CT protocols in evaluating pancreatic lesions varies from institution to institution,
but typically are multiphasic with thin-section imaging (≤3mm) and with multi-planar reconstructed (MPR) images (coronal and/or sagittal planes).
Despite optimal CT imaging,
PDAC can be falsely considered as locally resectable,
without CT signs of distant metastasis,
since focal areas of micro-metastasis might not be differentiated during CT scan [1].
In such cases MRI becomes essential.
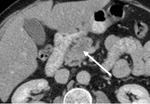
Case 1. 60 year old male with weight loss ~10 kg/6 months and painless jaundice; laboratory tests: slightly elevated bilirubin; ↑CRP 35.
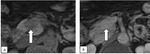
A hypodense lesion in the head of the pancreas in close contact with conflues; no distant metastases were detected.
The tumor was described as T3,
resectable disease.

Fig. 1: CT scan, axial image, portal venous phase. Year 2018 01.
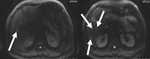
The patient underwent MRI with DWI (2018 02).
Tiny hiperintense on T2W sequence lesions in the liver with diffusion restriction were detected (Fig.2.).
These lesions were considered as micro-metastases.
The report was given: pancreatic head adenocarcinoma,
T3N1M1 (IV stage disease).
Patient underwent chemotherapy.
It was proven to be poorly differentiated (G3) PDAC.

Fig. 2: DWI B value 800.
Control CT images after 4 months.

Fig. 3: 2018 06 - MTS are seen in the liver parenchyma.
Discussion: evaluation of hepatic metastatic disease is most often performed with MDCT or MRI,
however,
MRI is more sensitive for the detection of small metastatic lesions.
Furthermore,
MRI provides better specificity in characterizing indeterminate liver lesions,
and is often used for further evaluation.
Regardless,
not every medical center perform MRI before surgery for the patients with resectable PDAC on CT images.
Cindy Chew et al.
performed the study of MRI of the liver on 69 patients,
who were previously diagnosed with resectable pancreatic cancer on CT [9].
According to their results,
47 (68.1%) patients had no evidence of liver metastasis,
while 6 (8.7%) had indeterminate lesions in the liver.
The remaining 16 (23.2%) patients had lesions consistent with liver metastasis,
which was not detected on CT.
They concluded that almost 25 % of the patients with resectable tumor on CT were positive for liver metastasis on MR imaging.
In conclusion,
the present study has demonstrated that liver MR imaging with DWI using hepatocyte-specific contrast agent could upstage patients with resectable pancreatic cancer on CT and could add valuable information to the diagnostic process,
thus allowing clinicians to determine the most appropriate treatment option for the patients,
avoiding unnecessary surgery in some cases.
Radiologically, PDAC might appear variously – imitating cystic lesion or inflammatory changes.
An appearance as a cystic lesion is shown in our second case.
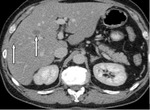
Case 2. 70 year old male with prostatic cancer; laboratory test within normal limits,
abdominal CT scan was performed for evaluation of spread of the disease (Figure 4).
Year 2017 03

Fig. 4: a) Axial CT image, arterial phase; b) Axial CT image, portal-venous phase
The report was given as “well circumscribed,
cystic lesion in the head of the pancreas”; for further evaluation - MRI was suggested.

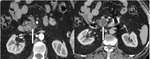
Fig. 5: MRI of the same patient. The lesion is hyperintense on T2W image (a), slightly hypointense on T1 W image (b); on DWI images the lesion shows no DWI restriction (c, d, e) and had high ADC value on ADC map (f).
No uptake of contrast material,
no mural nodes was identified on postcontrast images.
MRCP images showed no dilatation of MPD or CBD (Figure 6 a,b.) The report was given: ”cystic lesion in the head of the pancreas with no signs of malignancy”.
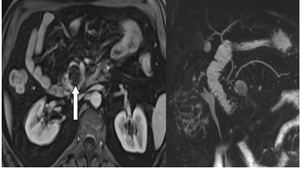
Fig. 6: a) T1_vibe image after contrast injection ; b)MRCP image MIP
The patient was left for further observation.

Fig. 7: Control CT images of the same patient in a year (year 2018 08) a) arterial phase shows a hypodense tumor, surrounding AMS with duodenal obstruction and dilatation of the stomach; b) and c) SMV is also involved. Dilatation of MPD is seen. We found T4 disease (locally advanced), it was proved to be poorly differentiated PDAC and the patient underwent paliative surgery.
Discussion: widespread use of computed tomography and ultrasound has led to the identification of increasing numbers of patients with incidental cystic lesions of the pancreas.
Most of these cysts are benign or low-grade neoplasms.
However,
it can sometimes hide a malignant process.
In study,
done by Carlos Fernandez-del Castillo et al.
[10],
212 patients with incidental cysts were evaluated.
Intraductal papillary mucinous neoplasms (IPMN) were found in 36% of the patients (53% of them had in situ or invasive cancer),
mucinous cystic neoplasm in 20.3% (26% of those had cystadenocarcinoma) and 14 (6.6%) patients were found with ductal pancreatic adenocarcinoma presenting as a cystic lesion.
In conclusion,
it is worth considering to observe patients with incidentally found cystic lesions in the pancreas more frequently and thoroughly.
Furthermore,
performing MRI DWI should be taken in consideration – searching for small foci with diffusion restriction behind cystic lesion,
indicating malignant process.
Case 3. 55 year old male with weight loss ~3 kg/6 months; laboratory tests: ↑CRP 38.
The patient underwent CT scan.

Fig. 8: Axial CT image; arterial phase; slightly hypodense lesion in the head of the pancreas is seen. No dilatation of MPD or CBD was detected.
The patient went for further evaluation with MRI.

Fig. 9: A) T2W image - slightly hyperintense mass in the head of the pancreas;
B) No dilatation of MPD or CBD was detected;
C) DWI b50;
D) DWI b400;
E) DWI b800;
F) ADC map;
MRI images. On DWI, the restriction of diffusion was questionable and ADC value was low.

Fig. 10: A)pre-contrast T1W image – a slightly hypointense mass in the head
of the pancreas; B) late phase – homogeneous uptake of contrast media is seen.
After MRI,
the report was given: focal form of pancreatitis is most likely diagnosis,
PDAC is less probable.
Discussion: the differentiation of focal pancreatitis and PDAC poses a diagnostic dilemma.
Both conditions may present with the same symptoms and radiological signs.
Complexity of differential diagnosis is difficult because PDAC is frequently associated with secondary inflammatory changes of surrounding parenchyma and chronic pancreatitis may develop into PDAC.
In many cases,
differential diagnosis using radiological methods is nearly impossible,
therefore endoscopic ultrasound with histological verification may be required.
Zhibing Ruan et al.
performed the study on 54 patients (24 mass-forming chronic pancreatitis (MFCP) and 30 pancreatic carcinoma(PDAC)) to identify features that may distinguish pancreatic carcinoma from mass-forming chronic pancreatitis [11].
All patients underwent CT,
MRI and PET/CT imaging .
Results shown that lesion size was higher in MFCP patients (5.44±1.27cm vs 3.34±1.23cm),
PC lesions showed increased lobulation compared to MFCP (83.3vs12.5%).
Also MFCP patients showed increased exudation around the lesion (83.33 vs.
13.33%),
pseudocyst formation (58.33 vs.
10%) and thickening of the right renal fascia (83.33 vs.
13.33%) than in the PDAC patients.
High signal on DWI was observed in 28.57% MFCP and 89.47% PDAC cases.
Moreover,
ADC values were higher in the MFCP than in the PC group.
In conclusion,
this study showed,
that in order to effectively differentiate PDAC from MFCP integration of CT,
MRI and PET/CT imaging methods are necessary.
Case 4. 45 year old male with weight loss ~6 kg/6 months and jaundice; laboratory tests: slightly elevated bilirubin; ↑CRP 50.
The patient underwent CT ,
followed by MRI with MRCP.

Fig. 11: A) Duct penetrating sign; B) Cystic degeneration of the duodenal wall; C) Late enhancement.
MRCP images (a,
b) we can see slightly dilated CBD with penetrating duct sign and also cystic degeneration of the duodenal wall – findings compatible with cystic form of paraduodenal pancreatitis.
In contrast enhanced image (c) we can see late enhancement of the paraduodenal space.
Discussion: Groove pancreatitis is a rare form of chronic pancreatitis affecting the “groove” between the superior aspect of the pancreatic head,
the duodenum and the common bile duct.
Groove pancreatitis has two forms.
The pure form affects only the pancreaticoduodenal groove (i.e.,
between the pancreatic head and duodenum),
whereas the segmental form is centered in the pancreaticoduodenal groove but also extends medially into the pancreatic head.
Differentiation of PDAC vs Groove pancreatitis can be extremely difficult (especially in solid form,
without cystic degeneration of duodenal wall),
and in many cases,
leads to surgery due to inability to reliably make this distinction.
This is particularly the case with malignancies,
which arise immediately adjacent to the groove itself and do not show the typical pancreatic ductal obstruction and upstream atrophy, presented with most PDAC’s.
However,
most PDAC’s do not show internal cystic changes and are much more likely to infiltrate posteriorly into the retroperitoneum and encase the vasculature (including the gastroduodenal artery) unlike Groove pancreatitis.
Nevertheless,
thickening of the medial duodenal wall - a common finding with Groove pancreatitis,
is quite uncommon in cases of PDAC [8].
The key: evaluation of the zone of transition from dilated to non-dilated or not visualized duct.
Abrupt termination of the bile duct and the absence of typical signs of biliary lithiasis have a high correlation with malignancy.
At the zone of transition; thin-section MR images should be obtained.
These are only 4 cases we encountered.
However,
PDAC has a lot more different “faces” radiologically.
To prevent pitfalls and possible mistakes,
we suggest to think about integrating different imaging modalities when diagnosis raises questions.
And do not forget,
that multidisciplinary approach in such cases is essential.