In the present work we review five cases of rTBADs treated in our institution with TEVAR.
A TBAD patient usually presents with acute onset chest pain irradiated posteriorly with an ongoing hypertensive crisis.
On the other hand,
in cases of rupture,
the patient can quickly become hypotensive as hemorrhagic shock ensues or there can be a variety of intermediate hemodynamic states if the rupture is contained.
Usually pleural effusion is present which can become massive left/right hemothorax but also hemomediastinum with compression of the trachea and esophagus and presence of dyspnea and hemoptysis.
Rupture can present immediately or at any time during the acute period. The protocol in our Institution is that every TBAD patient undergoes immediate contrast CT scan and is then admitted to the cardiovascular intensive care unit for monitoring.
A vascular surgeon performs clinical examination and reviews findings and in case the dissection is complicated the patient is transferred to the OR to undergo TEVAR under general anesthesia.
The operative technique that we routinely employ begins with a one-sided femoral cutdown and contralateral side percutaneous access,
the cutdown side chosen according to imaging and the most favourable approach to the true lumen.
A 6 Fr introducer is inserted and a naked Pig-tail catheter back-loaded with a 0.035” hydrophilic wire is slowly advanced frequently injecting contrast to ensure progression through the TL.
This takes advantage of the curved tip of the catheter which tends to remain inside the TL channel and not engage entry tears.
If resistance is felt angiography with road-mapping is performed and a more standard access is obtained with the help of the guidewire.
We prefer angiography to trans-esophageal echocardiography (TEE) or intravascular ultrasound (IVUS) to confirm TL access in emergency situations as we believe that unless an experienced operator is present the rate of false positives/negatives can unnecessarily delay treatment,
however both techniques are available if needed.
Once a rigid guidewire is safely and correctly positioned in the ascending aorta and the origin of the supraortic trunks,
the entry tear and the origin of the visceral arteries is identified the endograft is inserted and deployed.
Using 3D reconstructions and centerline measurements we have already identified the optimal length and size of the graft.
We prefer to deploy in Ishimaru Zone 3,
just distal to the left subclavian artery (LSA) and extend only the necessary amount to ensure about 5 cm distal to the supposed tear site zone,
always trying to deploy in straight segments of the aorta.
If proximal neck is insufficient Zone 2 deployment with coverage of the LSA is performed.
In emergency settings we do not routinely revascularize the LSA,
unless patient is very stable and we have covered a large length of the aorta,
but we have low threshold of revacularizing the LSA post-operatively.
Spinal cord drainage is also used only selectively.
We do not balloon the graft in dissected patients.
At least two angiographies 20 minutes apart are obtained to ensure no significant perfusion of the false lumen (FL) at the thoracic level.
Post-operatively the patient is transferred to the ICU for overnight monitoring before returning to the Vascular Surgery Ward.
A follow-up CT scan is usually performed the next day to ensure no leakage or endoleak.
Hemothorax is routinely drained 2-3 days after the operation guided by the respiratory status and imaging.
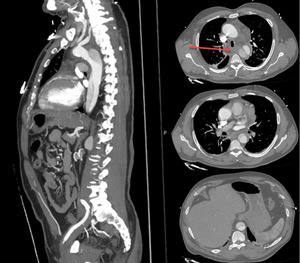
Fig. 1
Fig 1. Pre-operative contrast CT scan showing a ruptured TBAD at the mid-descending thoracic level with hemomediastinum (red arrow) and compression of esophagus and trachea.
Case 1: a 66-yr old male presents to the ED with acute chest pain irradiating posteriorly and ongoing hypertensive crisis.
He is a smoker,
affected by hypertension,
COPD,
mild-moderate aortic insufficiency,
mild mitral insufficiency.
A contrast CT scan is performed with evidence of TBAD extending until the common iliacs.
As per protocol he gets admitted to the cardiovascular ICU and after a few hours develops hypotension (PA 80/50) and dyspnea.
Repeat CT scan shows hemomediastinum with compression of the trachea ad esophagus (Fig 1).
Pre-operative planning is performed with findings of Type I arch,
primary entry tear 24 mm distal to LSA,
circumferential calcifications at the LSA origin,
proximal diameter 25 mm,
mid-descending diameter 23 mm at the suspected site of rupture,
all visceral branches originate from the TL with multiple secondary tears at the level of abdominal aorta,
the Adamkiewitz artery arises from the TL at the level of the body of T12.
Emergent TEVAR is performed using the described standard procedure deploying a distal 26x100 mm module first with a distal landing zone 8 mm from the celiac trunk (T9 level) and a 28x150 mm proximal module in Zone 3 with 7 cm overlap between the two.
The patient makes an uneventful recovery at follow-up CTs show complete thrombosis of the FL at the thoracic level (Fig 2).
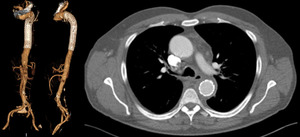
Fig. 2
Fig 2. A 12 month follow-up CT scan shows complete thrombosis of the false lumen at the descending thoracic level.
Case 2: a 57-yr old male present to the ED with acute chest pain and dyspnea.
A contrast CT scan reveals a complex ruptured TBAD with left hemothorax.
(Fig.
3)
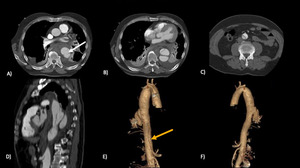
Fig. 3
Fig.3 Type B Aortic Dissection; a large entry tear (white arrow) is visible in the proximal portion of the descending thoracic aorta.
False lumen extends till the infrarenal abdominal aorta (yellow arrow).
3DVR reconstruction (e; f).
Large entry tear is present in the proximal-mid descending aorta and a rupture is evident at the distal descending aorta in the presence of another large secondary tear just distal to the rupture.
In this case we decide perform TEVAR across the false lumen in order to more adequately exclude the rupture site.
A guidewire is advanced into the TL and then made to cross into the FL through the secondary tear.
The wire is further progressed and made to re-enter the TL through the primary tear.
Two endografts are then placed telescopically 31x150 mm distally and 37x150 mm proximally with 5 cm overlap covering from TL to FL to TL again. The hemothorax is drained in post-operative day 2 with a chest tube.
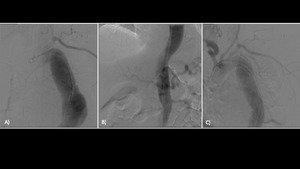
Fig. 4
Fig.4 Pre TEVAR aortography (a; b) with contrast medium injection through the true lumen.
Post TEVAR aortography (c).
Recovery is uneventful and follow-up CT scans show no complications at the descending thoracic level.
(Fig 5)
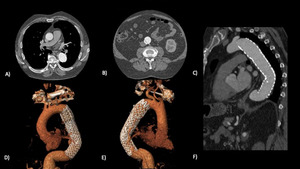
Fig. 5
Fig.
5 Post TEVAR follow up CT (12 months).
Two endografts placed in descending thoracic aorta,
covering the proximal wide entry point.
Case 3: a 61 yr-old female patient presents with massive left hemothorax and hemorrhagic shock in the setting of a ruptured TBAD.
(Fig 6)
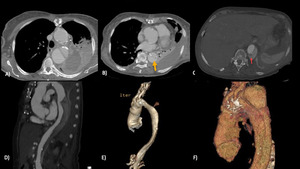
Fig. 6
Fig.6 Type B dissection,
after substitution of the ascending aorta.
(b) Small entry point in the upper descending aorta (yellow arrow).
(c,d) Another possible entry point in the distal portion,
hinted by increased opacification in the false lumen (red arrow).
(e,
f) 3DVR reconstruction.
The patient has a previous substitution of the ascending aorta for an aneurismatic disease.
Primary tear is visible in the proximal aorta with a possible secondary tear in the mid-distal descending aorta.
TEVAR is performed with a 31x150 graft and the patient stabilizes.
Post-operative imaging shows incomplete coverage of the secondary tear but despite the persistent FL in the uncovered portion no additional leakage of blood is detected and the patient makes uneventful recovery.
(Fig 7)
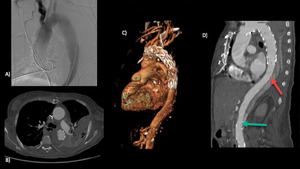
Fig. 7
Fig. 7 (a) Pre TEVAR aortography.
(b,c,d,e) TEVAR covering only the proximal entry point with shrinkage of the upper portion of the false lumen.
The possible distal entry point (red arrow) remain uncover with opacification of the false lumen.
An other entri point is visible in abdominal aorta (green arrow).
Case 4: a 64 yr-old female patient presents with type A acute aortic dissection and undergoes ascending aortic repair with hemiarch reimplantation.
On post-operative day 5 a contrast CT scan shows dilatation of the residual type B-post A dissection false lumen with suspected contained rupture at the mid thoracic level (Fig 8).
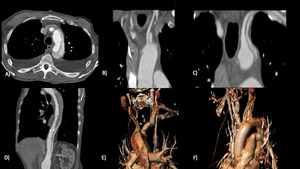
Fig. 8
Fig. 8 Type B - post A dissection,
after substitution of the ascending aorta.
Entry point are appreciable in left CCA and in left subclavian artery.
Other entry point is demonstrated in the abdominal aorta,
with opacification of the false lumen.
Carotid-carotid-subclavian by-pass is performed and a 150 mm endograft is deployed using the surgical prosthesis as a landing zone just distal to the innominate artery.
Two plugs,
one at the origin of the left carotid and the other at the origin of the left subclavian artery are placed.
(Fig 9)
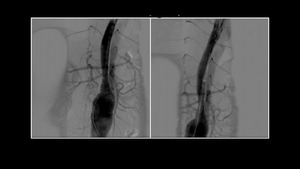
Fig. 9
Fig. 9 Pre TEVAR implant aortography demonstrate a thick dissection flap with no clear entry point.
the TEVAR il positioned landing on the surgical prosthesis only after carotid-carotid and carotid-subclavian bypass packaging.
Two plugs,
one at the origin of the left carotid and the other at the origin of the left subclavian artery are placed.
Recovery is uneventful and FU CT shows complete thrombosis of the FL at the thoracic level.(Fig 10)
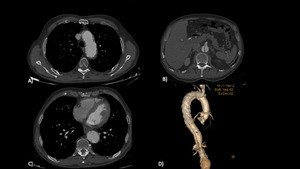
Fig. 10
Fig. 10 Post TEVAR follow up CT (6 months).
Shrinkage of the proximal false lumen due to closure of the entry point in left carotid and subclavian artery.
The distal entry point in descending thoracic aorta is not covered in order to guarantee blood flow in celiac trunk and SMA.
Case 5: a 28 yr-old suspected Marfan patient is transferred to our institution for hemorrhagic shock in ruptured TBAD at the mid thoracic level.
Emergent TEVAR is performed in Zone 3 with a 31x150 graft and the patient stabilizes.
Completion angiography shows very mobile aorta and persistence of the false lumen riperfused from below with high flow. As the imaging is being reviewed the patients develops a new hypotensive episode.
Open surgery is selected as the next choice of treatment with the intention to perform banding of the aorta around the endograft but as a left thoracotomy in the 6th intercostal space is being initiated the patient develops pulseless electrical activity and resuscitation proves futile even after cross-clamping the aorta.
It is known that the Windkessel function of the aorta is more pronounced in younger patients so we hypothesized that despite the initial reduction in flow after coverage of the primary tear and rupture subsequently the flow through the false lumen helped maintain the leakage in an already very compromised patient.











