BRAIN ATROPHY
Brain volume declines normally with aging at a certain rate due to atrophy of both grey and white matter. Loss of myelin, neuronal cell death and axonal degeneration cause a gradual loss of total brain volume that begins as early as the age of 30 (at a rate of 0.2% per year). After the age of 70 this loss is more substantial as the rate rises up to 0.5% per year (5% per decade). Although automated volumetry to assess quantitatively if brain atrophy is normal or abnormal is promising, it is still being developed.
Visual rating scales such as Global Cortical Atrophy (GCA) scale and Medial Temporal lobe Atrophy (MTA) scale are helpful to distinguish normal from abnormal atrophy.
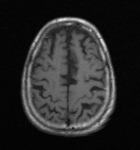
The GCA scale (fig. 1) is a qualitative score for cortical atrophy throughout the brain to assess cerebral atrophy.
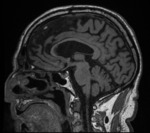
Asymmetric and regional or focal atrophy (fig. 4) should be considered abnormal, as neurodegenerative diseases such as Alzheimer disease (AD) or frontotemporal dementia (FTD) have those features. A GCA score of 3 is always abnormal. A score of 2 is considered abnormal on patients under the age of 75.
0 : No cortical atrophy
1 : Mild atrophy (opening of sulci)
2 : Moderate atrophy (volume loss of gyri)
3 : Severe atrophy (knife-blade atrophy)
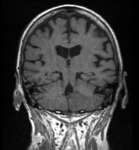
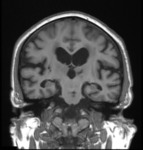
The Medial Temporal lobe Atrophy (MTA) scale (fig. 2 and 3) is based on the loss of volume of the hippocampi with aging and is useful for the diagnosis of AD. The score should be rated on coronal T1-weighted images through the hippocampus at the level of the anterior pons and three features are considered: width of the choroid fissure, width of the temporal horn of the lateral ventricle and hippocampal height. A MTA score of 3 or more is always abnormal. A score of 2 on one side is abnormal on patients under the age of 75.
0 : No atrophy
1 : Slight widening of the choroid fissure
2 : Moderate widening of the choroid fissure. Mild widening of the temporal horn. Mild loss of the hippocampal height
3 : Marked widening of the choroid fissure. Moderate widening of the temporal horn. Moderate loss of the hippocampal height
4 : Marked widening of the choroid fissure. Marked widening of the temporal horn. Marked atrophy of the hippocampus.
Parietal atrophy can be rated according to the Koedam scale (fig. 4). It can be helpful in the evaluation of patients with particular forms of dementia such as atypical or early-onset AD.
0 : Closed sulci and no gyral atrophy
1 : Mild cingulate and parieto-occipital sulcus widening and mild atrophy of the parietal lobes and precuneus
2 : Moderate cingulate and parieto-occipital sulcus widening and moderate atrophy of the parietal lobes and precuneus
3 : Marked cingulate and parieto-occipital sulcus widening and knife-blade atrophy of the parietal lobes and precuneus
WHITE MATTER (WM) CHANGES:
Cerebral WM T2-hyperintensities are visible as hypodense regions on CT and as hyperintensities on T2-weighted and FLAIR images on MRI, usually bilateral or diffuse, located in the periventricular region. The exact mechanisms are still a matter of debate, but it is widely accepted that WM hyperintensities are due to myelin loss caused by small vessel disease. It is a frequent finding in older patients as their volume increases with age and a strong link with vascular events is supported by recent studies.
The Fazekas scale (fig. 6, 7 and 8) quantifies the amount of WM hyperintensities and is best scored on axial T2-weighted or FLAIR images. A score of 2 or 3 is indicative of underlying small vessel disease. A score of 3 is always abnormal and can be helpful in the diagnostic process of vascular dementias.
0 : None or a single punctate WM hyperintense lesion
1 : Multiple punctate lesions
2 : Beginning confluence of lesions
3 : Large confluent lesions
ENLARGED PERIVASCULAR SPACES (EPVS):
PVS, also known as Virchow-Robin spaces, consist on microscopic spaces (usually
PVS can become enlarged with aging and are most frequently seen in the inferior basal ganglia region (fig. 9), centrum semiovale, mesencephalon and subinsular region. The term “état criblé” refers to numerous enlarged basal ganglia perivascular spaces (fig. 10).
Even though EPVS are usually well-circumscribed, round or linear shaped, and lack surrounding gliosis, they may mimic lacunar infarcts, especially large EPVS of the anterior temporal lobe that can present a rim of gliosis or edema in asymptomatic patients (fig. 11).
SILENT BRAIN INFARCTIONS:
Cortical microinfarcts, lacunar and cerebellar infarcts are very frequent in asymptomatic aging patients, can be caused by small emboli or small vessel disease and are related to a higher risk of dementia and stroke. Lacunar infarcts (fig. 12) are located in the distal distribution of deep penetrating vessels (basal ganglia and deep white matter) and measure between 3 and 15 mm. Lacunes are usually differentiated from EPVS by their rim of gliosis and their location (basal ganglia vs lower basal ganglia region). The term “état lacunaire” refers to the presence of multiple lacunar infarcts and must not be confused with “état criblé”.
Cerebellar infarcts (fig. 13) most commonly affect the posterior lobules, result from the occlusion of end-arteries and are also a common finding in the aging patient.
CEREBRAL MICROBLEEDS:
Microbleeds can be found in up to 20% of the general population over 60 years, thus a common finding in aging. They are better appreciated on T2*-weighted sequences as small, round, hypointense areas (2-10 mm) of signal void and represent chronic hemorrhages due to underlying small vessel disease.
Usually, a single microbleed is not considered to have any particular clinical relevance, whereas multiple microbleeds should raise suspicion of vasculopathy and are related to increased risk of dementia, stroke and worse cognitive function.
The pattern of distribution of microbleeds can raise suspicion of its etiology. Deep microbleeds (basal ganglia, thalamus, brainstem and cerebellum) reflect hypertensive microangiopathy (fig. 14). Strictly lobar microbleeds (fig. 15) are cortico-subcortical (grey-white matter junction) in location and are strongly linked to cerebral amyloid angiopathy (CAA), sparing the basal ganglia and brainstem.
IRON DEPOSITION:
The accumulation of non-heme iron in the basal ganglia (most frequently in the globus pallidus) increases with age. It is most apparent as hypointense areas on T2*-weighted sequences and it depends on technical factors such as field strength and sequence features (fig. 17).
There are, however, rare genetic disorders that can lead to the excessive accumulation of iron in the basal ganglia (fig. 18). As there is some impairment in the quantification of iron load, the distinction between normal aging and pathological processes can prove challenging.