Chest X-Ray (CXR) often represents the initial imaging examination,
being important in detecting and localizing mediastinal masses when clinically suspected.
Paravertebral masses disrupt the paraspinal stripes,
which is helpful in localizing the lesion in the posterior compartment,
but the diagnostic value of conventional radiography remains rather limited [3].
Ultimately,
cross-sectional imaging (CT,
MRI) plays a crucial role in distinguishing between different types of mediastinal masses and identifying their origin and cause,
based on morphological characteristics related to fat,
fluid,
soft tissue or calcium content.
The vascular involvement can also be assessed,
alongside enhancement patterns and relation to surrounding organs.
In assessing most mediastinal lesions,
multidetector CT with intravenous contrast material represents the imaging modality of choice [4].
Reporting considerations:
When reporting a mediastinal mass,
the following elements should be considered [2],
[5].
- Localization to a mediastinal compartment,
size
- Descriptive features: density (cystic versus solid); heterogeneity; enhancement
- Presence of intralesional components: fat,
cystic components,
calcification
- Evaluation of edge contours (connection with or invasion of neighboring structures)
MR imaging due to its high contrast resolution can further characterize the lesion,
being the preferred modality in evaluating neurogenic tumors because it provides information regarding the nature and extent of intraspinal involvement [3].
Furthermore,
MRI can be performed to patients who cannot undergo contrast-enhanced CT imaging due to severe contrast allergy and/or impaired renal function and can also be used to evaluate younger patients to lower radiation dose.
MRI is superior to CT in distinguishing cystic versus solid lesions,
identifying cystic or necrotic components within solid lesions,
and identifying septations or soft tissue components within cystic lesions [5].
However,
this modality does not demonstrate calcification as well as CT and has a lower spatial resolution [3].
1.
Neurogenic tumors
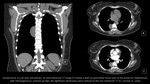
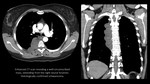
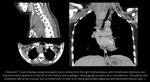
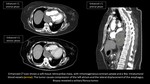
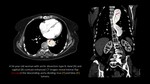
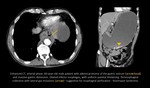
1.1 Peripheral nerve sheath tumors (PNST) - typically arise from spinal or proximal intercostal nerves and account for approximately 70% of mediastinal neurogenic tumors [6].
1.1.1.
Benign PNST include schwannomas and neurofibromas.
Schwannomas are the most common PNST,
often seen in Neurofibromatosis type 2 patients,
while multiple neurofibromas are associated with Neurofibromatosis type 1 [7].
CXR:
- well-circumscribed,
round/oval paraspinal masses with variable extension.
CT:
- soft tissue density masses with the widening of the neural foramina,
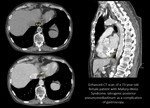
usually a more markedly convex mass arising from the vertebral body compared to sympathetic nerve tumors ( Fig. 3, Fig. 4, Fig. 5);
- "dumbbell" or "hourglass" configuration and communication with the spinal canal;
- possible benign pressure erosions of adjacent ribs;
- schwannoma: frequent internal cystic changes and hemorrhage [7],
[8].
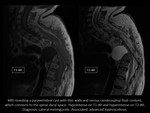
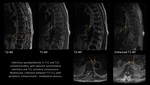
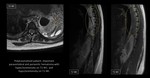
MRI: optimally demonstrates the extent of intraspinal/extradural extension.
Schwannomas:
- typically present the fascicular sign (multiple small,
hypointense,
ring-like structures corresponding to fascicular bundles);
- variable signal intensity on T1-WI sequences and intermediate to high signal intensity on T2-WI;
- homogeneous/heterogeneous enhancement depending on regions of cellular and acellular (or myxoid) components [9].
Neurofibromas:
- on T2-WI typically present the “target sign” (central low signal with high signal rim); gradual and weak contrast enhancement (due to myxomatous and fibrous tissues within the tumor) [8].
1.1.2.
Malignant PNST are rare spindle cell sarcomas arising de novo from a peripheral nerve or can result from the malignant transformation of neurofibromas.
They are typically heterogeneous and show peripheral enhancement due to central necrosis in the tumor on contrast-enhanced CT and MRI [8].
Suggestive imaging features also include rapid growth, bone destruction,
invasion of adjacent structures and pleural effusion [7].
1.2.
Sympathetic nerve tumors include neuroblastomas,
ganglioneuromas and ganglioneuroblastomas,
which present themselves as vertically oriented masses with a tapered appearance,
alongside the anterolateral surface of several vertebrae [8].
Imaging features of these tumors are similar; the age of onset is an important differentiation factor [7].
CT:
- paraspinal soft tissue masses with variable enhancement;
- coarse calcifications in 80% neuroblastomas; punctate calcifications in 20% ganglioneuromas [7].
MRI:
- ganglioneuromas: T2-WI heterogeneous high signal due to myxomatous components; “whorled appearance” - curvilinear bands of low signal intensity due to the collagenous fibrous tissue in the tumor [10].
1.3.
Paragangliomas are neuroendocrine hypervascular tumors.
Contrast-enhanced CT:
- intense homogeneous enhancement; may show peripherally and within the mass feeding vessels.
MRI:
- T1-WI: intermediate signal; T2-WI: high signal; avid enhancement;
- “salt and pepper” appearance (high signal = slow flow within tumor vessels or hemorrhage; low signal = high flow within tumor vessels) [11].
2.
Non-neurogenic solid tumors
2.1.
Extramedullary hematopoiesis represents a physiological compensatory mechanism that occurs when the bone marrow has an ineffective function and is unable to provide sufficient red blood cells [8].
It is usually part of various chronic hematologic pathologies such as myelofibrosis,
lymphoma,
hemolytic anemias [12].
CXR:
- well-delineated paravertebral masses,
usually bilateral [8].
CT:
- active lesions: usually soft-tissue density;
- inactive lesions: low/high attenuation values depending on the presence of fat,
or the iron content.
MRI:
- active lesions: intermediate signal intensity on both T1-WI and T2-WI; mild homogenous enhancement;
- inactive lesions: hypersignal on both T1-WI and T2-WI if fatty replacement or hyposignal with iron deposition; heterogeneous enhancement [13].
2.2.
Lymphoma
Mediastinal Hodgkin and non-Hodgkin lymphoma may arise either as a primary malignancy or as a manifestation of systemic disease.
CXR: a widened mediastinum; posterior tracheal,
or retrocardiac mediastinal opacity [14].
CT:
- enlarged lymph nodes present homogenous soft-tissue density,
possible cystic or necrotic changes; mild to moderate enhancement [4].
MRI:
- used to evaluate response to chemotherapy:
- untreated lymphomas: homogenous low signal on T1-WI and a moderate signal on T2-WI;
- inactive residual lesions due to an increase in fibrotic content present lower T2-WI signal intensity and decreased/absent enhancement [4,
9].
2.3.
Uncommon mediastinal tumors
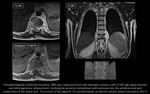
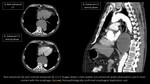
Chordomas are slow-growing tumors derived from the remnant cells of the primitive notochord.
They only rarely occur in the dorsal spine and posterior mediastinum.
CXR: discrete soft tissue masses with the widening of the mediastinum; bone erosion.
CT: well-circumscribed mass with soft tissue density,
hyper-attenuating relative to the spinal cord,
inhomogeneous due to intrinsic calcifications,
hemorrhage or necrotic tissue; mild to intense enhancement; bone destruction sometimes with peripheral sclerosis.
MRI: T1-WI intermediate/low signal intensity + focal high signal areas due to hemorrhage/mucus; T2-WI high signal intensity due to increased water content (Fig. 6); inhomogeneous enhancement,
honeycomb pattern due to fibrous septa [15].
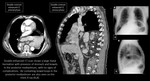
Solitary fibrous tumors are a rare group of slowly growing tumors of mesenchymal origin (less than 2% of all soft tissue masses) [16].
On cross-sectional imaging they do not have distinguishable features from other soft tissue mediastinal neoplasms,
therefore,
the final diagnosis is based on biopsy (Fig. 7).
CT: well-circumscribed,
often lobulated,
hypervascular tumor.
MRI: T2-WI markedly inhomogeneous with large areas of bright signaling reflecting extensive areas of necrosis [16,
17].
3.
Non-neurogenic cystic tumors
3.1.
Lateral meningocele
An intrathoracic meningocele is caused by the abnormal herniation of the leptomeninges through an intervertebral foramen or a defect in the vertebral body.
CT and MRI reveal a paravertebral cyst with thin walls and serous cerebrospinal fluid content, connecting to the spinal dural space [18].
CT: well-circumscribed,
homogeneous,
low-attenuated paravertebral masses; frequently associated with intervertebral foramen enlargement,
vertebral scalloping,
scoliosis [2].
MRI: T1-WI hypointense; T2-WI hyperintense; no enhancement (Fig. 8).
3.2.
Esophageal duplication cysts are uncommon foregut developmental anomalies.
CT: well-defined,
homogeneous,
low-to-high attenuation lesions (depending on their fluid or proteinaceous content),
closely adjacent to the esophagus wall (Fig. 9) [18].
MRI: proves the cystic nature - various patterns of signal intensity on T1-WI if proteinous,
mucoid or hemorrhagic content is present [9].
3.3.
Pancreatic pseudocyst
In a patient with a clinical setting of pancreatitis,
a cystic mass in the paravertebral mediastinum that develops over a short period of time may represent an intrathoracic extension of a pancreatic pseudocyst via the esophageal or aortic hiatus.
Cross-sectional imaging plays an important role in assessing complications such as pleural effusion,
hemorrhage,
fistulization,
or mediastinitis [13].
4.
Hiatal hernia refers to the protrusion of elements of the abdominal cavity through the diaphragm into the chest.
CXR: retrocardiac air-fluid level suggestive for paraesophageal hiatal hernia; intestinal herniation: multiple opacities in the posterior mediastinum containing air or air-fluid levels; smaller hernia might require barium studies.
CT: shows the diaphragmatic defect and the contents of the hernia sac (Fig. 10); assesses possible complications (obstruction/strangulation); cranial displacement of the gastroesophageal junction or gastric fundus through the hiatus can be visualized on oral contrast-enhanced CT;
MRI: no advantages over CT,
therefore,
it is not routinely used in the diagnosis of hiatal hernia [19]
5.
Vascular lesions
Aortic dissection is a medical emergency resulting from a tear in the intimal layer of the aortic wall,
which allows the blood inflow through the medial layer.
An intimal flap separates the “false lumen” from the “true lumen”.
A descending aortic dissection accounts for type B in Stanford classification (Fig. 11).
CXR: non-specific: mediastinum enlargement; abnormal cardiac contour; displacement of aortic wall calcifications.
Contrast-enhanced CT: permits early recognition of the dissection type and extension; the intimal flap is visualized in most cases.
MRI: alternative imaging modality [13].
6.
Infection
Infectious spondylitis is usually caused by pyogenic or tuberculous infection,
most commonly spread through the hematogenous pathway.
The infection of a vertebral body may extend into the pre – or paravertebral soft tissue.
Unlike X-ray,
both CT and MRI can detect early osteolytic destruction of the vertebra and the intervertebral disc involvement.
Imaging features of paraspinal abscesses:
CT:
- soft-tissue density; “horseshoe” mass surrounding the affected vertebral body; (pyogenic abscess – associates complete destruction of the intervertebral disc; tuberculous abscess – associates calcifications); internal foci of air may be present [20].
MRI:
- focal hypointense (to muscle) region on T1-WI,
hyperintense on T2-WI (Fig. 12)
- peripheral enhancement is characteristic: thick and irregular-pyogenic infection; thin and smooth-tuberculous infection
- allows early diagnosis of spinal cord compression syndrome and involvement of the epidural space [9,13].
Mediastinal abscesses can also form as a consequence of esophageal perforation – Boerhaave syndrome (Fig. 13).
7.
Traumatic lesions
Paravertebral lesions related to trauma may also be encountered in the PM.
Mediastinal hematoma imaging characteristics:
CT: abnormal soft tissue density around the mediastinal structures; contrast extravasation is suggestive for vessel injury and indicative of the source of hemorrhage.
MRI: highly sensitive in detecting acute hemorrhage,
but it is not used in an emergency setting [21]; variable imaging features (Fig. 14),
depending on the hemoglobin's chemical state in relation to the onset time of the hemorrhage.
Pneumomediastinum is a rare condition in which extraluminal free air is present within the mediastinum.
Causative factors are traumatic injuries or pathologies which determine air escaping from the lung, tracheobronchial tree or digestive tract.
It is also one of the possible complications of upper gastrointestinal tract endoscopy (Fig. 15).
CXR: air outlining mediastinal structures; visible mediastinal pleura.
CT: assesses extension; identifies the underlying cause,
differentiates between pneumomediastinum and pneumopericardium [22].