1.
Perfusion MR Imaging
Perfusion is a biological function that refers to the delivery of oxygen and nutrients to tissue by means of blood flow.
Perfusion MR Imaging is a technique that evaluates the cerebral microvasculature through changes in signal due to a tracer passing through blood vessels.
1.1 Perfusion Techniques
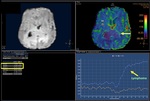
There are two major approaches to measure cerebral perfusion with MRI: those with and without the use of an exogenous contrast agent (Figure 1).
The first group of techniques includes application of an exogenous,
intravascular,
non-diffusible contrast agent,
usually a gadolinium-based contrast agent,
using dynamic susceptibility contrast (DSC)-MR and dynamic contrast-enhanced (DCE)-MR.
The second group includes the application of an endogenous contrast agent using magnetically labeled arterial blood water as a diffusible flow tracer using arterial spin-labeling (ASL).
The most commonly used technique is DSC-MR,
which is based on the susceptibility changes after injecting the contrast agent.
The first pass of a bolus of gadolinium-based contrast agent through brain tissue is monitored by a series of T2- or T2*-weighted MR images and involves the serial measurement of signal loss during the passage of the bolus through the tissue.
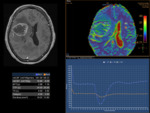
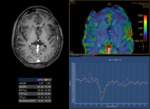
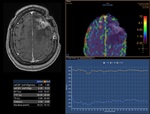
This can be plotted as a signal intensity–time curve (Figure 2),
with the area under the curve being proportional to the cerebral blood volume (CBV).
This is not an absolute value,
it is expressed as a relative measurement (rCBV) that allows the lesion to be compared with the contralateral normal white matter or corresponding structure.
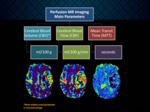
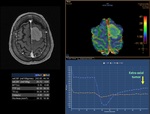
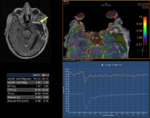
The most commonly evaluated parameters used in Perfusion MR Imaging are cerebral blood volume,
cerebral blood flow,
and mean transit time (Figure 3).
Cerebral blood volume (CBV) is defined as the total volume of blood traversing a given region of brain,
measured in milliliters of blood per 100 grams of brain tissue (ml/100 g).
Cerebral blood flow (CBF) is defined as the volume of blood traversing a given region of brain per unit time,
measured in milliliters of blood per 100 grams of brain tissue per minute (ml/100 g/min).
Mean transit time (MTT) is the average time it takes for blood to traverse between arterial inflow and venous outflow,
measured in seconds.
CBV is the most widely used parameter in neurooncology.
1.2 Perfusion Parameters
To perform a perfusion study there are technical requirements on the acquisition and on the postprocessing side (Figure 4).
The DSC-MR Perfusion is based on a fast echo-planar imaging acquisition that can be performed both at 1.5 T and 3 T.
The amount of contrast media used is 0.1 mmol/kg.
Bolus injection of the gadolinium-based contrast agent should start after about a 20 second delay (range 5–30 seconds) from the start of the DSC MR perfusion sequence.
A minimum 3–5 mL/s bolus injection rate of gadolinium-based contrast agent is recommended using a power injector to allow adequate bolus arrival in the cerebral tissue via an 18 or 20-gauge IV catheter.
This should be followed by a 25 mL (range 10–30 mL) saline flush at the same rate to push the bolus toward the heart.
1.3 Advantages and Disadvantages of Perfusion
Perfusion MR Imaging provides additional information about the vascular morphology and the degree of angiogenesis,
important elements in evaluating different tumor types and determining the biologic aggressiveness of intracranial neoplasms.
The maps of rCBV show the overall tumor vascularity allowing indirect assessment of tumor angiogenesis.
Besides,
these maps direct biopsy towards the most aggressive part of the tumor so tumors are not undergraded.
DSC techniques are the most widely used method to measure brain perfusion with MRI.
It has several advantages over ASL: higher signal-to-noise ratio,
shorter acquisition time and less motion artifacts.
Nevertheless,
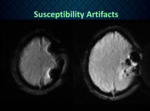
given that it is often performed as a gradient sequence to enhance the detection of T2 signal changes,
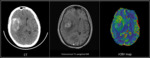
it is adversely affected by hemorrhage or postsurgical changes that cause susceptibility artifact (Figure 5).
Other disadvantages include the difficulty in determining absolute quantification and user dependence.
Blood-brain barrier breakdown leads to underestimation of rCBV,
usually seen in the setting of high-grade neoplasms.
High permeability in these regions leads to extravasation of contrast material into the interstitium,
which increases signal above baseline due to the T1 shortening effects of gadolinium.
Since the algorithm for calculation of rCBV assumes a constant baseline,
the area above baseline is interpreted by the algorithm as negative blood volume and subtracted from the area below baseline caused by the drop in T2* signal leading to a significant underestimation of rCBV.
2.
Perfusion MR Imaging in Tumor Characterization
Different tumor types and grades differ in their perfusion characteristics.
The higher vascularity of brain neoplasms is most commonly quantified with perfusion MR techniques in terms of the cerebral blood volume of the tumor.
2.1 Gliomas
Contrast enhancement is not a reliable indicator of tumor grade.
It is possible to estimate the histopathological grade of cerebral gliomas using dynamic susceptibility MR perfusion imaging.
A strong correlation is found between the grade of astrocytoma and relative CBV measurements.
As the tumor grade worsens,
the relative CBV ratios typically increase.
Nevertheless,
there can be significant overlap between the grades and variability depending on histologic type.
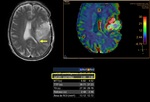
Perfusion MR imaging confidently demonstrates tumor angiogenesis and a normalized rCBV value >1.75 indicates a high-grade neoplasm and a value <1.75 a low-grade neoplasm (Figure 6,
7,
8).
An exception to this rule is oligodendroglioma,
which can show foci of high CBV irrespective of tumor grade,
likely owing to the extensive angiogenesis and dense capillary networks that characterize these tumors (Figure 9).
Because of the high level of histological variability within cerebral gliomas,
rCBV maps of high-grade tumors are often heterogeneous,
containing both high and low rCBV foci.
Therefore,
the focus of maximal CBV is taken to be representative of the tumor grade.
Low-grade tumors,
on the other hand,
tend to have homogenously low rCBV throughout the lesion.
Foci of maximal tumor rCBV have been associated with increased mitosis and vascularity in pathologic specimens.
2.2 Metastases
Vascular metastases such as renal cell carcinoma and melanoma also have higher rCBV than normal gray and white matter (Figure 10).
Since both high-grade gliomas and vascular metastasis have high rCBV,
a way to differentiate them is analyzing the peritumoral edema.
High-grade gliomas are tumors that along vascular channels infiltrate the surrounding parenchyma,
whereas the peritumoral edema of metastatic lesions rarely contain tumor cells.
The presence of high rCBV in the peritumoral region of a lesion is more like to represent a high-grade glioma like glioblastoma rather than a metastasis.
2.3 Primary CNS Lymphoma
The differentiation between a primary CNS lymphoma from other tumors is crucial since the management is different; in case of suspicion of a lymphoma the neurosurgeon will perform a biopsy instead of surgical resection.
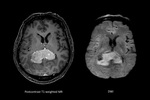
Primary CNS lymphoma in immunocompetent patients usually has a strong homogeneous enhancement and presence of diffusion restriction (Figure 11),
but also has a characteristic perfusion pattern.
Compared with GBM,
lymphomas typically have lower relative CBV ratios due to differences in tumor vascularity.
Although it is not a specific finding,
lymphomas often demonstrate increased signal relative to baseline in the recovery phase due to contrast material leakage within the interstitial space (Figure 12).
2.4 Meningiomas
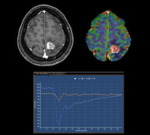
Highly vascular benign neoplasms such as meningiomas often show high rCBV values (Figure 13).
Extra-axial lesions demonstrate a characteristic signal intensity-time curve displaying immediate leakage of contrast agent due to absence of a blood-brain barrier,
as well as the curve does not return to baseline.
The determination of rCBV inside the tumor is not useful in the differentiation between benign and malignant meningiomas,
however,
there is greater rCBV in the peritumoral edema of malignant meningiomas than in the benign meningiomas,
being a useful tool for their preoperative differentiation (Figure 14).
3.
Perfusion MR Imaging in Treatment Response Assessment
Differentiating treatment-related changes from recurrent tumor remains challenging due to overlapping clinical and radiologic features.
Both may present with new and/or increasing enhancing mass lesions.
Radiation therapy and chemotherapy can increase capillary permeability and alter the blood-brain barrier,
leading to increased leakiness of contrast agent and increased contrast enhancement in the absence of tumor mimicking tumor recurrence.
3.1 Pseudoprogression and radiation necrosis versus recurrence
Treatment-related changes may be clinically separated into early pseudoprogression and late radiation necrosis on the basis of timing.
3.1.1 Pseudoprogression
Pseudoprogression is a phenomenon in which there is increase in size and T2 prolongation of the enhancing lesion in patients with CNS neoplasms who undergo radiation therapy and adjuvant chemotherapy,
occurring in approximately 20-40%.
These findings can simulate tumor progression and treatment failure resulting in the inappropriate discontinuation of effective therapy.
Pseudoprogression occurs more often in patients who undergo concurrent radiation therapy and chemotherapy,
typically within 6–12 weeks after treatment (<6 months).
These enhancing lesions are self-limited,
spontaneously stabilize and resolve without any new treatment.
Patients who develop pseudoprogression are often clinically asymptomatic and have improved survival,
due to favorable correlation with methylated O6- methylguanine-DNA methyltransferase (MGMT) promoter status.
Pseudoprogression is histologically similar to radiation necrosis,
with enlarging areas of enhancement that are thought to represent local inflammatory response and increased vascular permeability.
In perfusion MR imaging,
there is a decrease in the relative CBV ratios within the enhancing lesion (Figure 15).
3.1.2 Radionecrosis
As a potential long-term complication of radiation therapy and chemotherapy,
radiation necrosis can occur months to decades after treatment.
Usually occurs >12–18 months after radiotherapy,
the same time period during which recurrence is most likely,
with progressive enhancing lesions that are associated with worsened morbidity.
It is related to the total radiation dose and fraction size,
as well as lesion location and size,
patient age,
and vascular risks.
Histologic findings are characterized by endothelial damage and vascular hyalinization,
leading to occlusive vasculopathy and ischemic changes.
Radiation necrosis usually occurs near the site of the original tumor and within the margins of the irradiation field.
Radiation necrosis appears as a ring-enhancing lesion with characteristic stellate margins,
T2 prolongation and significant vasogenic edema that can mimic tumor recurrence on conventional images.
The pattern of enhancement has been described as “Swiss cheese” or “soap bubble” appearance.
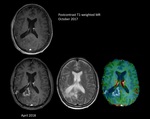
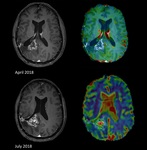
Perfusion MR imaging is useful in the diagnosis where there is a decrease in the relative CBV ratios within the enhancing lesion (Figure 16).
3.1.3 Tumor recurrence
The distinction of radiation necrosis from tumor recurrence is a particularly challenging problem in neuro-oncology,
as treatment is radically different for the two entities.
The radiological differentiation using conventional contrast-enhanced MR or CT scanning is not reliable.
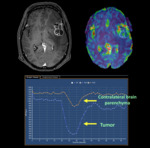
Perfusion MR imaging is relatively specific: tumor recurrence typically demonstrates significantly elevated relative CBV ratios,
whereas pseudoprogression and radiation necrosis are associated with a reduction in relative CBV or hypoperfusion.
rCBV greater than 2.6 indicated tumor recurrence and an rCBV of less than 0.6 is consistent with radiation necrosis (Figure 17,18).
Nevertheless,
it should be taken into account that there is often concomitant tumor and radiation necrosis that sometimes can produce confusing results and follow-up imaging is crucial in these circumstances.
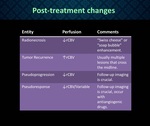
Summary of Treatment Response Assessment (Figure 19).