We had series of 20 patients (age range 18-72) with pathologically proven SBOT between July 2015 and July 2018.
We demonstrated MR imaging features of SBOTs using our cohort.
Histologically,
typical borderline serous tumours are non-invasive proliferative neoplasms characterized by multiple fibrous papillae with extensive and complex hierarchical branching [2].
Similarly in MR images,
they are usually seen as a cystic or a solid mass showing abundant hyperintense papillary projections with low signal internal branching and ovarian stroma preservation with a hypointense ovarian capsular margin on T2-weighted imaging.
Naqvi et al.
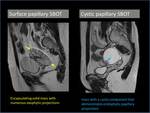
classified the pure epithelial SBOTs as surface papillary and cystic papillary subtypes(Figure 2) [4].
1. Surface papillary SBOT
It appears as an encapsulating solid mass with numerous exophytic projections but no cystic component or endophytic projections.
2. Cystic papillary SBOT
It appears as a mass with a cystic component that may demonstrate endophytic,
in addition to exophytic papillary projections.
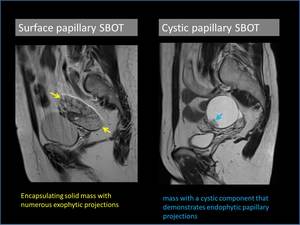
Fig. 2: Examples of surface and cystic papillary subtypes of serous borderline ovarian tumors.
Although serous borderline tumours were classified as papillary cystic and papillary surface subtypes histopathologically in previous version of WHO classification (1999-2000),
this nomenclature is not used in the last version of WHO classification (2014) [3].
Principal MR imaging features
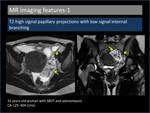
1. T2 high signal papillary projections with low signal internal branching (Figure 3)
T2 low signal represents the central fibrous stroma of the papillary projections and the hyperintense regions represent oedematous papillae[4].
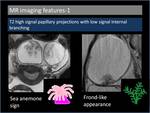
Tanaka et al described this appearance similar to sea anemone and deemed it as a "hallmark” feature of surface BOT [5].
Those projections might also be assumedto have frond-like appearance (Figure 4).
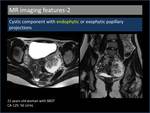
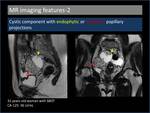
2. Cystic component with endophytic or exophytic papillary projections (Figure 5,6)
A spherical high signal intensity cyst is seen with internal low to intermediate signal endophytic papillary projections or mural nodules on T2W imaging [4].
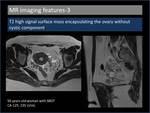
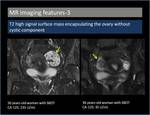
3. T2 high signal surface mass encapsulating the ovary without cystic component (Figure 7,
8)
T2 high signal papillary projections related to the surface of the ovary without any cystic component suggest the diagnosis of serous BOT [6].
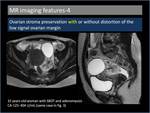
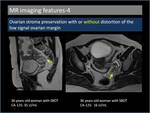
4. Ovarian stroma preservation with or without distortion of the low signal ovarian margin(Figure 9,
10)
An important finding that differentiates serous BOT from other malignant lesions is the presence of normal ovaries or at least some normal ovarian tissue,
completely or partially surrounded by the solid tumour [6,
7].
Due to characteristic growth pattern of serous surface papillary BOTs,
both ovaries are likely to be of normal size and shape [6].
On T2W imaging,
a low signal rim can be delineated around the ovarian margin.
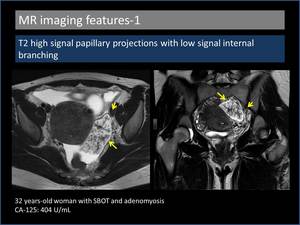
Fig. 3: T2 high signal papillary projections with low signal internal branching
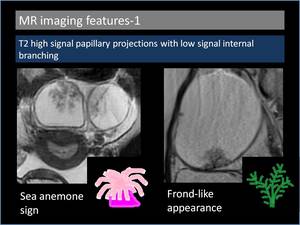
Fig. 4: Sea anemone sign and frond-like appearance in serous borderline ovarian tumours.
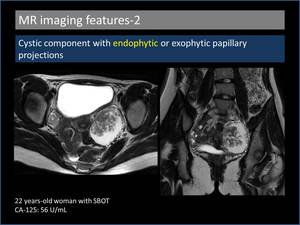
Fig. 5: Cystic component with endophytic papillary projections.
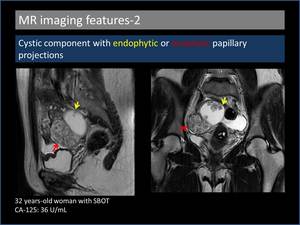
Fig. 6: Cystic component with endophytic and exophytic papillary projections.
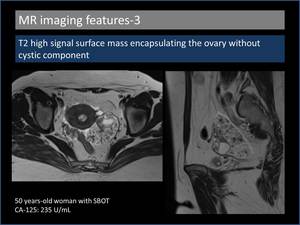
Fig. 7: T2 high signal surface mass encapsulating the ovary without cystic component.
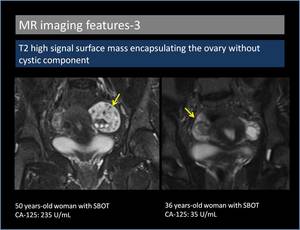
Fig. 8: T2 high signal surface mass encapsulating the ovary without cystic component.
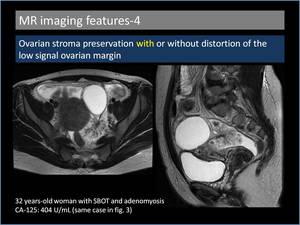
Fig. 9: Ovarian stroma preservation with distortion of the low signal ovarian margin.
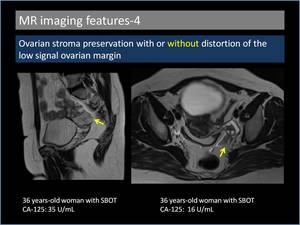
Fig. 10: Ovarian stroma preservation without distortion of the low signal ovarian margin
Features associated with SBOT
1.
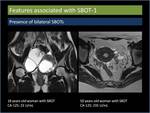
Presence of bilateral SBOTs (Figure11)
2.
Accompanying peritoneal implants (Figure 12): Peritoneal implants are more likely to be seen in serous BOTs with exophytic projections.
The prognostic implications of the implants depend on the invasiveness of the implants,
which cannot be accurately determined by MRI [4].
3.
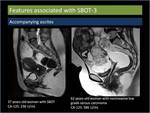
Accompanying ascites (Figure 13): Ascites is an unreliable indicator for predicting the presence of BOT [4].
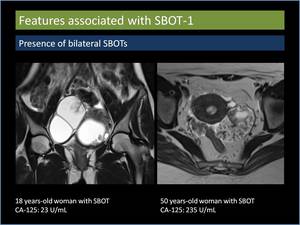
Fig. 11: Presence of bilateral serous borderline ovarian tumours.
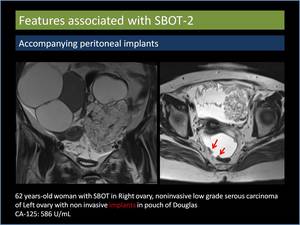
Fig. 12: Accompanying peritoneal implants.
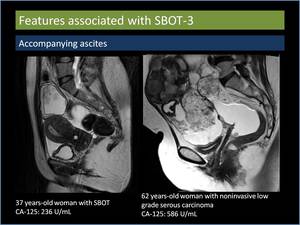
Fig. 13: Accompanying ascites.
Tips and tricks in differential diagnosis of SBOTs
1.
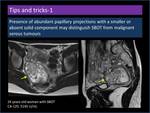
Presence of abundant papillary projections with a smaller or absent solid component may distinguish SBOT from malignant serous tumours (Figure 14).
2.
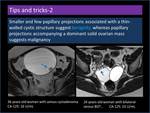
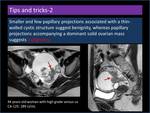
Smaller and few papillary projections associated with a thin-walled cystic structure suggest benignity,
whereas papillary projections accompanying a dominant solid ovarian mass suggests malignancy (Figure 15,
16)
3.
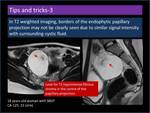
In T2 weighted imaging,
borders of the endophytic papillary projection may not be clearly seen due to similar signal intensity with surrounding cystic fluid.
Look for T2 hypointense fibrous stroma in the centre of the papilla (Figure 17).
4.
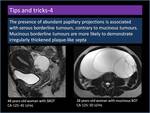
The presence of abundant papillary projections is associated with serous borderline tumours,
contrary to mucinous tumours.
Mucinous borderline tumours are more likely to demonstrate irregularly thickened plaque-like septa (Figure 18).
5.
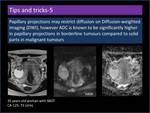
Papillary projections may restrict diffusion on Diffusion-weighted imaging (DWI),
however ADC is known to be significantly higher in papillary projections in borderline tumours compared to solid parts in malignant tumours (Figure 19).
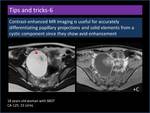
6.
Contrast-enhanced MR imaging is useful for accurately differentiating papillary projections and solid elements from a cystic component since they show avid enhancement (Figure 20).
 7.
Clear cell carcinoma (CCC) of the ovary may represent as unilocular large cysts with mural solid component and thus can be a mimicker of cystic subtype of SBOTs (Figure 21). The ADC values of solid portions tend to be lower in CCC compared with SBOT.
7.
Clear cell carcinoma (CCC) of the ovary may represent as unilocular large cysts with mural solid component and thus can be a mimicker of cystic subtype of SBOTs (Figure 21). The ADC values of solid portions tend to be lower in CCC compared with SBOT.
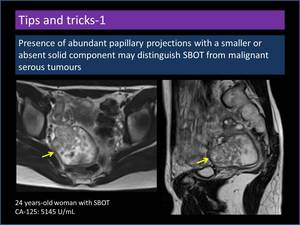
Fig. 14: Presence of abundant papillary projections with a smaller or absent solid component.
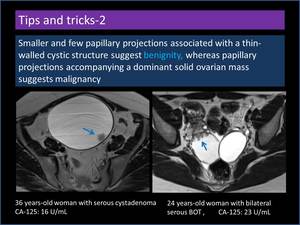
Fig. 15: Smaller and few papillary projections associated with a thin-walled cyst
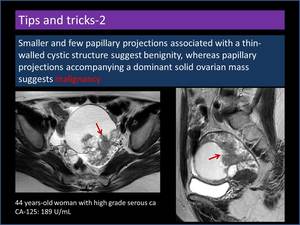
Fig. 16: Papillary projections accompanying a dominant solid ovarian mass.
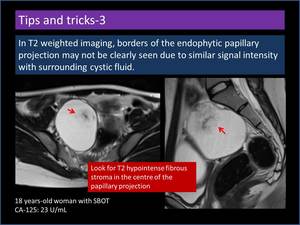
Fig. 17: Endophytic papillary projection with subtle borders.
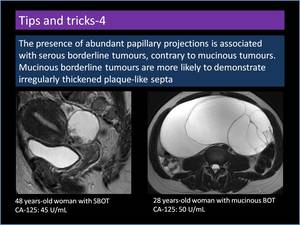
Fig. 18: Difference between serous and mucinous borderline tumours.
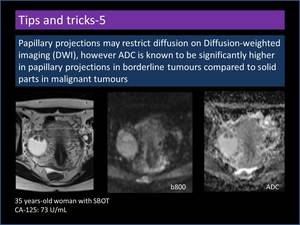
Fig. 19: Diffusion-weighted imaging features of papillary projections.
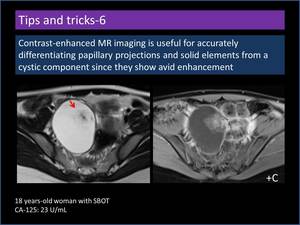
Fig. 20: Contrast-enhanced MR imaging features of papillary projections.
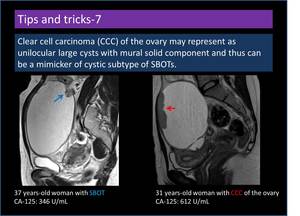
Fig. 21: Clear cell carcinoma of the ovary as a mimicker of cystic subtype of serous borderline ovarian tumours.
Clinical approach to serous borderline ovarian tumors:
- Although CA 125 is an important tumour marker in distinguishing benign and malignant adnexal masses,
it is rather non-specific for the diagnosis of SBOT.
Since the range of CA 125 level overlaps between serous surface papillary BOT and serous papillary carcinomas,
CA 125 levels are usually not sufficient to differentiate two entities [6].
However,
CA 125 level has a role in the follow-up of BOT patients.
Wide range of CA 125 level in SBOTs is demonstrated in the figures.
- Important prognostic factors in patient outcome are FIGO stage,
presence of peritoneal implants,
micropapillary pattern and microinvasion.
However,
lymph node invasion does not seem to be a prognostic factor [8].
- The knowledge of SBOT and presence of normal ovarian tissue before surgery is important in determination of treatment options which includes fertility-sparing surgery in a high proportion of women in the reproductive age group.
- Surgical staging generally includes resection of the primary borderline tumour (either unilateral salpingo-oophorectomy or ovarian cystectomy),
cytologic washings,
omentectomy and peritoneal biopsies.
Routine lymphadenectomy is not recommended [8].
- Surgery alone is the standard therapy for these tumours as the relapse rate is extremely low (for SBOT relapse rate <1%).
Postoperative therapy is recommended only for those with SBOT and invasive implants,
despite the lack of definitive evidence of benefit [8].
- There is no standard in terms of monitoring patients after primary treatment of a BOT.
Most true recurrences involve extra-ovarian sites and the majority of recurrent tumours are invasive low-grade serous carcinoma rather than SBOT [8].