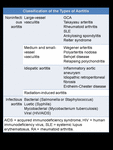
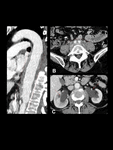
Classification of aortitis into two categories, noninfectious and infectious, is useful for clinical purposes ( Fig. 1 ).
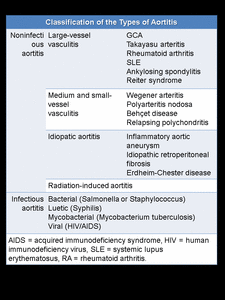
Fig. 1: Classification of the Types of Aortitis.
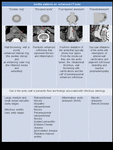
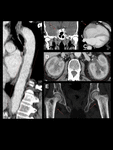
The pattern of aortic involvement and imaging features help distinguish between noninfectious and infectious causes ( Fig. 2 ).
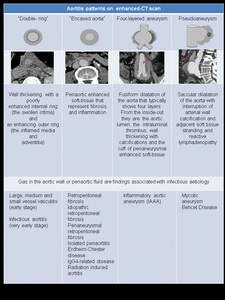
Fig. 2: Aortitis patterns on enhanced-CT scan
Takayasu arteritis (TA)
TA is a large-vessel vasculitis that predominantly affects the aorta, its major branches and the pulmonary arteries.
The initial lesions frequently occur in the left subclavian artery and progress to the other supra-aortic trunks. The abdominal aorta is involved in approximately 50% of patients.
The exact cause is unknown. TA has a predilection for young Asian women.
The pathologic analysis demonstrates granulomas and inflammation of the arterial wall, followed by fibrosis and calcification in the late stage.
The diagnosis is based on clinical and imaging findings. TA is commonly divided into two clinical stages. In the early stage, manifestations are mainly vague, systemic and constitutional. The late-stage is characterized by manifestations related to arterial stenosis, occlusion, or dilatation with clinical findings that depend on the vascular territory affected. Aneurysmal dilatation and rupture of the affected aorta are not rare, with a prevalence of 33%.
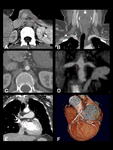
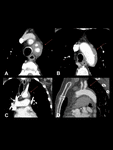
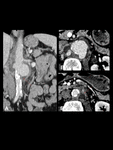
Contrast-enhanced CT scan allows early diagnosis. Findings include wall thickening with a “double ring” appearance (typically seen in the early stage), thrombosis, stenosis, and occlusion. Other associated findings include vessel ectasia, aneurysms, and ulcers. Wall calcification develop in chronic cases and usually spares the ascending aorta. Fig. 3.
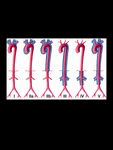
TA has been classified based on location, the involvement of the coronary and pulmonary arteries should be indicated as C(+) or P(+), respectively. Fig. 4.
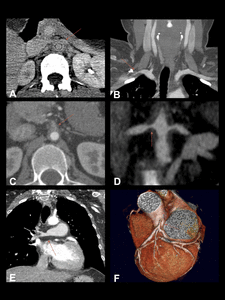
Fig. 3: Takayasu arteritis in a 30-year-old woman. Contrast-enhanced CT scan. Extensive thickening of the aortic wall. Notice the double ring pattern (A). There are thickening and stenosis of the aortic branches, such as the right subclavian artery (B), celiac trunk (C)and renal arteries (D). The pulmonary and coronary arteries are also affected (F and G respectively).
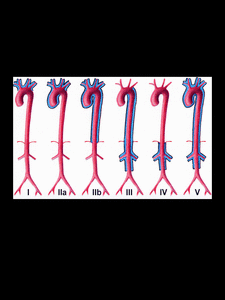
Fig. 4: System for classifying Takayasu arteritis according to the site of involvement.
References: Nastri MV, et al. Gadolinium-enhanced three-dimensional MR angiography of Takayasu arteritis.Radiographics. 2004 May-Jun;24(3):773-86.
Giant Cell Arteritis (GCA)
GCA is a large and medium-vessels vasculitis that usually involves the extracranial carotid artery and its branches, such as the temporal artery. Vascular inflammation may be widespread, involving the aorta, its branches and the vertebral arteries. Aortic involvement occurs in 15-20% of GCA patients.
GCA is the most common primary vasculitis and the most common form of aortitis, accounting for more than 75% of cases.
The exact cause is unknown although it is thought to be secondary to an autoimmune process. The pathogenesis is secondary to a systemic granulomatous vasculitis followed by fibrosis. There is a recognized predilection for women over 50 years old. Polymyalgia rheumatic is seen in 50% of GCA patients.
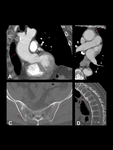
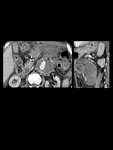
Contrast-enhanced CT scan findings are wall thickening with a “double ring” appearance (early stage). Late complications are stenoses, occlusions, ectasia, aneurysm and dissection. Fig. 5.
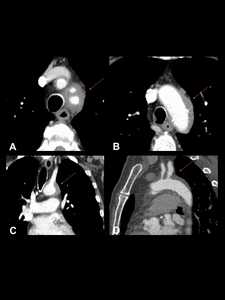
Fig. 5: GCA in a 65-year-old woman. Contrast enhanced CT images show diffuse wall thickening of the thoracic aorta. (A obtained at a higher level than B, C and D are coronal and sagittal reconstructions).
Ankylosing Spondylitis (AS)
AS is a seronegative spondyloarthropathy. HLA-B27 is the gene with the strongest association (5%). There is a young male predilection and 18% of cases manifest with inflammatory back and buttock pain.
Symmetrical and bilateral sacroiliitis is usually the first finding. AS results in ankylosis of the spine and sacroiliac joints. AS can also affects peripheral joints and extraarticular tissues such as the skin, the mucous membranes, the eye, the lung and the bowel. AS is the most common rheumatic disease associated with aortitis and periaortitis.
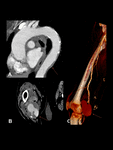
Contrast-enhanced CT scan shows wall thickening of the ascending aorta (60%). The aortic root and valve are frequently affected due to thickening, nodularity and rolling of valve cusps with annular dilatation that leads to valvular insufficiency. The extension of the subaortic fibrosis into the interventricular septum may cause conduction abnormalities. AS can also cause secondary chronic periaortitis (CP) that includes retroperitoneal fibrosis (RPF) and inflammatory abdominal aortic aneurysms (IAAA). Fig. 6.
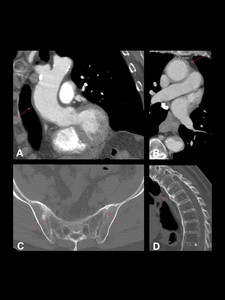
Fig. 6: Aortitis secondary to AS in a 72-year-old woman. Contrast-enhanced CT images showing wall thickening of the ascending aorta (A and B). Notice symmetrical and bilateral sacroiliac joint ankylosis (C) and diffuse syndesmophytic ankylosis giving a "bamboo spine" appearance (D).
Behçet disease (BD)
BD is a multisystemic, relapsing and chronic inflammatory vasculitis of unknown aetiology. There is a young male predilection. BD is most prevalent in the Mediterranean region, the Middle East and East Asia.
The clinical triad of BD consists of oral and genital ulceration and ocular manifestations. This disease can also affect the skin, joints, the bowel, genitourinary tract, central nervous system and the lung. Vascular complications develop in about 30% of affected patients. The aorta is the most common artery affected (5%) with a predilection for the abdominal aorta, followed by the pulmonary arteries.
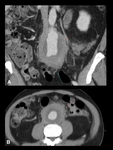
Contrast-enhanced CT scan shows wall thickening and wall-enhancing saccular pseudoaneurysms that may develop in the thoracic and abdominal aorta. In one-fifth of patients, multiple pseudoaneurysms develop in large vessels such as the aorta and the iliac, femoral, popliteal, and subclavian arteries. Fig. 7.
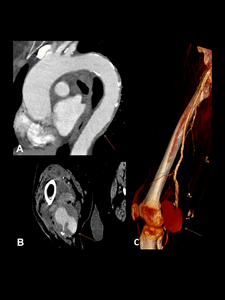
Fig. 7: Aortitis in Behçet disease in a 77-year-old woman. Contrast-enhanced CT images showing wall thickening of the thoracic aorta (A) and a saccular pseudoaneurysms of the popliteal artery ( B and C).
Inflammatory abdominal aortic aneurysm
IAAA is characterized by inflammatory thickening of the aneurysm wall and perianeurysmal fibrosis. IAAA is thought to be secondary to an abnormal inflammatory response to atherosclerosis combined with autoimmunologic factors. The prevalence is 5%–25% of all abdominal aortic aneurysms. IAAA has a predilection for a young male with a family history of this disease.
Contrast-enhanced CT scan shows a four-layered appearance of the abdominal aorta that usually spares the posterior wall. Complications are secondary to ureteral involvement with resultant hydronephrosis, Aortoenteric fistula, and secondary bacterial infection. It is important to assess the extension and the adhesions to adjacent tissue to avoid planning surgery. Fig. 8.
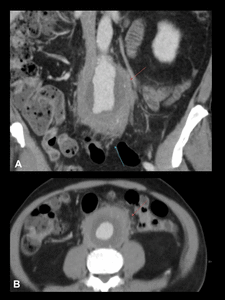
Fig. 8: IAAA in a 65-year-old man. Contrast-enhanced CT images show a significant amount of soft tissue surrounding a fusiform dilatation of the distal aorta (A coronal and B axial). Notice the four layers pattern and the inflammatory soft tissue that mainly involves the anterior and lateral aortic walls, sparing the posterior wall. There is fat stranding adjacent to bowel wall (Purple arrow).
Retroperitoneal fibrosis (RPF)
RPF is characterized by masslike fibroinflammatory tissue mainly in the retroperitoneum that extends to retroperitoneal structures. RPF is idiopathic but has been associated with certain drugs, malignant disease, and systemic disorders.
CT scan shows a retroperitoneal mass of soft-tissue attenuation that typically enhances in the early stages. This process tends to initiate about the aortic bifurcation. There are cases confined to the thoracic aorta.
RPF encases but does not invade or stenose the ureters, vessels or bones. However, ureteric obstruction and venous thrombosis can occur. As a rule, the mass does not displace the aorta and inferior vena cava anteriorly from the spine. Multiple reactive lymph nodes are frequently seen. Fig. 9.
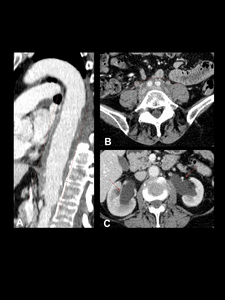
Fig. 9: Idiopathic retroperitoneal fibrosis (IRF) in a 70-year-old man. Contrast-enhanced CT scan. Notice the fibro-inflamatory soft-tissue encompassing the thoracic aorta (A) and retroperitoneal structures causing ureteric obstruction (D).
Erdheim-Chester Disease (ECD)
ECD is a non-Langerhans cell histiocytosis characterized by multiorgan xanthomatous
infiltration. This rare, non-inherited disease has a predilection for middle-aged men.
Patients with ECD experience bone pain, diabetes insipidus, exophthalmos, constitutional symptoms, interstitial lung disease, ureteral obstruction, renal impairment, cardiac dysfunction and tamponade and xanthelasmas due to xanthomatous infiltration.
On CT bilateral and symmetric osteosclerosis of the lower extremity metaphysis and diaphysis, is the most common finding (96%). Vascular involvement manifests as circumferential infiltration of the thoracic and abdominal aorta that appears as enhancing soft-tissue attenuation. Infiltration can progress in the aortic branches resulting in renovascular hypertension and myocardial infarction. Retroperitoneal infiltration is present in 30% of patients, inferior vena cava and pelvic ureters are typically spared, differing from RPF. Fig. 10.
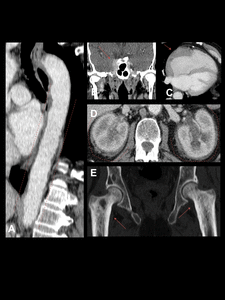
Fig. 10: Erdheim-Chester disease in a 65-year-old man. Contrast-enhanced CT scan. Notice the wall thickening of the aorta due to fibro-inflamatory surrounding soft tissue leading to a “coated aorta” appearance (A). There is right cavernous sinus (B), right atrial free wall (C) and perirenal infiltration, observe the “hairy kidney” appearance typically seen in this disease (D). In addition, there is cortical thickening and bilateral metaphyseal sclerosis of both femurs (E).
Mycotic aneurysm (MA)
MA is a consequence of infectious aortitis that ultimately disrupts the vessel wall, creating a false lumen or pseudoaneurysm. MA is infrequent (< 2.6%). Predisposing factors include atherosclerosis, intravascular devices and conditions that cause immunosuppression. Salmonella spp is generally the organism implicated.
The most common location is the infrarenal aorta. MA of the aortic root has been associated with infectious endocarditis. CT scan shows a typical MA pattern (Table 1). Other findings include perianeurysmal gas, fat stranding, fluid, vertebral body destruction, psoas abscess and kidney infarct. Fig. 11 and Fig. 12.
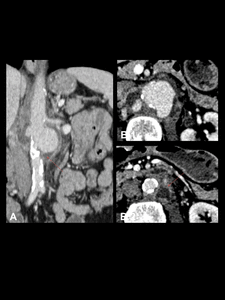
Fig. 11: MA due to Salmonella species in a 59-year-old woman. Contrast-enhanced CT scan. Saccular aneurysm of the infrarenal aorta. Notice the multilobulated appearance and the interruption of arterial wall calcification (A), the adjacent stranding and soft tissue mass (B 1-2 successively lower levels) and the paraortic abscess that affects the left psoas muscle (orange arrow).
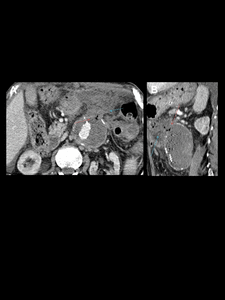
Fig. 12: Aneurysm sac infection following prior EVAR in a 79-years-old man. Contrast-enhanced CT scan showing gas in aneurysm sac (orange arrows). Notice adjacentfat stranding and free fluid (purple arrow). Fistula between the excluded aortic sac and the duodenum was confirmed on surgery.