Video capsule endoscopy (VCE) is an excellent, minimally invasive diagnostic tool. It is remarkably well-tolerated and enables visualisation of otherwise unreachable sections of bowel (1). It has multiple diagnostic utilities including: diagnosing and monitoring Crohn’s and Coeliac disease, assessing obscure gastrointestinal bleeding and identifying small bowel tumors, amongst others Fig. 2 .
Imaging the small bowel
Prior to the development of VCE in 2000, existing methods to visualise the small bowel were limited and invasive. VCE has enabled the entirety of the small bowel mucosa to be scrutinised in order to provide definitive diagnoses.
VCE Fig. 1
- Developed for medical use in 2001 by Given Imaging Ltd. and called the ‘Mouth to Anus’(M2A). Subsequently renamed ‘PillCam SB’ (small bowel) (2).
- Various manufacturers currently
- 26 x 11mm capsule
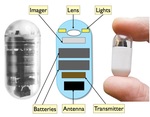
- Consists of two small button batteries, an imaging sensor surrounded by four light emitting diodes(LED), a radio transmitter unit and an antenna (3)
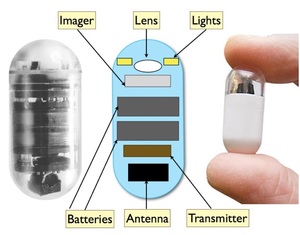
Fig. 1: Internal components of a video capsule endoscope

Fig. 2: Indications for capsule endoscopy
Advantages of VCE
- Minimally invasive
- No sedation or insufflation
- Pan-intestinal visualisation with excellent mucosal detail Fig. 3
- No radiation exposure
- Ability to review images after completion of procedure
Limitations of VCE
Despite its extensive advantages, VCE has limitations:
- Inability to obtain biopsies/perform therapeutic interventions
- Inability to control movement through the GI tract
- Intestinal debris impairs visualisation
- High rate of missed lesions through duodenum/proximal jejunum due to rapid transit
- Significant risk of capsule retention in patients with known small bowel stricturesFig. 4
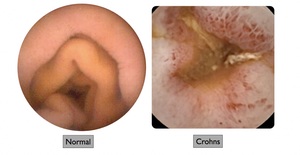
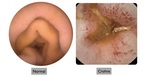
Fig. 3: Select images from VCE study demonstrating normal gastrointestinal mucosa (left) versus ulceration in a patient with Crohn’s disease (right).
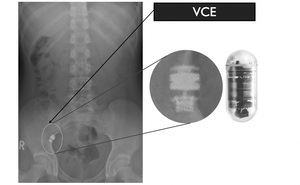
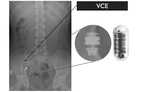
Fig. 4: A terminal ileal stricture in a 16-year-old Crohn’s disease patient causes capsule retention. The abdominal radiograph below demonstrates the radiographic appearance of a capsule compared with that which is swallowed.
Capsule retention
Capsule retention is defined as “having a capsule endoscope within the gastro-intestinal tract for > 14 days post ingestion”(4). Retention may result in intestinal obstruction and perforation, requiring invasive surgery for retrieval.
Retention rates in patients have been reported as follows:
- Established Crohn’s: 5% (5) but anywhere between 3-15% reported in the literature(6, 7)
- Suspected Crohn’s: 2.4% (5)
- Obscure upper GI bleeding (OGIB): 1.4% (8).
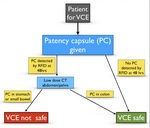
Selecting patients to undergo a patency capsule test
The following patients are deemed to be at higher risk of capsule retention and would therefore benefit from a patency capsule (PC) test prior to undergoing a VCE:
- Known narrowing/ stricturing of the intestinal lumen, in many cases secondary to Crohn’s disease
- Symptoms suggestive of small bowel obstruction
Our centre adopts a non-selective approach whereby everyone planned for a VCE is given a PC test beforehand.
Patency capsule Fig. 5 Fig. 6
The patency capsule was first developed in 2005 (9)and its design has been modified to produce the Agile patency capsule:
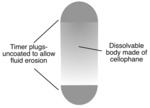
- Measures 26 x 11mm (same dimensions as VCE)
- Two timer plugs located at each end of the cellophane body of the PC. When intestinal fluid makes contact with the plugs, the dissolution process begins (starts at ~30 hours and ends by ~40 hours)
- Central Radio-Frequency Identification Tag (RFID), which enables a hand-held device to detect a retained capsule
Despite having a dissolvable body, patency capsules have a theoretical risk of RFID tag impaction. Thus, a further tagless capsule has been developed (Figure 6), which has a theoretical advantage, although hand-held detection is not possible with this subtype.
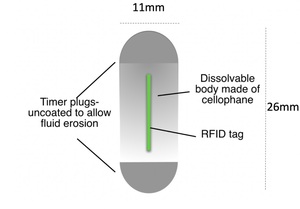
Fig. 5: Patency capsule with RFID tag
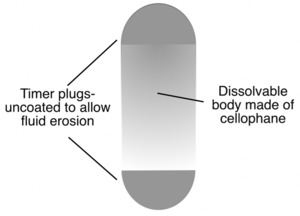
Fig. 6: Tagless patency capsule
Localisation of the patency capsule and Royal Free Hospital Protocol Fig. 7
All patients undergoing VCE regardless of risk factors have a patency capsule (PC) test at our institution, the Royal Free Hospital.
The patient ingests the PC and is reviewed in clinic 48 hours later. If the PC has not been excreted, further investigations are warranted in order to localise the capsuleFig. 7.
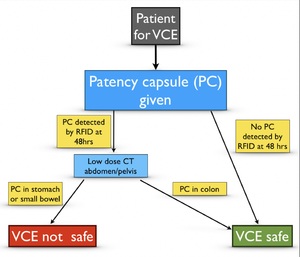
Fig. 7: Royal Free Hospital protocol for investigations prior to VCE