We review the MRI request to avoid request or appointment errors.
The patient's clinical history is then reviewed to evaluate if he or she has any device or implant that contraindicates the MRI examination.
Joint prostheses and common surgical materials are usually compatible.
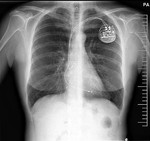
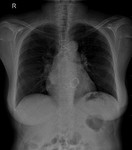
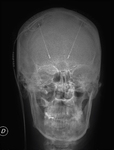
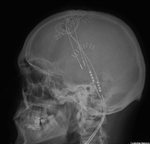
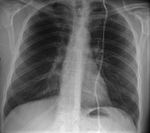
We pay special attention to devices such as pacemakers,
implantable cardioverter defibrillator (ICD),
cardiac valves,
cochlear implants,
neurostimulators and any other devices that may be incompatible with MRI.
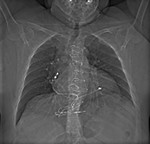
In the case of pacemakers and ICD we need to know if the device is compatible,
the cardiac leads,
if there are disconnected leads or epicardial leads.
Cardiologist connect the compatible pacemaker on a MRI mode before the test and review the device once the study is completed.
ICD must be stopped,
the cardiologist is present during the study to act if any complication happens.
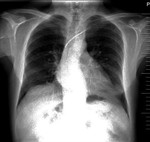
For the heart valves the model is checked for compatibility.
All compatible neurostimulators and subcutaneous devices must be disconnected or placed in MRI mode before the scan and must be reconnected at the end by the specialist.
For all other devices we need information about the model to check the compatibility with MRI.
The technologist (radiographer) does the review process,
supervised by the radiologist.
Patient and Radiologist sign the consent for the study.
Results
We evaluated from November 2017 to January 2018 all the MRI request’s ordered to the radiology department and the incidents related to MRI.
Patient’s medical records were reviewed to detect the following devices or safety related issues:
Implant of cardiac valves,
pacemakers,
implantable cardioverter defibrillator (ICD),
cochlear implants,
neurostimulators,
joint prostheses,
or any other devices susceptible of being MRI incompatible,
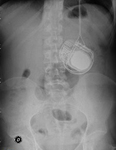
subcutaneous devices like pumps of insulin,
heparin,
etc.
erroneous request,
MRI contrast contraindication and claustrophobic or large volume patients are.
3114 MRI requests were reviewed during this period 40 incidents were detected,
this represents 1.28% of the total number of cases.
The principal causes are:
1-Patients with implantable or non-implantable devices,
not referred in the medical request and/or unknown brand or model 75%
2-The request is confusing or not well specified.2.5%
3-Contraindication for contrast administration.12,
5%
4-Claustrophobic or large volume patients.7.5%
5-Recent surgery.2.5%
The study was cancelled in 10% of the patients reviewed.
The reasons were Claustrophobia 2.5%,
non MRI compatible pacemaker 5% and recent surgery 2.5%