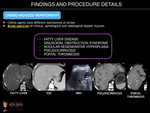
Cancer therapy has evolved to include classic cytotoxic agents in addition to newer options such as molecularly targeted agents. Their different mechanisms of action may result in a broad spectrum of clinical (ranging from asymptomatic patients to fulminant liver failure), pathological and radiological hepatic injuries, including diffuse hepatopathy (i.e. hepatitis, steatosis, fibrosis, pseudocirrhosis, or sinusoidal obstruction) or focal hepatopathy. Fig. 3 Fig. 4
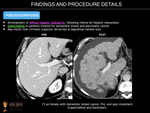
FATTY LIVER DISEASE
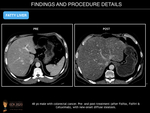
Chemotherapy-associated steatosis and steatohepatitis (CASH) are observed in up to 47% and 28% of patients, respectively. Fatty infiltration of liver parenchyma, known as steatosis, results from oxidative stress from chemotherapy. CASH is a more severe form of fatty liver disease on which hepatocyte degeneration leads to liver inflammation and fibrosis.
Clinically, it is often asymptomatic; however, it can often manifest through mild elevations of alanine transaminase and aspartate transaminase.
Fatty changes have been linked, in particular, to irinotecan, when used in combination with folinic acid and 5-fluorouracil (FOLFIRI) to treat patients with liver metastases of colorectal carcinoma. Thus, it should be avoided in patients with severe steatosis identified on imaging.
Other associated chemotherapeutic agents include platinum derivatives, taxanes, and methotrexate with initial steatotic appearance occurring from 2 weeks to 2 months after therapy.
CASH may be diffuse or focal. Typical locations of focal fat deposition include falciform ligament, porta hepatis, and gallbladder fossa. CASH can be detected on ultrasound (US) as increased parenchymal echogenicity and beam attenuation. On computed tomography (CT), it is characterised by decreased parenchymal attenuation. On magnetic resonance (MR) imaging, it can be detected by a drop in signal intensity on opposed phase images compared with in-phase images. This is particularly useful in the diagnosis of focal hepatic steatosis, when it may be difficult to differentiate fat from metastasis if fatty infiltration occurs in unusual locations.
Detecting and reporting fatty changes is clinically meaningful. It may prompt changes in treatment, especially in metastatic colorectal cancer patients who are planned for hepatic metastasectomy, as underlying steatosis may alter hepatic function and increase the risk for postoperative complications, particularly of those patients receiving neoadjuvant therapy.
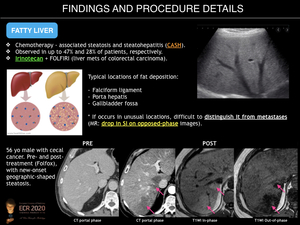
Fig. 5: Fatty Liver
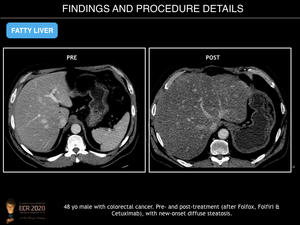
Fig. 6: Fatty Liver
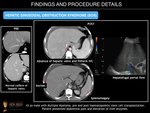
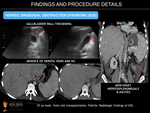
SINUSOIDAL OBSTRUCTION SYNDROME
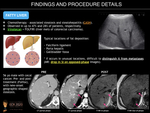
Hepatic sinusoidal obstruction syndrome (SOS), previously called veno-occlusive disease, is a serious, potentially life-threatening complication consisting of vascular liver injury caused by microvascular fibrosis. This fibrous deposition leads to obstruction of small intrahepatic vessels and liver sinusoids, with subsequent sinusoid dilatation and congestion, finally provoking hepatocyte disruption and necrosis.
The clinical presentation of SOS includes hepatosplenomegaly (with resulting thrombocytopenia), jaundice, abdominal pain, weight gain with increasing ascites and encephalopathy. When haematologic patients are affected, these features may be similar to those of graft-versus-host disease, thus biopsy may be necessary to differentiate these conditions.
SOS is an established complication of bone marrow ablation therapy and stem cell transplantation, affecting up to 10-60% of patients. It also occurs with chemotherapy for solid tumours or long-term immunosuppression. Associated chemotherapy agents include oxaliplatin (54% of patients with metastatic colorectal carcinoma who undergo this therapy develop SOS), 5-fluorouracil, mercaptopurine, dacarbazine, cyclophosphamide and vincristine. It may present as early as 1 to 3 weeks after initiation of therapy.
US findings include ascites, gallbladder wall thickening, and hepatosplenomegaly as well as decreased flow through the portal vein on Doppler US. CT reveals similar findings, as well as perioesophageal varices, and recanalisation of the umbilical vein. Post-contrast enhancement CT and MR imaging commonly reveal patchy liver enhancement (mosaic appearance, usually located in the peripheral parenchyma of the right lobe) and narrowing of the main hepatic veins. On gadoxetic acid enhanced MRI, a reticular pattern is noted on hepatobiliary phase images. This finding is highly specific for correct diagnosis of hepatic SOS.
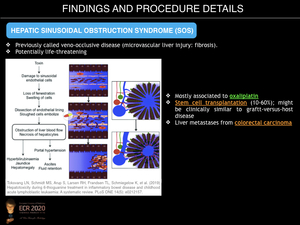
Fig. 7: Hepatic Sinusoidal Obstruction Syndrome (SOS)
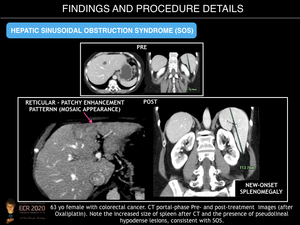
Fig. 8: Hepatic Sinusoidal Obstruction Syndrome (SOS)
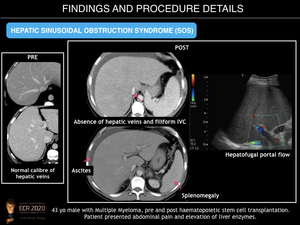
Fig. 9: Hepatic Sinusoidal Obstruction Syndrome (SOS)
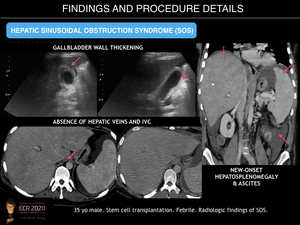
Fig. 10: Hepatic Sinusoidal Obstruction Syndrome (SOS)
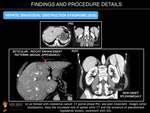
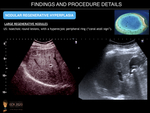
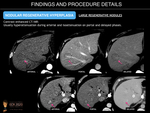
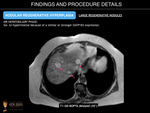
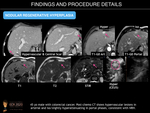
NODULAR REGENERATIVE HYPERPLASIA
Nodular hyperplasia is defined by the micronodular transformation of hepatic parenchyma. In contrast to cirrhosis, these nonneoplastic regenerative nodules are not surrounded by fibrous septa. This transformation may lead to compression and atrophy of the surrounding liver parenchyma, non-cirrhotic portal hypertension (including ascites, splenomegaly and varices) and increased postoperative morbidity.
Post-chemotherapy regenerative nodules include:
- Monoacinar regenerative nodules (nodular regenerative hyperplasia). Considered to be end-stage SOS, these are characterized by small-sized nodule formation. The sensitivity of multimodal imaging is low for the diagnosis of these lesions because of their small size, and at present, the final diagnosis is based on pathology.
- Multiacinar regenerative nodules (large regenerative nodules, LRN, or focal nodular hyperplasia (FNH)-like lesions). These can be detected on imaging because may be large (>5mm). US may reveal small, isoechoic round lesions, with a hyperechoic peripheral ring (“coral atoll sign”). LRN are isodense on unenhanced CT and can be hypo/iso or hyperintense on T2-weighted MRI images. On contrast-enhanced CT and MR imaging, they usually show bright and homogenous arterial contrast enhancement and isoattenuation to the surrounding liver parenchyma during the portal venous and delayed phases. A central scar is present in fewer than 50% of cases. FNH-like lesions are usually iso- to hyperintense during the hepatobiliary phase because of a similar or stronger OATP1B3 expression in the lesion than in the background liver. In addition, a ring or doughnut-like enhancement may be present in up to 50% of cases and is characterized by a hyperintense periphery and a hypointense centre.

Fig. 11: Nodular Regenerative Hyperplasia
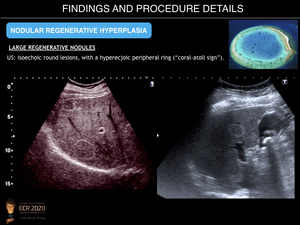
Fig. 12: Nodular Regenerative Hyperplasia
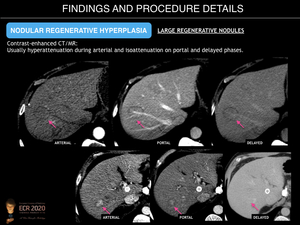
Fig. 13: Nodular Regenerative Hyperplasia
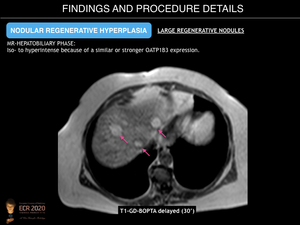
Fig. 14: Nodular Regenerative Hyperplasia
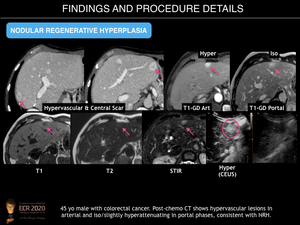
Fig. 15: Nodular Regenerative Hyperplasia
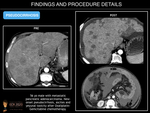
PSEUDOCIRRHOSIS
Pseudocirrhosis is defined as the development of diffuse hepatic nodularity, in patients following chemotherapy for hepatic metastases. While a potential cause of portal hypertension and liver failure, it does not show the major clinical features of cirrhosis.
It is most commonly found following chemotherapy with gemcitabine in patients treated for metastatic breast cancer (occurring in 75% of patients, with 9% of them showing signs of portal hypertension) and pancreatic cancer. It has also been reported with oxaliplatin therapy for metastatic colorectal carcinoma, and less commonly, in carcinoid tumour and Hodgkin disease.
The morphologic changes of the hepatic contour are initially detected between 4 and 8 months after therapy, may be diffuse or focal, and mimic liver cirrhosis, causing segmental volume loss, capsular and tumour tissue retraction and fibrosis, and enlargement of the caudate lobe. Additionally, the imaging findings of portal hypertension such as ascites, portosystemic collaterals, and splenomegaly may also be present.
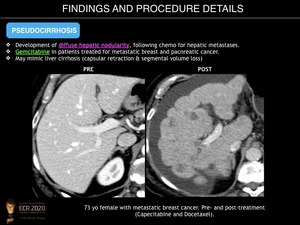
Fig. 16: Pseudocirrhosis
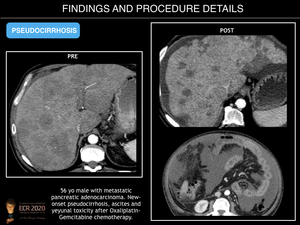
Fig. 17: Pseudocirrhosis
PORTAL THROMBOSIS
It is associated with FOLFIRI and bevacizumab therapy for treatment of colorectal cancer metastases. Bevacizumab decreases the healing capacity of endothelial cells, which causes bleeding and thrombosis. It is usually clinically silent.
At imaging, portal thrombosis is seen as a filling defect in the portal vein or its branches with subsequent wedge-shaped perfusion disorder area.
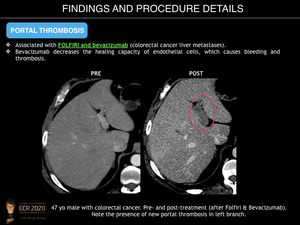
Fig. 18: Portal Thrombosis
Other patterns of chemo-induced hepatotoxicity include acute hepatitis, reactivation of hepatitis B and C and hepatic peliosis.
MANAGEMENT OF CHEMO-INDUCED HEPATOTOXICITY
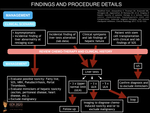
Optimal management of chemo-related hepatotoxicity depends on the clinical scenario, the agent in use and the severity of the imaging findings.
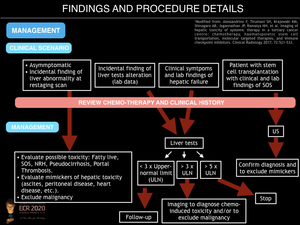
Fig. 20: Management of chemo-induced hepatotoxicity.