NORMAL ANATOMY
- Pancreas is a retroperitoneal organ opposite the first lumbar vertebral body located in the anterior para renal space consisting of an uncinate process,
head ,
neck,
body and tail.
- The uncinate process and head are located within the duodenal C loop.
- The neck is a small area of constriction anterior to the superior mesenteric artery and vein and retro-pancreatic part of portal vein.
- The body courses obliquely upwards towards the left side with the splenic vein and artery running along its posterior surface.
- The tail is totally covered by the peritoneum and directed towards the splenic hilum.
- Pancreas is drained by two ducts –
- Duct of Wirsung or main pancreatic duct draining the tail,
body,
neck and part of head into the major duodenal papilla along with common bile duct usually via ampulla of Vater
- Duct of Santorini or accessory pancreatic duct draining part of head into the minor duodenal papilla
MODALITIES AVAILABLE FOR PANCREATIC IMAGING:
Ultrasound (USG)-
Transabdominal sonography- usually the first investigation ordered as easily available and not expensive
- Normal pancreas appears homogenous and slightly more echogenic as compared to the liver.
- Can detect pancreatic head masses (appear hypoechoic to the pancreatic parenchyma) causing ductal and biliary radicles dilatation
- But has limited role- pancreas can be obscured by bowel gas,
high operator dependability.
Endoscopic US (EUS)- highly sensitive in detecting masses as small as 0.2–0.3 cm,
local tumor staging and can also aid in its biopsy.
EUS can clarify equivocal findings at CT or MR imaging but has the limitations of being highly operator dependent with a steep learning curve,
having a narrow field of view and provides little information about distant spread.
Computed Tomography:
Contrast-enhanced MDCT (CECT) is currently the most commonly used modality for the diagnosis and staging of pancreatic cancer.
The scanning protocol specifically for pancreas needs to be optimized as the organ receives most of the blood supply in the arterial phase as compared to liver which shows enhancement more in the venous phase.
Also it needs to be tailored according to the indication and the patient factors further but broadly includes-

Table 1: Optimal scanning protocol for pancreas for Computed Tomography
Pitfalls of MDCT include-
- Lower accuracy in detection of small lesions
- Poor diagnostic power in differentiating between a residual/recurrent tumor,
and post treatment changes
- Significant radiation exposure especially when follow up scans are needed
- MRI,
MRCP,
and EUS are superior for evaluation of cystic neoplasms,
ductal injuries,
and chronic pancreatitis
Magnetic Resonance Imaging (MRI)
MRI including Magnetic Resonance Cholangiopancreatography (MRCP) are a comprehensive,
accurate,
safe and non-invasive modalities for evaluation of pancreatic parenchyma,
ductal system,
peripancreatic soft tissue,
vessels and cystic lesions.
A combination of T1-weighted,
T2-weighted,
Fat saturation with early and late post-gadolinium-enhanced images,
MRCP and MR angiography (MRA) are used usually for optimal pancreatic assessment.
- Normal pancreas has high T1 signal due to high aqueous protein
- Pancreatic tumours being desmoplastic,
even a small focal hypointense lesion can be detected
- T2-weighted images: useful for anatomic delineation of the ductal system,
characterization of the nature of fluid in the cystic lesions
FINDINGS-
NEOPLASTIC LESIONS:
PANCREATIC ADENOCARCINOMA
- Most common tumour of the pancreas
- Is aggressive showing early local and distant spread
- Poor prognosis with only 10–20% of patients having a surgically resectable disease at the time of diagnosis and the overall 5-year survival rate is of only 5%.
- Presents with a vague symptomology- abdominal pain,
weight loss late in the course of disease or obstructive jaundice when pancreatic head is involved
- Twice as common in males as females and occurs in elderly
Imaging features
- Most commonly arise from head or uncinate process
- On ultrasound- appear hypoechoic as compared to the rest of the pancreatic parenchyma
- On CECT- seen as a poorly enhancing focal area within the densely enhancing normal pancreatic tissue.
- Classically described direct sign of tumour is the presence of a hypodense/hypointense mass within the pancreas on cross sectional imaging.
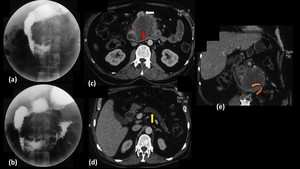
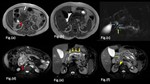
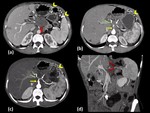
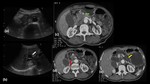
Fig. 22: Adenocarcinoma of pancreatic body with duodenal invasion.
Barium meal spot films (a,b) show widening of c-loop of duodenum with irregularity and loss of normal mucosal pattern in the 3rd part of duodenum. Axial and Coronal contrast enhanced computed tomography images (c,d,e) show a well defined hypoenhancing lesion involving the uncinate process of pancreas (Red arrow in c) causing atrophy of distal pancreas with dilated main pancreatic duct (Yellow arrow in d). The lesion is invading into the lumen of 3rd part of duodenum (Curved arrow in e) and is displacing the superior mesenteric vessels anteriorly (White arrow in c) forming less than 180 contact with them.
- Other secondary signs which give us a clue about the presence of a pancreatic lesion are:
- Biliary duct dilatation when the mass is in the pancreatic head and involves the common bile duct (CBD)
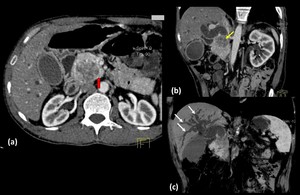
Fig. 15: Adenocarcinoma Head of pancreas
Axial (a) and oblique coronal (b and c) contrast enhanced computed tomography images show a well defined, heterogeneous, hypoenhancing lesion in the head of pancreas which shows a peripheral rim of hyperenhancement (red arrow in a). The lesion is causing widening of the C-loop of duodenum and is infiltrating into the distal common bile duct (yellow arrow in b) causing its abrupt narrowing and gross dilatation of proximal duct and intrahepatic biliary radicles (white arrows in c). The lesion is abutting the superior mesenteric vein, however no encasement or luminal compromise is seen.
- Main pancreatic duct (MPD) dilatation distal to the mass
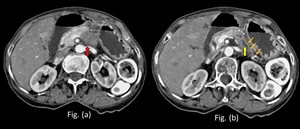
Fig. 18: Adenocarcinoma in a 68 year old patient:
Contrast enhanced computed tomography axial sections (a) and (b) at the level of the pancreas reveal a hypo-enhancing lesion in the pancreatic body region (red arrow in Fig. a) with dilatation of distal main pancreatic duct (yellow arrow in Fig. b) and distal parenchymal atrophy of the rest of the body and tail (multiple thin orange arrows in Fig. b).
- Double duct sign i.e.
dilatation of both CBD and MPD seen in pancreatic head masses
- Atrophy of the gland distal to the lesion
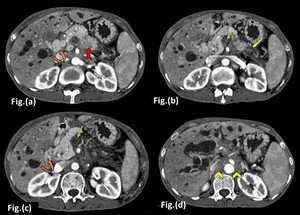
Fig. 16: Adenocarcinoma in a 47 year old female
Axial contrast enhanced CT images in the arterial phase show an ill defined hypo-enhancing lesion in the pancreatic body region (Red arrow in a) with dilatation of distal main pancreatic duct and distal parenchymal atrophy (Yellow arrow in b). Non enhancing thrombus is seen within the superior mesenteric vein causing its non-opacification (Green arrow in b,c). Portal vein appears to be replaced by multiple dilated venous channels (Curved arrow in a,c) showing filling in the porto-venous causing significant intrahepatic biliary radicle dilatation suggestive of a portal cavernoma. Ill defined non-enhancing soft tissue density is seen in the retroperitoneum surrounding the abdominal aorta and its branches suggestive of retro peritoneal fibrosis (yellow arrow head in Fig. d).
- A large mass can lead to an abnormal convex contour of the gland
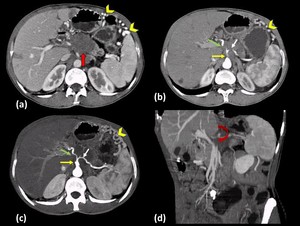
Fig. 20: Adenocarcinoma of pancreas in a 53 year old male.
Axial contrast enhanced CT images (a and b) show an ill-defined hypoenhancing lesion seen involving the neck and body of the pancreas (Red arrow in a) encasing the distal trunk of coeliac axis (Yellow arrow in b,c), proximal part of common hepatic artery (Green arrow in b,c) and splenic artery (White arrow in b,c) causing circumferential luminal narrowing, better seen on axial maximum intensity projection image (c). Oblique coronal CT image (d) shows invasion and obliteration of the splenic vein (Curved arrow in d) causing development of multiple venous collaterals in the perigastric and perisplenic area (Arrow heads in a,b and c)
- Normal pancreas has a lobulated outline- its loss can be an early sign for a hidden pancreatic mass
- Shows early vascular invasion and in the absence of distant metastatic disease,
it is the most important criteria for unresectability.
Tumour infiltration causes loss of the perivascular fat planes and indistinct vessel margins,
calibre change,
shape deformity,
thrombosis,
and development of collateral vessels.
This sometimes can be seen in the superior mesenteric vein which becomes teardrop shaped from its normal round shape on axial images due to tumor infiltration or peritumoral fibrosis,
is called “tear drop sign”
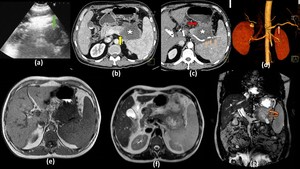
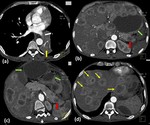
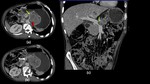
Fig. 13: Adenocarcinoma in a 76 year old man with abdominal pain and weight loss:
Ultrasound image (a) shows evidence of a hypoechoic solid lesion involving the body of pancreas (Green arrow in a). Axial contrast enhanced images in the pancreatic phase show a hypoenhancing mass in the pancreatic body (Star in b and c) which is extending into the peripancreatic region with loss of fat planes with the posterior stomach wall (Red arrow in c). The lesion is invading into the splenic vein (Yellow arrow in b) causing its non opacification and is seen encasing the splenic artery causing significant circumferential long segment luminal narrowing (Multiple thin orange arrows in c), which is seen better in the volume rendering technique image (d). The lesion is hypointense on T1-weighted MR image (e) and is heterogeneously hyperintense on T2 weighted MR image (f). Invasion into the gastric mucosa (Curved arrow in g) is confirmed in the coronal T2 weighted MR image (g). Note made of multiple gall stones.
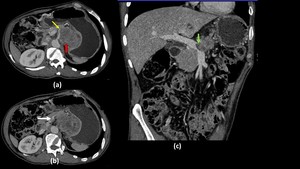
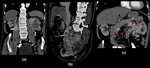
Fig. 21: Pancreatic adenocarcinoma in a 81 year old patient
Axial (a and b) and oblique coronal (c) contrast enhanced CT images shows a heterogeneously hypo-enhancing mass lesion in the pancreatic body (Red arrow in a) encasing the superior mesenteric artery (Yellow arrow in a) and invading into the splenic vein with the tumour thrombus extending into its confluence with superior mesenteric vein to form portal vein (Green arrow in c). Superior mesenteric vein appears partly compressed (White arrow in c) but shows normal opacification in a slightly more cranial section level (b) demonstrating tear drop sign.
- Metastasis commonly occurs to liver and peritoneum,
which may be assumed if abdominal ascites is present.
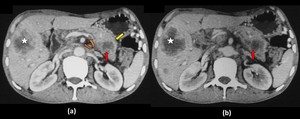
Fig. 14: Adenocarcinoma in a 65 year smoker:
Contrast enhanced computed tomography axial sections (a) and (b) of the upper abdomen in the porto-venous phase show evidence of an ill defined hypo-enhancing lesion in the pancreatic body region (red arrow) causing an abnormal convex contour of the pancreas (yellow arrow) with invasion and thrombosis of the splenic vein(curved arrow) and its non opacification distally in image (a). An ill defined hypoattenuating lesion is also seen in the liver (star) showing central necrotic areas suggestive of metastases.
- MR imaging is very accurate in detection and staging with a better contrast resolution than CT showing a hypo-enhancing T1- and T2-hypointense lesion on MR due to its desmoplastic nature with an enhancing thin peritumoral rim which guides towards disease focality.
Staging
TNM Staging of Pancreatic Cancer
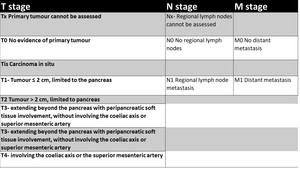
Table 6: TNM Staging of Pancreatic Cancer
On cross sectional imaging,
increased lymph node size is taken as the main criteria for predicting nodal status which poses a problem in their staging as they may be involved in pancreatic adenocarcinoma without enlargement.
Resectability criteria of tumor:
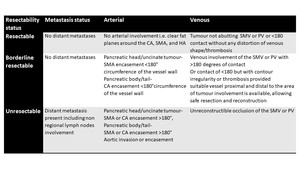
Table 5: Criteria for Defining the Resectability Status of Pancreatic Adenocarcinoma according to NCCN guidelines
Treatment
In the surgically resectable tumours,
distal pancreatectomy or radical pancreaticoduodenectomy i.e.
Whipple’s procedure is performed which itself is associated with significant morbidity and mortality necessitating careful preoperative local staging to avoid unnecessary laparotomies.
For palliation,
endoscopic biliary stenting can be done to relieve biliary obstruction.
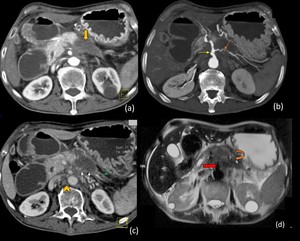
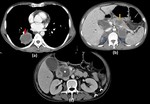
Fig. 12: Adenocarcinoma of pancreas with peripancreatic spread
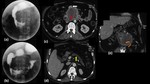
Contrast enhanced computed tomography image of upper abdomen in the arterial phase (a) shows a well defined hypoenhancing mass involving the body of pancreas (Thick yellow arrow in a) with peripancreatic extension. Maximum intensity projection image (b) shows encasement of celiac axis (Yellow arrow) and splenic artery (Orange arrow) with significant circumferential long segment luminal narrowing. Obliteration of splenic vein (White thin arrows in c) associated with atrophy of tail of pancreas & prominent distal pancreatic duct (Green arrow in c). Multiple enlarged lymph nodes are noted in the retroperitoneum (Arrow head in c). The lesion is hypointense (Curved arrow in d) on T2 weighted magnetic resonance imaging (d) and encasement of celiac axis is well seen (Red arrow in d). These constellation of imaging findings is highly suggest Pancreatic adenocarcinoma. The diagnosis was confirmed on Fine needle aspiration cytology.
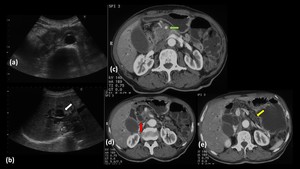
Fig. 23: Head of pancreas adenocarcinoma in a 49 year old female.
Ultrasound images (a,b) shows a hypoechoic lesion in the region of head of pancreas which is causing gross dilatation of CBD and intrahepatic biliary radicles (White arrow in b) . Axial contrast enhanced CT images (c,d,e) show an ill defined hypoenhancing lesion involving the head of pancreas causing dilatation of CBD (Red arrow in d) and IHBRD. Pancreatic body and tail appears atrophied and shows prominent MPD (Yellow arrow in e). The lesion is encasing the superior mesenteric artery (Green arrow in c) and is invading and obliterating the superior mesenteric vein.
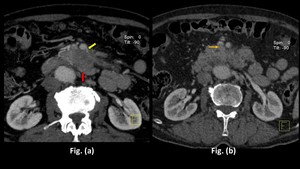
Fig. 17: Adenocarcinoma in a 62 year old patient detected incidentally:
Contrast enhanced computed tomography axial sections through the upper abdomen in the porto-venous phase shows evidence of a hypo-enhancing lesion in the pancreatic body(red arrow in Fig. a) abutting the superior mesenteric artery and encasing its branch in image (yellow arrow in Fig. a), with invasion into the superior mesenteric vein (thin orange arrow in Fig. b)
 NEUROENDOCRINE TUMOUR (NET)
NEUROENDOCRINE TUMOUR (NET)
- Previously known as ‘islet cell tumours,’ responsible for 1%–5% of all pancreatic tumors
- Usually present in the 6th decade
- Can be associated with- multiple endocrine neoplasia type 1,
von Hippel–Lindau syndrome,
neurofibromatosis type 1,
and tuberous sclerosis
- Classified into functioning or non functioning depending upon whether any active hormone is produced by the tumour.
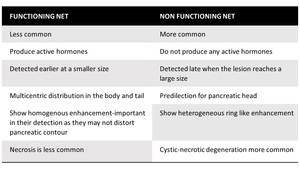
Table 4: Features of Functioning and Non-functioning Neuroendocrine tumours
Depending on the type of hormone synthesized Functioning NET are further classified as-
INSULINOMA-
- most common NET
- Present with- hypoglycemic attacks due to hyperinsulinemia,
atypical seizures
- Usually solitary,
benign,
most being less than 2 cm in size.
- Can be completely resected surgically thus primary aim of imaging is localization.
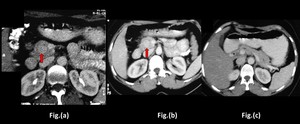
Fig. 8: Insulinoma in a 50 year female presenting with syncopal attacks
Axial Contrast enhanced Computed Tomography images show a small, well defined, homogenously hyperenhancing lesion within the head of pancreas (red arrow in a and b) with no obvious dilatation of common bile duct and main pancreatic duct in image (c). No obvious features of malignant degeneration seen.
GASTRINOMA-
- Second most common functioning NET
- Malignant in 60 % of cases
- Secrete gastrin thus present with Zollinger-Ellison syndrome- gastric hyperacidity,
recurrent gastric and duodenal ulceration; malabsorption and diarrhoea
- Localised to ‘gastrinoma triangle’,
bounded superiorly by junction of cystic and common hepatic ducts,
inferiorly by junction of second and third parts of duodenum,
and medially by pancreatic body
GLUCAGONOMA-
- Uncommon tumors
- Show necrolytic migratory erythema and hyperglycaemia
Others- VIPoma,
Somatostatinoma,
Corticotropinoma
Imaging features
- With increase in tumour size,
risk of malignancy increases
- On MDCT,
small tumors are generally solid and homogeneous,
and larger being heterogeneous with cystic-necrotic degeneration.
Calcification is more common with a lesser propensity for ductal obstruction as compared to adenocarcinoma.
- Are hyper-vascular showing intense rapid enhancement during the arterial phase
- Can show- local spread,
vascular invasion and tumour thrombus,
lymph node involvement,
and metastases,
having an enhancement pattern similar to that of the primary tumor
- On MR,
have low signal intensity on T1-weighted and intermediate-high signal intensity on T2-weighted images showing intense enhancement
- Indium 111 octreotide SPECT/CT is taken up by most NETs except insulinoma,
can be used for whole-body scanning for the detection of small tumors and metastases
- FDG PET can be used for assessment of poorly differentiated NETs,
which are octreotide negative.
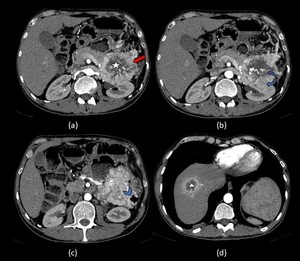
Fig. 7: Non functioning Neuro endocrine tumor of pancreas with liver metastasis:
Axial contrast enhanced CT images in the arterial phase (a) show a large, well defined, hyperenhancing lesion involving the tail of pancreas (Red arrow in a) showing central cystic necrotic degeneration (star). The lesion is seen infiltrating the left kidney (Curved arrow in b) and the descending colon (Arrow head in c). A lesion with similar image morphology is seen in the right lobe of liver (Yellow arrow in d), suggestive of metastases.
PANCREATOBLASTOMA
- most common pancreatic tumor of young children
- Seen in first decade with a predilection for boys; If seen in adults,
is more aggressive
- Cystic pancreatoblastoma is associated with Beckwith Wiedmann syndrome and presents earlier
- Usually slow growing thus manifests as a large asymptomatic mass or abdominal pain,
early satiety,
vomiting,
and constipation due to mass effect
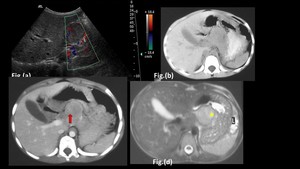
Fig. 9: Pancreato-blastoma in a 6 year old boy:
Ultrasound image (a) shows a well defined heteroechoeic lesion involving the body of pancreas abutting the inferior surface of liver showing internal vascularity on colour doppler.
On cross sectional imaging, a well circumscribed, smooth marginated mass is seen which is iso-dense to the normal pancreas in the non contrast image (b) and is heterogeneously hyperintense on the axial T2 weighted MR image (d). The lesion has internal solid (Red arrow in c) and cystic components (Star in d) and enhancement of solid components is seen on the contrast enhanced axial CT image (c). The lesion is heterogeneously hyperintense on the T2 weighted axial Magnetic resonance image (d)
Imaging features
- Due to its large size at presentation sometimes organ of origin may be difficult to identify,
thus biopsy may be required to differentiate from other paediatric tumors
- More common in head region
- USG-appears as a well circumscribed,
heterogeneous mass with solid and cystic spaces and intervening hyperechoic internal septa
- CECT- appears as a well/partially circumscribed heterogeneous mass with internal cystic areas due to necrosis,
with smooth lobulated outline and enhancing septa.
- Can have small punctate,
clustered or curvilinear calcification and rarely causes biliary or duodenal obstruction
- MR- shows low signal intensity on T1-weighted and heterogenous on T2-weighted images with mild contrast enhancement
- Metastases occurs most commonly to liver,then lymph nodes,
lung,
bone,
posterior mediastinum,
peritoneum,
and omentum
LYMPHOMA
- Commonly B-cell subtype of non-Hodgkin lymphoma
- More common in middle-aged patients with a non specific symptomology
- Can be classified as –
- Primary: When pancreas is involved primarily- occurs rarely,
and usually in immunocompromised patients
- Secondary: more common,
and results from direct extension from involved peripancreatic lymph nodes or adjacent structures
Imaging features
Two morphologic patterns seen- Focal well-circumscribed form and a diffuse form
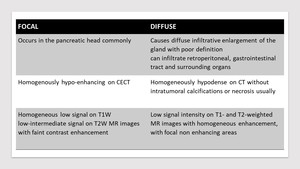
Table 3: Morphological patterns of pancreatic lymphoma
- Lymphadenopathy is seen,
typically below the level of renal veins
- Vascular invasion and ductal dilatation are less frequent in lymphoma than in adenocarcinoma
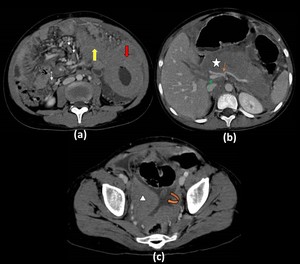
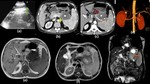
Fig. 6: Disseminated Non Hodgkin Lymphoma with pancreatic involvement in an 11 year old boy:
Contrast enhanced computed tomography axial image (a) shows marked circumferential thickening of the jejunum (red arrow) with normal lumen. Contiguous extension of the lesion in seen in the adjacent mesentery (yellow arrow). Multiple discretely enlarged mesenteric lymph nodes are also seen (white thin arrows). Image at the level of the pancreas shows (b) diffuse enlargement of the organ (star) with mild enhancement on post contrast study. Peripancreatic soft tissue stranding is also present. Encasement of the celiac axis with its branches (orange thin arrow) and the superior mesenteric vein(thin green arrow) is also seen. The section through the pelvis (c) shows similar soft tissue density lesion (triangle) along with ascites (curved arrow)
SOLID PSEUDO-PAPILLARY TUMOR
- These are rare,
typically benign with some showing malignant degeneration
- More common in young women in their 2nd - 3rd decades of life
- Usually asymptomatic
Imaging features
- Most frequently involves the head or tail
- On CT,
a well encapsulated mass with peripherally located enhancing solid and more centrally located non enhancing cystic component is seen but do not show any hypervascularity as seen in NETs.
- MR typically shows a well-defined lesion with heterogeneous signal intensity on T1- and T2-weighted images,
with haemorrhagic areas in between
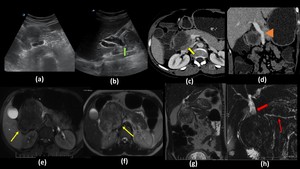
Fig. 19: Solid Pseudopapillary tumor in a 20 year old female presenting to OPD with upper abdominal pain and fullness:
Ultrasound images(a,b) show a well defined heterogeneously hypoechoic lesion involving the head of pancreas which is compressing the distal common bile duct causing proximal dilatation (Green arrow in b). In contrast enhanced axial (c) and oblique coronal (b) CT images, the lesion shows peripheral heterogenous hypo-enhancement (Yellow arrow in c) with internal necrotic areas and is displacing the portal vein (arrowhead in d) with no obvious encasement or invasion. The lesion is also abutting the 2nd part of duodenum with no obvious signs of invasion. On MR images, a well encapsulated solid lesion is seen in the pancreatic head which is hypointense on axial T1WI (e) and is iso to hyperintense on axial (f) and oblique coronal (g) T2WI. The capsule appears hypointense on both T1 and T2 weighted images (Yellow arrows in e,f). On heavily T2-weighted fat-sat image (h) common bile duct proximal to the lesion is dilated (Thick red arrow in h) however the main pancreatic duct is of normal caliber and is displaced by the lesion (Thin red arrow in h).
GASTRO-INTESTINAL STROMAL TUMOR
GIST is the most common mesenchymal tumor of the alimentary tract but very rare to primarily arise from the pancreas
These are seen are large heterogenous masses usually involving pancreatic head,
with areas of cystic degeneration
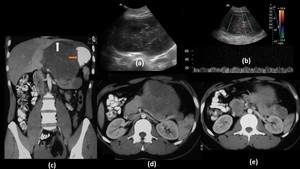
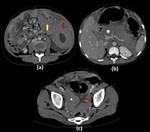
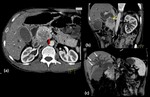
Fig. 2: Gastrointestinal Stromal tumor of pancreas in a 22 year old male:
Ultrasound images of the abdomen show a large heteroechoic lesion in the central abdomen (a) showing internal vascularity on spectral doppler application (b). Axial (d,e) and Coronal CT images (c) with intravenous and oral contrast show a large well defined heterogenous lesion arising from the body and tail region of the pancreas displacing the stomach laterally (Orange arrow in c) and is abutting the liver superiorly. Similar smaller hypodense lesions are also seen in the tail region. Post operative biopsy of the lesion revealed findings consistent with Gastro-intestinal stromal tumor.
PANCREATIC LIPOMA
- Usually detected incidentally on cross sectional imaging for some other indication
- Asymptomatic
- Appears as well-circumscribed,
non-enhancing lobulated hypodense lesion in the pancreas showing fat attenuation
- Hyperintense on T1 and T2 weighted MR images
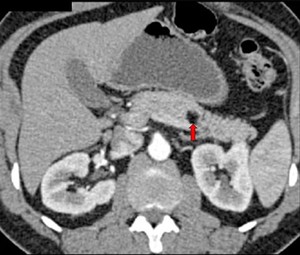
Fig. 10: Pancreatic lipoma detected incidentally in a patient undergoing CECT for abdominal tuberculosis
Contrast enhanced CT on the axial section shows a well-circumscribed round to oval non-enhancing hypodense lesion in the pancreatic body showing fat attenuation ( HU value of -45)
METASTASIS
- Metastases to the pancreas are uncommon in patients with widespread metastatic disease
- Usually asymptomatic and detected incidentally
- Occur frequently from renal cell carcinoma (RCC) and lung carcinoma,
followed by breast carcinoma,
colorectal carcinoma,
and melanoma
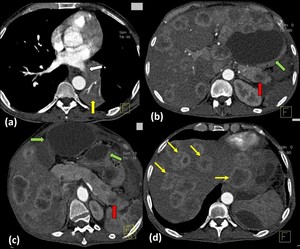
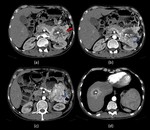
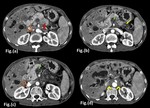
Fig. 4: Bronchogenic carcinoma with liver and pancreatic metastases
Axial contrast enhanced Computed Tomography images show a well-defined enhancing lesion involving the superior segment of left lower lobe (yellow arrow in a) which is obliterating the left inferior pulmonary vein(white arrow in a) and abutting the descending thoracic aorta. Multiple well defined rim enhancing lesions (thin yellow arrows) are seen diffusely involving both lobes of the liver. Few well defined hypoenhancing lesions are also seen involving the body and tail of pancreas (red arrow). Multiple well defined non-enhancing hypodense collections are seen in the peripancreatic region and in the lesser sac (green arrow in c) (likely pseudocyst formation as a sequelae of pancreatitis).
- Three morphologic patterns of involvement are recognized:
- Solitary- most common type,
generally well marginated
- Multifocal
- Diffuse
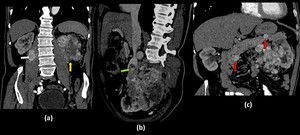
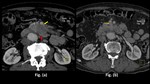
Fig. 5: Renal Cell Carcinoma with pancreatic metastases.
Coronal(a), oblique sagittal(b) and oblique coronal(c) contrast enhanced computed tomography images show an ill defined heterogeneously enhancing lesion involving the left kidney (yellow arrow in a) which is distorting the renal architecture and is invading into the perinephric fat and into the renal vein with tumor thrombus reaching upto the IVC(green arrow in b). Multiple hyper-enhancing lymph nodes are seen in the left pararenal area. Similar well defined hyper-enhancing lesions are also seen involving the head and body of the pancreas(red arrows in c) and also involving the right psoas muscle(white arrow in a).
- Are usually solid but can be cystic also
- Typically have low signal intensity on T1- weighted and high on T2-weighted MR images and iso to hypodense on CT
- Show similar morphology to that of primary carcinoma on contrast-enhanced imaging with homogenous enhancement in small lesions and ring enhancement in larger ones
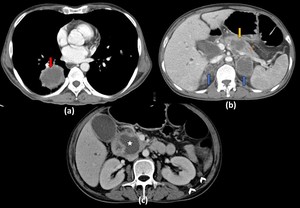
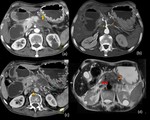
Fig. 3: Bronchogenic carcinoma with multiple necrotic metastasis to pancreas:
Axial contrast enhanced CT image through the lower lungs (a) shows a large necrotic mass with irregular margins in the right lower lobe (Red arrow in a). Section at the level of pancreas (b) shows a necrotic lesion in the pancreatic body causing a bulge in the pancreatic contour. Bilateral necrotic adrenal masses (Blue arrows in b) are also seen. Section at a lower level in the region of the pancreatic head (c) shows another large necrotic mass (star) causing enlargement of the pancreatic head causing compression of the distal common bile duct causing mild dilatation of intrahepatic biliary radicles. Two enhancing peritoneal nodules (Arrow heads in c) are also seen in the perinephric space on the left side.
Biopsy of the pulmonary lesion revealed squamous cell carcinoma.
NON- NEOPLASTIC LESIONS:
FATTY INFILTRATION
- Common finding in elderly or obese patients
- Associated with chronic pancreatitis and cystic fibrosis
- Normally diffuse but rarely can be focal
- Focal fatty infiltration and focal fatty sparing,
can mimic a tumor and needs to be differentiated
- On ultrasound,
pancreatic echogenicity becomes raised
- If there is adequate macroscopic fat,
nonenhanced CT shows characteristic fat attenuation aiding diagnosis but contrast-enhanced CT is not helpful
- MR is the modality of choice being highly specific- shows high signal on T1- and T2-weighted image and signal loss with fat-suppressed sequences
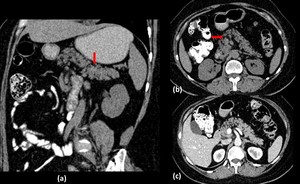
Fig. 11: Fatty infiltration of pancreas
Oblique coronal (a) and axial (b) non contrast CT images with positive oral contrast show multiple diffuse hypodense areas within the entire pancreatic parenchyma with a HU value of -95 with atrophy of the gland. On the contrast enhanced axial section the surrounding pancreatic parenchyma is enhancing which is erroneously increasing the HU value to -12.
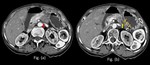
INTRA PANCREATIC ACCESSORY SPLEEN:
- Occurs as a result of failure of fusion of the splenic buds during development
- Most commonly in splenic hilum followed by the pancreatic tail
- Usually small 1–3-cm sized,
well-defined ovoid mass in the pancreatic tail.
- Showing homogeneous echogenicity on ultrasound with a vascular hilum on color Doppler
- Imaging features resemble those of the spleen- shows lower signal intensity on T1-weighted and higher signal intensity on T2-weighted MR images than pancreas with arciform splenic enhancement pattern in the arterial phase
- Technetium 99m (99mTc) sulfur colloid scintigraphy are highly sensitive and can be used to confirm
PSEUDOTUMERAL PANCREATITIS/FOCAL PANCREATITIS:
- Chronic pancreatitis can present as a focal inflammatory mass usually in pancreatic head mimicking adenocarcinoma resulting in unnecessary pancreatectomies
- Difficult to differentiate,
as have similar symptomology,
imaging features,
and even histological appearances
- Inflammation can be seen with adenocarcinoma and,
adenocarcinoma may arise in long-standing inflammatory states like chronic pancreatitis
Imaging features-
- Both are hypoechoic on USG,
hypoattenuating on CT showing similar signal intensities on T1- and T2-weighted MR imaging.
- Both can show double duct sign,
ductal narrowing,
adjacent fat stranding,
arterial encasement,
and peripancreatic venous obstruction
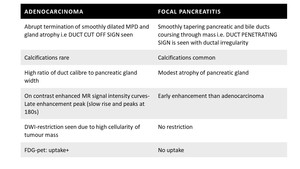
Table 2: Imaging features of adenocarcinoma versus focal pancreatitis
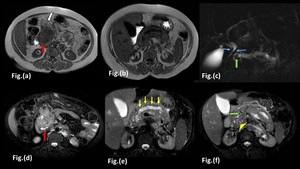
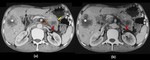
Fig. 1: Focal pancreatitis in a 34 year old male with a long standing history of chronic pancreatitis:
Magnetic Resonance images show an ill defined lesion (Red arrow in Fig a and d) involving the head of pancreas which appears hypointense on T1-Weighted Image (a,b) and is heterogeneously hyperintense on fat saturated T2-Weighted Images (c, d and e) and is invading the peripancreatic fat (White arrow in Fig a). Distal main pancreatic duct appears dilated with irregular margins (Yellow arrows in Fig e) and shows a calculus within its lumen, which is seen as a filling defect in highly T2-Weighted image (Green arrow in c,f). Mild atrophy of distal pancreatic parenchyma is seen with a hypointense foci (Arrow head in Fig f) within the pancreatic parenchyma suggestive of parenchymal calcification. Smoothly tapering common bile duct (Blue arrows in Fig.c) is seen coursing through the mass with mild proximal dilatation.
GROOVE PANCREATITIS
Form of chronic pancreatitis involving pancreato-duodenal groove-potential space between pancreatic head,
duodenum and CBD
Presents with pain,
recurrent vomiting due to duodenal obstruction,
jaundice and weight loss
- SEGMENTAL: Involves pancreatic head with scar tissue present in groove; may resemble a focal mass at imaging
- PURE- Involves groove only,
and spares the head
- IMAGING:
- On CT seen as a sheet like delayed enhancing fibrotic scar lesion between pancreatic head and duodenum.
- Duodenal wall thickening leading to stenosis and cystic dysplasia are commonly seen.
- May show smooth tapering of intrapancreatic portion of CBD
- On MR,
lesion is T1-hypointense T2-iso to hyperintense