PET-MRI dimensional, morphological and functional characteristics of metastatic neck lymph nodes
The diagnosis of metastatic cervical lymph nodes is challenging and made even more difficult by the multiple criteria available. A normal sized lymph node can still be reported as metastatic if one or more of the other criteria are met. Suggested dimensional, morphological and functional criteria for the diagnosis of metastatic cervical lymph nodes are (Tab.1):
Short-axis diameter cutoff on MRI is 10 mm [3] except for lateral retropharyngeal nodes (VIIa) that have a 6 mm cutoff and submandibular (Ib) and upper jugular (II) groups that have a 15 mm cutoff; medial retropharyngeal lymph nodes are considered pathologic if detectable [4].
Three or more contiguous and confluent lymph nodes with borderline short-axis are highly suggestive of metastatic lymph node involvement, especially along the drainage chain of a primary tumor [5].
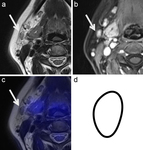
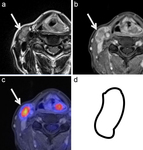
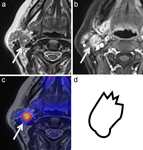
Lymph node borders can be: smooth (benign; Fig.1), lobulated (benign; Fig.2), spiculated (metastatic; Fig.3) or indistinct (metastatic; Fig.4)[6].
Round lymph node shape with a maximum longitudinal to maximum short axial diameter ratio (L/T ratio) ≤2 should be considered abnormal. However, this includes both benign and malignant causes for lymphadenopathy.
ENE is defined as the extension of metastatic cells through the nodal capsule into the perinodal tissue (Fig.4). Caution should be used when reporting ENE, assigning ENE-negative if there are doubts to avoid upstaging [7].
Signal intensity heterogeneity has a better performance of nodal metastases detection when evaluated on T2w images rather than on contrast-enhanced T1w (C+ T1W)[6](Fig.5).
Central necrosis has a high specificity [5](Fig.6). The differential diagnosis of an abscess formation is almost always made by the simultaneous presence of signs and symptoms of infection.
Cystic lymph nodes are usually harboring metastasis from HPV+ HNSCC [8].
According to a recent systematic review and meta-analysis, the median ADC cutoff value reported in the literature is 0.965x10 −3mm2/s [9].
Hypermetabolic lymph nodes with high focal uptake of 18F-FDG (SUVmax > 2.5) should be considered malignant independently of their size (Fig.7). False-negative findings may be caused by nodal necrosis; false-positive findings may be caused by inflammatory and infectious processes [5].
Pathologic lymph nodes should be then reported using neck level segmentation [10](Tab.2, Fig.8).
Role of PET-MRI for N staging of HNSCC: review of the literature
A comprehensive literature search of Pubmed, Web of Science, Cochrane, Embase and Scopus databases was conducted to find relevant articles on the role of PET-MRI for N staging of HNSCC, based on combinations and variants of “PET-MR” and “HNSCC” in the title/abstract/keyword fields. All studies published up to August 30th, 2019 were considered. We excluded non-English articles and publications that:
- did not assess the N staging;
- considered a post-processing fusion of PET and MRI data acquired without simultaneous or sequential PET-MR scanners;
- were editorials, letters, review articles, comments, author replies or case reports.
We found eight studies (381 total patients) that reported data about N staging in HNSCC using PET-MR (Tab.3). All of them concluded that PET-MR is at least comparable to PET-CT or MR alone for this purpose. Chan and Schlittenbauer reported a superior performance of PET-MR than PET-CT or MR for metastatic cervical lymph nodes detection. Other authors did not find statistically significant advantage PET-MR compared to PET-CT and MR (Kuhn and Schaarschmidt) or MR (Platzeck 2014).
Four authors (Chan, Schaarschmidt, Platzeck (2014) and Schlittenbauer) calculated sensitivity and specificity values on a per level basis. The sensitivity ranged from 81% (Schaarschmidt) to 100% (Schlittenbauer), while the specificity ranged from 95,2% (Chan) to 100% (Schlittenbauer).
The included studies were heterogeneous and reported several limitations, the main one being the small number of patients.
The patient selection criteria and the reference standards of these studies were different. Three studies evaluated patients that were staged or re-staged after treatment (Kubiessa, Kuhn and Schaarschmidt) while the others included only patients that were referred for staging. In four studies histopathological findings served as reference standard, whereas histopathology, clinical inspection and imaging follow-up were used alternatively as reference by Chan, Kubiessa and Kuhn et al; Platzek (2012) investigated the feasibility of HNSCC staging with PET-MR comparing the lymph nodes detected by PET-MR and PET dataset, thus not requiring histopathological confirmation.
Six studies were prospective while two were retrospective (Chan and Schaarschmidt).
All studies made the diagnosis of metastatic lymph node according to different dimensional, morphological and functional criteria (Tab.4).