade the medium and small-sized pulmonary vessels causing thrombosis and hemorrhagic infarction with coagulative necrosis of the pulmonary parenchyma.
(8) The incidence of HS in patients with IMD is particularly high during the initial phases; in fact,
over time,
the HS tends to disappear.
Caillot et al.
documented how the incidence of HS in neutropenic patients with IMD on days 0,
3,
7 and 14 from the onset of infection was 96%,
68%,
22% and 19%,
respectively.
Although it is widely recognized that HS is a suggestive sign of IMD (Yeghen and Bernard have shown that the presence of HS has a high positive predictive value> 90%),
it is early and fleeting.
Moreover,
the incidence of this sign varies considerably among the various authors,
from 25% to 95%.
This is probably due the different meaning attributed to the definition of HS and to the different timing with which the CT were performed in several studies.
(9-10)

Fig. 1: Figure 1 - HRTC: solid parenchymal nodule with peripheral halo (HS)
The ACS (Figure 2) is defined as a peripheral sickle of radiotransparency (crescent moon appearance) in the context of a parenchymal nodule.
It is a late sign of IMD,
it appears at the resolution of neutropenia and it is accompanied by the presence of neutrophil infiltrates in the context of fungal lesions (Figure 3).
It follows the retraction of the necrotic pulmonary parenchyma to the adjacent healthy parenchyma,
with evidence of an aerial sickle in the context of the lesion.
In the study of Caillot (10) the ACS was observed in 8%,
28% and 63% of cases respectively on days 3,
7 and 14 after the onset of infection.
Therefore,
the ACS has a limited diagnostic and therapeutic utility as a late sign linked to the patient's haematological recovery phase.
It has a prognostic significance as indicative of a favorable outcome with a tendency to lesion resolution.
(11-12)

Fig. 2: Figure 2 - HRCT: solid parenchymal nodules with simultaneous air crescent (ACS)
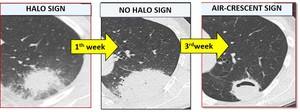
Fig. 3: Figure 3 - HRTC: temporal evolution of angioinvasive mycotic lesions from HS to ACS at the time of patient's neutrophil hematological recovery
The RHS (Figure 4) is defined as a nodular area of ground glass opacity surrounded by a ring,
complete or incomplete,
of parenchymal consolidation; histopathologically, is attributable to pulmonary hemorrhagic infarction with coagulation necrosis of the alveolar septa caused by the invasion and occlusion of peripheral pulmonary arteries by fungal hyphae with a greater amount of hemorrhage at the periphery (the ring-shaped consolidation) than in the center,
where alveolar air remains (the central ground-glass opacity). In the neutropenic patient,
RHS is highly suggestive for IMD and was observed in 94% (13) and 100% (16/16) (14) of patients with mucormycosis,
so its presence is considered highly suggestive of invasive pulmonary mucormycosis.
However,
as with the halo sign,
the sensitivity of the RHS is limited by its transitory nature.
(1,
15-16)
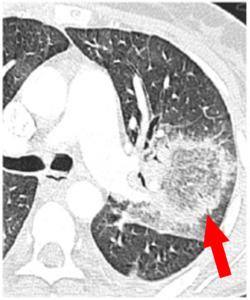
Fig. 4: Figure 4 - Thorax CT acquired in the venous phase after administration of intravenous contrast medium: nodular area of ground glass opacity surrounded by a parenchymal consolidation ring (RHS)
The HyS (figure 5) refers to the presence of a central hypodensity in the context of a parenchymal consolidation; it is due to the initial parenchymal necrosis secondary to vascular obstruction by fungal hyphae.
HyS is a lesion that precedes cavitation and ACS formation of about 2-19 days (mean 8.3 days).
The Hys has been described to HRCT as a sign with low sensitivity (30.2%) compared to a high specificity (100%).
(17) Recently it was demonstrated that the visibility of the HYS improves in studies with contrast media,
with sensitivity increasing up to 63.9% and high specificity (98%) (Figure 6).
(18)

Fig. 5: Figure 5 - CT scan of the acquired thorax without administration of contrast medium: solid parenchymal nodule with central hypodensity (HyS)

Fig. 6: Figure 6 - CT scan of the thorax acquired in the venous phase after administration of intravenous contrast medium: clear appreciation of a solid parenchymal nodule with central hypodensity (HyS)
The VOS (Figure 7) is a recently described sign,
visualized after intravenous administration of contrast agent with CTPA method (19-20) in the context of dense lung lesions,
of size> 1 cm,
not cavitated.
This sign is associated with the presence of angioinvasion documented as an interruption of the pulmonary vessels afferent to the parenchymal lesion,
in the absence of opacified vessels in the context of the lesion itself.
Even the irregular caliber reduction of the vessel without complete occlusion can be considered indicative of initial angioinvasion.
VOS is absent when the pulmonary vessels are present and with regular caliber both at the periphery and in the context of the lesion (Figures 8,
9,
10).
Both Sonnet and Stanzani have shown that VOS,
documented at the CTPA in febrile neutropenic oncohematological patients,
is an early,
sensitive (88%) and specific (97%) sign for IMD.
(21)

Fig. 7: Figure 7 - CT scan of the thorax acquired in the arterial phase after administration of intravenous contrast agent: amputation of the pulmonary vessels at the periphery of the lesion (VOS). Intralesional vascular structures are not recognizable

Fig. 8: Figure 8 - Pulmonary angio-CT and "Via Labicana Augustus": Vessel occlusion sign. No vascular structures are detectable inside the parenchymal consolidation
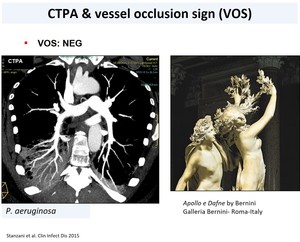
Fig. 9: Figure 9: Example of negative VOS in which the afferent vessel and its arboriform branches are visible within the parenchymal consolidation during Pseudomonas Aeruginosa infection. In analogy the "Apollo e Dafne" by Gian Lorenzo Bernini in which Dafne has hands covered with fronds

Fig. 10: Figure 10: utility of VOS in distinguishing angioinvasive fungal infections (left, positive VOS) from infections supported by other pathogens (right, negative VOS)











