Indications and Outcomes
Stereotactic ablative radiotherapy (SABR) is delivered in several fractions,
typically 3-8,
and precisely administers high doses of radiation,
in excess of 100 Gy,
to the lung tumour over 1-3 weeks in an outpatient setting (3,
4).
Complex beam arrangements reduce the impact on adjacent normal lung and mediastinal structures (3).
SABR is indicated in early stage NSCLC in patients who are unfit for or decline surgery and in some cases of recurrent NSCLC (5).
SABR has been reported to obtain locoregional control in greater than 90% of patients (6,
7) and many studies have found no significant difference in rates of local recurrence,
disease-specific or overall survival between SABR and surgery (8,9).
Radiological appearances post SABR
Lung changes post SABR differ from conventional radiotherapy due to complex beam arrangements which deliver high doses to the affected tissue with relatively low doses to the surrounding normal lung.
This is in contrast to conventional radiotherapy which results in a characteristic appearance of fibrosis with straight borders as a result of conventional treatment field edges (10).
CT appearances of the lungs post SABR can be divided into acute changes,
representing pneumonitis (<6 months post treatment) and late changes,
representing fibrosis (>6 months post treatment) (11,
12).
These changes are displayed in Table 2.
Median time to acute changes is 17 weeks (4) and 62% of patients will develop some form of acute change,
the majority of which is consolidation (45%) (13).
Modified conventional fibrosis is the most common late finding on CT (13).
Table 1.
CT changes post SABR
|
Acute (<6 months)
|
Late (>6 months)
|
|
Diffuse Consolidation
|
Modified Conventional Fibrosis
|
|
Patchy Consolidation
|
Mass-like Fibrosis
|
|
Diffuse Groundglass Opacification
|
Scar-like Fibrosis
|
|
Patchy Groundglass Opacification
|
No Change
|
|
No Change
|
|
Acute Radiological Changes/Pneumonitis (12):
1)Diffuse consolidation. Homogenous increased lung attenuation obscuring the bronchi and vasculature which involves an area of greater than 5cm.
There is more consolidation than aerated lung in the affected are.
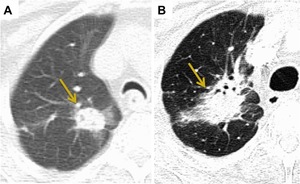
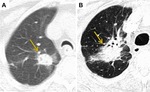
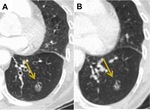
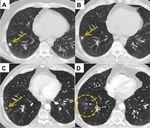
Fig. 1: Diffuse Consolidation. (A) CT Thorax shows a 2.5 cm right upper lobe lepidic-predominant adenocarcinoma. (B) 3 months post SABR CT demonstrates a large focus of airspace opacification with indistinct bronchi and vasculature at the site of the primary tumour consistent with diffuse consolidation.
2) Patchy consolidation.
Patchy areas of increased lung attenuation less than 5cm in diameter.
There is less consolidation than aerated lung in the involved region.
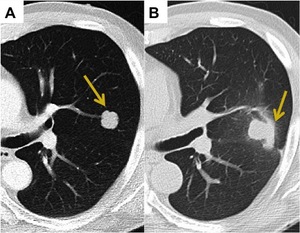
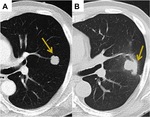
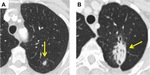
Fig. 2: Patchy Consolidation. (A) CT Thorax shows a 2 cm left lower lobe adenocarcinoma. (B) CT 4 months post SABR shows little change in the overall size of the tumour however there has been interval development of increased opacification laterally consistent with patchy consolidation.
3) Diffuse Groundglass Opacification (GGO). Hazy increased lung attenuation with preservation of bronchial and vasculature margins which involves an area of greater than 5 cm.
There is more GGO than aerated lung in the affected area.
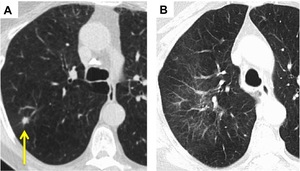
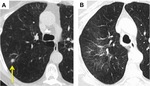
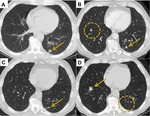
Fig. 3: Diffuse Groundglass Opacification. (A) 1 cm right upper lobe squamous cell carcinoma. (B) CT 3 months post SABR demonstrates diffuse groundglass opacification at the site of the treated tumour.
4) Patchy Groundglass Opacification (GGO).
Hazy increased lung attenuation with preservation of bronchial and vasculature margins which involves an area of less than 5 cm.
There is less GGO than aerated lung in the affected area.
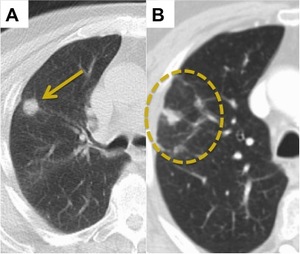
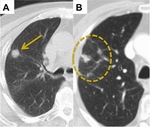
Fig. 4: Patchy Groundglass Opacification. (A) 1.5 cm right upper lobe adenocarcinoma. (B) CT 3 months post SABR reveals reduction in the size of the tumour with surrounding patchy GGO.
5) No change. No increasing density.
Includes stable appearance of the primary tumour,
resolution or reduction in size of the tumour or fibrosis at the site of the primary tumour which does not exceed the size of the primary tumour.
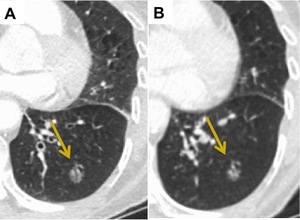
Fig. 5: No change. (A) 2cm left lower lobe lepidic-predominant adenocarcinoma. (B) CT 3 months post SABR demonstrates a subtle decrease in the size of the lesion without a change in density.
Late Radiological Changes/Fibrosis (11):
1) Modified Conventional Pattern. Consolidation,
bronchiectasis and volume loss similar to conventional radiation fibrosis but usually less extensive.
Affects a larger area than the original tumour size and may have associated GGO.
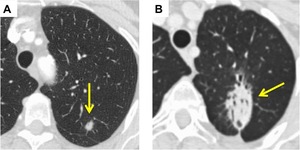
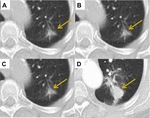
Fig. 6: (A) 1 cm left upper lobe adenocarcinoma. (B) CT 18 months post SABR demonstrates modified conventional fibrosis with minimal volume loss, consolidation and bronchiectasis at the site of the treated tumour.
2) Mass-like Fibrosis. Well circumscribed consolidation limited to the region of the treated tumour but larger than the size of the primary tumour.
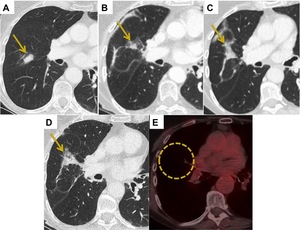
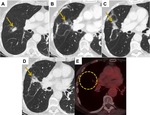
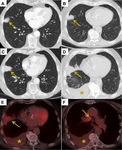
Fig. 7: Mass-like Fibrosis. (A) 2 cm right middle lobe adenocarcinoma. (B-D) Increasing mass at the site of the previous tumour on 6, 12 and 18 monthly post treatment CT scans. (E) PET CT at 19 months demonstrated no abnormal uptake. Findings are consistent with mass-like fibrosis.
3) Scar-like Fibrosis. Linear opacity in the area of the treated tumour with associated volume loss.
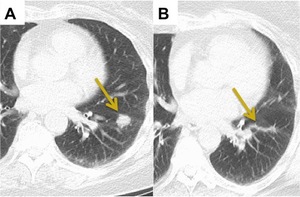
Fig. 8: (A) 1.5 cm left lower lobe adenocarcinoma. (B) Linear scar-like focus at the site of the left lower lobe tumour which underwent SABR 2 years previously.
4) No change. No evidence of increasing density.
Includes stable appearance of the primary tumour,
resolution or reduction in size of the tumour or fibrosis at the site of the primary tumour which does not exceed the size of the primary tumour.
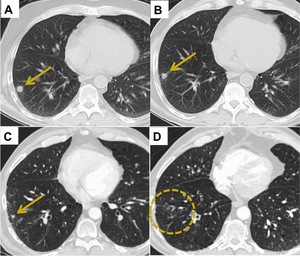
Fig. 9: Complete resolution. (A) 1 cm primary right lower lobe adenocarcinoma. (B+C) Gradual reduction in size of the tumour at 12 and 18 months post SABR. (D) Complete resolution of the primary tumour at 24 months.
Recurrence
There is no internationally accepted protocol for follow up imaging post SABR.
The National Comprehensive Cancer Network (NCCN) recommends a CT every 6-12 months for the first two years and yearly thereafter (14).
More recently published literature however recommends a more intensive surveillance protocol,
for example,
conventional CT at 3 montly intervals for the first 2 years post treatment (15).
PET CT has been found to be a specific but insensitive test for local recurrence in the acute phase and it is recommended that conventional contrast enhanced CT should be used for surveillance (16).
PET CT can then be used as an adjunct when there is suspected recurrence on conventional CT.
While recurrence within the SABR field is uncommon and post treatment CT changes are very common,
it is important that the radiologist be able to distinguish them on imaging as those with recurrence may benefit from salvage treatment,
including surgical resection (17).
Assessing for recurrence is difficult as the standard post treatment changes,
particularly mass-like fibrosis,
mimic recurrence and the changes are known to evolve over time (18).
Recurrence occurs in approximately 18% of patients and most commonly involves out of field or distant recurrence (66%).
Locoregional recurrence accounts for the remaining 34% of patients (19).
Second primary tumours occur in 6-8% of patients (19,
20) (Figure 10).
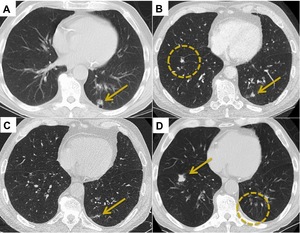
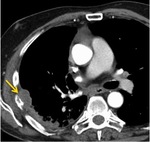
Fig. 10: (A) CT Thorax demonstrates a 1.5 cm left lower lobe squamous cell carcinoma. (B) CT 6 months post SABR demonstrates reduction in the size of the treated tumour (arrow). There is a new right perifissural nodule (circle). (C) CT 9 months post SABR demonstrates almost complete resolution of the primary tumour. The right perifissural nodule is less conspicuous. (D) 12 months post SABR. The left lower lobe tumour has completely regressed. Unfortunately there has been a significant increase in size of the right perifissural nodule consistent with a second primary lung cancer.
There are no completely reliable signs of local recurrence on follow up contrast enhanced CT.
An enlarging opacity,
bulging margins,
filled in bronchogram,
new effusion,
new mass,
and increasing density have all been described as high risk features of local recurrence however several of these are also seen in evolving post treatment change.
An enlarging mass and bulging margins have both been reported to have a high specificity for recurrence (20,
21) and the presence of multiple high risk features increases the specificity for recurrence (21).
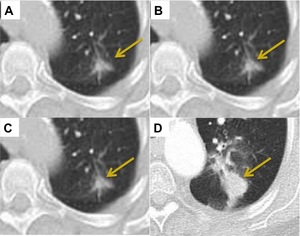
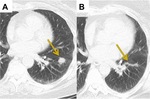
Fig. 11: Recurrence. Increasing opacity at the site of a previously treated 3cm left upper lobe adenocarcinoma over a period of 12 months consistent with recurrence.
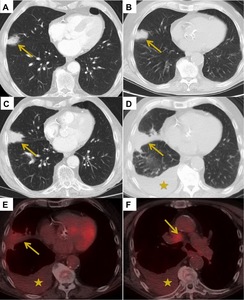
Fig. 12: Recurrence. (A) 3 cm right middle lobe squamous cell carcinoma which underwent SABR. (B+C) 6 and 12 month post treatment CT demonstrates gradual increase in size of the primary lesion. (D) CT at 18 months demonstrated further increase in size of the tumour with interval development of a right pleural effusion (star). (E) PET CT was arranged and showed mild increased uptake in the primary tumour and the presence of the pleural effusion. (F) PET CT also identified an FDG avid right paratracheal lymph node. Percutaneous sampling of the effusion confirmed recurrent squamous cell carcinoma.
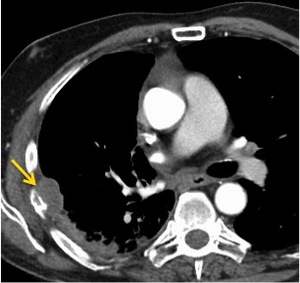
Fig. 13: Recurrence. CT 3 years post SABR for a right upper lobe squamous cell carcinoma revealed a right pleural based soft tissue mass with associated rib destruction consistent with recurrence.
Complications
A list of complications associated with SABR are displayed in Table 2 (22).
Table 2.
Complications post SABR.
|
Toxicity
|
Manifestation
|
|
Airway Toxicity
|
Atelectasis
|
| |
Stenosis/stricture
|
| |
Necrosis
|
| |
Fistula formation
|
|
Pulmonary Toxicity
|
Spontaneous pneumothorax
|
| |
Pneumonitis
|
|
Oesophageal Toxicity
|
Oesophagitis
|
| |
Stricture
|
| |
Perforation
|
| |
Tracheo-oesophageal fistula
|
|
Vascular Injury
|
Pulmonary haemorrhage/Haemoptysis
|
| |
Aortic rupture
|
| |
Aortic aneurysm
|
| |
Aortic dissection
|
|
Chest wall and Skin Toxicitiy
|
Chest wall pain
|
| |
Rib fracture
|
| |
Radiation skin burn
|
|
Nerve Toxicity
|
Brachial plexus injury
|
| |
Vagus nerve injury
|
Centrally located tumours,
those within 2cm of the proximal bronchial tree,
are at higher risk of toxicity,
particularly airway,
oesophageal and vascular toxicities.
Pneumonitis is one of the major limiting toxicities to maximal radiation dose with up to 30% of patients developing clinically relevant pneumonitis.
Chest wall and skin toxicity is associated with peripherally located tumours however tumours with a distance of greater than 2 cm from the chest wall are generally associated with a low chest wall toxicity rate.
Injury to the brachial plexus or vagus nerves,
likely due to demyelination,
is a concern in apical tumours (brachial plexus and right vagus nerve) and medial left upper lobe tumours (left vagus nerve) (22) .