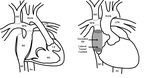
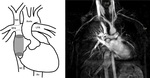
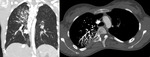
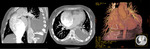
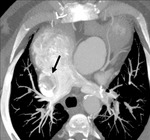
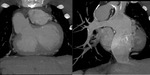
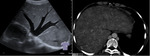
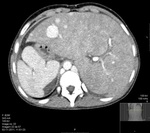
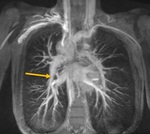
Normal Fontan Anatomy
The Fontan procedure refers to any surgical technique aimed at the complete separation of pulmonary and systemic blood flows,
redirecting the normal venous blood flow to the lungs and bypassing the right ventricle.
It is used in cases of complex congenital heart defects such as pulmonary valve atresia with intact ventricular septum,
tricuspid valve atresia,
mitral valve atresia,
hypoplastic left heart syndrome and double-inlet ventricle.
Since its first description by Fontan and Baudet in 1971,
the procedure has undergone numerous modifications.
Originally,
the procedure consisted on connecting the superior vena cava (SVC) to the right pulmonary artery (RPA),
a unidirectional “Glenn shunt”,
and connecting the right atrium (RA) to the left pulmonary artery (LPA).
The intended goal was to bypass only the right ventricle,
attempting to preserve a more physiological blood flow,
but the procedure often resulted in right atrial dilatation and consequent arrythmias,
vein compression and thrombosis.
In its latest iterations,
the Fontan circulation is established by connecting the superior and inferior vena cava directly to the pulmonary arteries,
bypassing both the right ventricle and atrium.
The circuit is created in two different stages,
allowing for a better adaptation to the new hemodynamic status.
At 4-12 months,
a hemi-Fontan is preformed and the SVC is connected to the RPA,
in a “end-to-side” anastomosis (the SVC cardiac “stump” is also connected to the RPA).
When the patient reaches 1-5 years of age the circuit is completed by attaching the inferior vena cava (IVC) to the RPA through a conduit that can run internally or externally to the right atrial chamber.
A small opening,
called “fenestration”,
is sometimes created between the conduit and the right atrium to relieve the pressure and prevent pulmonary volume overload (Fig.
1).
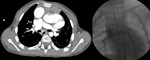
Imaging modalities such as MR and CT are invaluable tools for the long-term management of Fontan patients,
as they provide excellent anatomical and hemodynamical information for medical therapy or subsequent surgical interventions (Fig.
2 and 3).
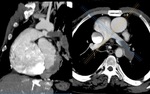
Special considerations in contrast-enhanced CT should be taken into account,
due to the extent of the circulatory alterations.
As a contrast agent is usually injected in the right upper arm,
it passes to the SVC and then to the RPA,
filling the homolateral veins first.
The LPA only enhances after the contrast reaches the IVC through the systemic circulation.
Consequently,
care should be taken in early arterial phase scans as they might be non-diagnostic (Fig.
4).
Complications
Despite boasting good results,
with survival rates around 85% at 20 years,
the Fontan procedure is palliative by design.
Complications arising from the procedure are all but inevitable and increasingly viewed in adults due to the increase of life expectancy with newer surgical techniques.
All patients (n=19) had at least one assessable complication,
apparent on the available imaging methods.
Cardiac Complications
The most common cardiac complications were atrial dilatation in 52,6% (n=10),
followed by ventricular hypertrophy/dilatation in 42,1% (n=8),
while 21,4% (n=4) experienced conduit complications,
including leaks,
stenosis and thrombosis.
Atrial thrombi were also visible in 15,8% (n=3)(Table 1).
• Atrial hypertrophy/dilatation and Atrial thrombus
The most common complication in the “classic” procedure,
right atrial dilation occurs due to constant exposure to elevated systemic pressure.
In our study,
10 patients presented with atrial dilatation,
9 of which underwent “classic” Fontan procedures,
as would be expected (the remaining case was a post hemi-Fontan patient with critical pulmonary stenosis)(Fig.
5).
The resulting stasis often leads to the development of atrial thrombus,
as it was seen in 3 patients.
Slow-flowing blood in the atrium can also cause “swirling artefacts” mimicking thrombus,
a pitfall of CT imaging (Fig.
6).
• Left Ventricular Hypertrophy/Dilatation
Chronic volume overload,
present even before the procedure,
often leads to ventricular hypertrophy and dilatation.
This was observed in 7 patients.
The bypass of the right side of the heart causes a severe preload reduction to the systemic ventricle,
that in turn results in systolic and diastolic failure.
In some cases,
the original malformation is also a predisposing factor for ventricular dysfunction (for instance,
when the systemic ventricle is also the morphological right ventricle or in indeterminate primitive ventricle) (Fig.
7).
• Conduit Complications
The intraarterial or extracardiac conduits,
used to direct flow from the IVC to the pulmonary arteries in modern Fontan procedures,
may also be a site for complications.
These include leaks and stenosis,
adjacent to the anastomosis,
or intra-conduit thrombosis.
Our study found 4 patients with such complications: 2 cases of thrombosis,
1 leak and 1 stenosis.
Extracardiac complications
The most common extracardiac complication was chronic hepatic congestion occurring in 84,2% of patients (n=16),
followed by pulmonary artery stenosis in 31,6% (n=6),
arteriovenous malformations in 31,6% (n=6),
hepatic nodules in 26,3% (n=5),
major thromboembolic events in 21,1% (n=4) and lymphatic system dysfunction (n=3) 15,8%.
(Table 2)
• Chronic hepatic congestion and hepatic nodules
In the present study,
the most common complication was chronic hepatic congestion,
occurring in 16 patients.
This is caused by an increase of systemic venous pressure,
directly transmitted to the liver parenchyma with consequent congestion and dilatation of sinusoids,
atrophy of hepatocytes,
progressive fibrosis and cardiac cirrhosis.
Patients exhibited hepatomegaly and IVF/hepatic vein dilatation,
cirrhotic livers and signs of portal hypertension to various degrees.
While parenchymal perfusion alterations like “nutmeg” or “Budd‐Chiari syndrome-like” liver are widely reported,
they were only found in 6 cases,
probably due to the absence of liver specific contrast enhanced studies (Fig.
8).
Hypervascular nodular lesions were found in 5 patients,
as expected in chronic hepatic congestion.
They were consistent with either nodular regenerative hyperplasia (NRH) or focal nodular hyperplasia (FNH),
though histological confirmation was only present in two cases (both cases of NRH).
Hypervascular nodules located in the periphery are often hyperplastic and may indicate a failing Fontan.
Hepatocarcinoma should be excluded,
as it has been reported in these patients (Fig.
9).
• Arteriovenous Malformations
Arteriovenous malformations were present in 6 patients.
Post-Fontan patients are at an increased risk of developing such malformations,
creating right-to-left shunts and contributing to cyanosis.
They have been attributed to the lack of pulsatile blood flow and the underfilling of the pulmonary vascular bed (Fig.
10).
• Pulmonary artery stenosis
Morphological alterations in the pulmonary arteries,
namely artery stenosis,
are commonly observed,
usually at the anastomosis site.
This complication was present in 6 patients.
Detection and early treatment are very important as stenosis negatively affects pulmonary vascular resistance,
a determinant factor in post-Fontan cardiac output (Fig.
11).
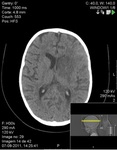
• Major Thromboembolic Events
Four patients experienced thromboembolic complications: 4 patients had pulmonary embolism (one patient had 3 reported episodes) and 1 experienced a stroke,
(following the initial pulmonary embolism).
Dilatated atrium and low cardiac output,
associated with hepatic congestion and cyanosis-related polycythaemia are believed to be amongst the etiological factors responsible.
(Fig.
12 and 13).
• Lymphatic System Dysfunction
The Fontan circulation places a significant amount of pressure on the lymphatic system.
In our study,
chylothorax was reported in 1 patient,
and chylopericardium in 1 patient.
A more uncommon complication,
protein losing enteropathy,
was also reported in 1 case.
Plastic bronchitis,
a mucinous obstruction of the airways,
is a rare reported complication,
not present in our study.