Heterogeneity of clinical presentation linked to variations in the genotype at codon 129 of PRNP and the type of PrPSc deposited in the brain. Genotypes can be methionine homozygous (MM), valine homozygous (VV), or heterozygous (MV). Type 1 and 2 are two biochemically distinct forms of PrPSc.
Main neuroimaging findins related to the 6 molecular subtypes of sCJD are:
- MM1 and MV1 subtypes (55-70%). Typical clinical and MRI findings, with frequent basal ganglia and cortical involvement (thalamus relatively spared).
- VV1 subtype, younger age of onset and slow progression (resembling variant CJD). Prominent cortical involvement (basal ganglia and thalami spared; variant CJD has the bilateral "pulvinar sign").
- MV2 and VV2 subtypes. Prominent basal ganglia and thalamus involvement.
- MM2 often divided into a cortical and thalamic subtype.
MR Imaging Findings
Restricted diffusion and hyperintensity (FLAIR) in cortical gyri (cortical ribonning). Reduced ADC may be due to neuronal vacuolation, PrPSc accumulation or both combined.
CJD diagnosis by MRI can be classified as definitive, probable and unlikely.
Clues for definitive diagnosis include DWI over FLAIR hyperintensity in:
- Cortex (>1 gyrus), striatum and >1 neocortical gyrus
- Cortex only (>3 gyrus)
Complementary criteria for subcortical involvement: DWI striatum anterior-posterior hyperintensity gradient (posterior putamen relative spared)
Complementary criteria for cortical involvement: asymmetric involvement of midline neocortex or cingulate.
Usually there is no mass effect or post-contrast enhancement.
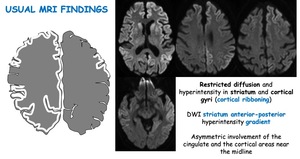
Fig. 3: Usual MRI Findings. Restricted diffusion and hyperintensity in striatum and cortical gyri
Isolated limbic involvement is not found in sCJD, and the perirolandic area is usually spared.

Fig. 4: Usual MRI Findings. Perirolandic area usually spared; absence of limbic involvement
Best diagnostic key for sCJD is progressive DWI or FLAIR hyperintensities in cerebral cortex, caudate and putamen. Serial MRI demonstrates that most cases with unilateral DWI restriction in the first MRI scan shows bilateral involvement in the following studies. Increases in signal intensity associate with disease duration and spongiform degeneration extention, serving as a noninvasive biomarker of disease severity.

Fig. 5: Serial MRI reveals that both cortical and basal ganglial DWI hyperintensities tend to increase in time
As the disease progresses, signal intensity abnormalities can decline or even disappear on DWI. This feature, linked to advanced stage of the disease, has been associated with neuronal death and increased atrophy.
At imaging, white matter is not commonly affected or is affected only with disease progression when cerebral atrophy, thickening of the cerebral cortex and enlargement of the lateral ventricles occur.
Despite a normal appearance, there is histopathologic evidence that white matter dysfunction occurs in the early stages of CJD.

Fig. 6: Cerebral atrophy with thickening of the cerebral cortex in advanced stage of the disease
The pulvinar sign (part of the WHO diagnostic criteria for variant CJD (vCJD) means that the posterior thalamus is brighter than the anterior putamen.
The double hockey stick sign is defined as a high signal in both the pulvinar and dorsomedial thalamic nuclei.
Both signs are relatively specific but not pathognomonic of vCJD. Thalamic involvement has also been reported in some molecular subtypes of sCJD (incidence up to 45%).
When thalamic involvement is present, the keys to differentiate sCJD from vCJD are:
- Thalamic changes in vCJD are symmetrical.
- sCJD associated with striatum and neocortical abnormalities.

Fig. 7: Unusual MRI findings. Pulvinar and double hockey stick signs
Hyperintensity in the globus pallidus on T1WI is another unusual finding associated with sCJD (reflects heavy deposition of prion protein). The putamen has an even higher protein content, but does not appear hyperintense on T1WI due to the coexistent high degree of spongiform degeneration.
There is a high prevalence of cerebellar symptoms and a profound neuropathologic phenotypic involvement of the cerebellum in sCJD. However, the hallmark of cerebellar involvement in sCJD is atrophy.

Fig. 8: Unusual MRI findings. Hallmark of cerebellar involvement in sCJD is atrophy
Main differential diagnosis of sCJD with MRI
Abnormalities in the cerebral cortex, basal ganglia, thalamus, and cerebellum at MR imaging might represent severe acute disorders with several different causes:
Severe hypoxic ischemic encephalopathy. In adults, usually results from cardiac arrest or cerebrovascular disease with secondary hypoxemia. Severe damage primarily affects the gray matter structures: basal ganglia, thalami, cerebral cortex (sensorimotor and visual cortices, including perirolandic area), cerebellum and hippocampi. Diffusion is the earliest imaging modality to become positive, frequently within the first few hours.

Fig. 9: Differential diagnosis of sCJD. Severe hypoxic ischemic encephalopathy
Hypoglycemic encephalopathy. Results from prolonged or severe hypoglycemia (overdose of medication in diabetic patients or pancreatic insulinoma). Abnormalities are usually bilateral and asymmetric affecting internal capsule, corpus callosum (boomerang sign), cerebral cortex (in particular parieto-occipital and insula), hippocampus and basal ganglia. The cerebellum, brainstem and thalami usually spared in adults (involved in neonates).
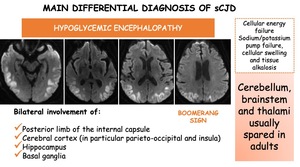
Fig. 10: Differential diagnosis of sporadic Creutzfeldt-Jakob disease. Hypoglycemic encephalopathy
Autoimmune-mediated encephalopathy. Antibody-mediated brain inflammatory process. Limbic encephalitis (paraneoplastic or non-neoplastic) is the most common pattern of autoimmune encephalopathy and is characterized by T2/FLAIR hyperintensity in the mesial temporal lobes, typically involving the amygdala and hippocampus (not affected in sCJD).

Fig. 11: Differential diagnosis of sCJD. Autoimmune-mediated encephalitis
Infectious disease (encephalitis). Herpes simplex encephalitis is the most common agent of acute sporadic encephalitis, and has been recognized as a trigger for anti-NMDAR encephalitis. Typically involves the anterior and medial aspects of the temporal lobes and the orbital frontal lobes, often asymmetrically. Easy differentiation from sCJD (serious febrile medical condition with mesiotemporal involvement).

Fig. 12: Differential diagnosis of sCJD. Herpes simplex encephalitis
Post-ictal State. Transient and reversible signal intensity changes in patients with focal or generalized seizures or status epilepticus. Focal transient edema manifested as gyral swelling, T2/FLAIR hyperintensity, gadolinium enhancement, and DWI reversible signal intensity abnormalities involving the hippocampus and cortex (specific clinical and EEG scenario).

Fig. 13: Differential diagnosis of sporadic Creutzfeldt-Jakob disease. Postictal state
SMART syndrome. Uncommon delayed complication of cerebral radiation therapy. Seizures and subacute-stroke like episodes. Prominent unilateral gyral enhancement at MRI with mild mass effect, usually included in an area included in the radiation ports.

Fig. 14: Differential diagnosis of sCJD. SMART syndrome
Hepatic encephalopathy. Patients with liver dysfunction and portal hypertension (exposure of the brain to excessive amounts of ammonia). Extensive cortical signal intensity changes with bilateral involvement of the insular cortex and cingulate gyrus. Involvement of the perirolandic cortex helps to differentiate from sCJD.
Mitochondrial disorders. Mitochondrial encephalopathy with lactate acidosis and stroke-like episodes (MELAS), manifests as multifocal stroke-like cortical lesions in different stages of evolution, which cross the cerebral vascular territories and predilection for the posterior parietal and occipital lobes. There is also a prominent lactate “doublet” at 1.3 ppm in normal-appearing brain.

Fig. 15: Differential diagnosis of sporadic Creutzfeldt-Jakob disease. Mitochondrial disorders. MELAS



























